Press release
Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and Related Profiles
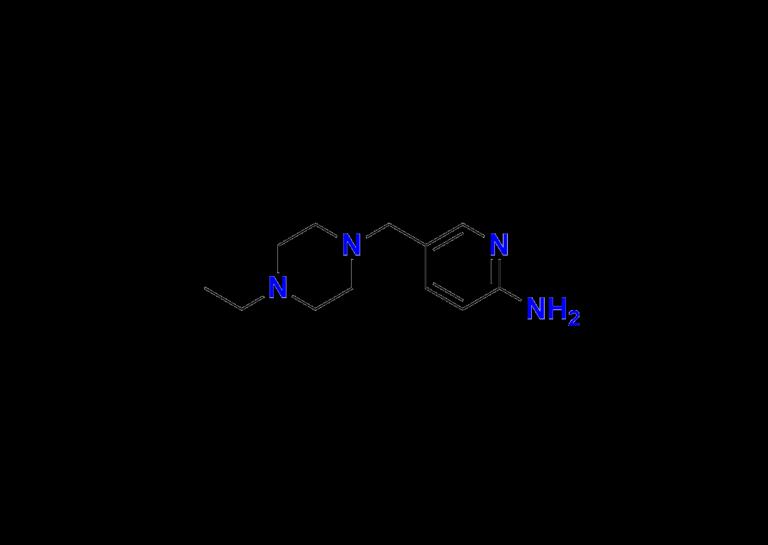
In the ever-evolving field of targeted cancer therapy, Abemaciclib has emerged as a pivotal agent in the treatment of hormone receptor-positive (HR+), HER2-negative advanced or metastatic breast cancer. As researchers and pharmaceutical developers continue to innovate in oncology, the importance of impurity profiling and the availability of reliable Abemaciclib impurity standards has never been greater.At the forefront of pharmaceutical impurity standards, Aquigen Bio is proud to support global manufacturers, CROs, and regulatory bodies with high-purity Abemaciclib Impurity 1, along with an expanding suite of related impurities including Abemaciclib Impurity 5, Abemaciclib Impurity 2, and Abemaciclib Nitroso Impurity 1.
Know more about Abemaciclib Impurity 1
https://aquigenbio.com/product/abemaciclib-impurity-1/
What Is Abemaciclib Impurity 1?
Abemaciclib Impurity 1 represents a critical reference material necessary for assessing the stability, efficacy, and safety of Abemaciclib-based formulations. This impurity, like others formed during synthesis or degradation, must be carefully monitored and quantified using validated analytical methods such as HPLC, LC-MS, or NMR spectroscopy.
The presence and profile of Impurity 1 serve as a fingerprint for the drug's purity level and manufacturing consistency. Regulatory authorities including the US FDA and EMA require stringent reporting and qualification of all relevant impurities, particularly those found at levels above identification thresholds.
At Aquigen Bio, our Abemaciclib Impurity 1 is offered with:
Purity >98% (HPLC)
Complete characterization (1H-NMR, 13C-NMR, MS, IR, COA)
Custom pack sizes and bulk availability
Immediate global dispatch and technical support
Why Are Abemaciclib Impurities Crucial for Compliance?
The demand for impurity standards is rising in tandem with stricter ICH Q3A/B guidelines. With oncology drugs being under heightened regulatory scrutiny, identifying and quantifying trace-level impurities is essential-not just for regulatory filings, but also for safeguarding patient safety.
Aquigen Bio's impurity solutions support:
Drug substance and drug product method validation
Stability testing and forced degradation studies
Qualification studies under ICH guidelines
ANDA/DMF/NDA regulatory submissions
Our analytical-grade Abemaciclib Impurity 1 serves as a cornerstone in these applications, enabling laboratories to meet global pharmacopoeial standards.
Expanding the Impurity Profile: Abemaciclib Impurity 5
Another key structural variant, Abemaciclib Impurity 5, arises as a process impurity or degradation product under certain stress conditions. It plays a critical role in the overall impurity mapping and qualification of Abemaciclib.
To know more about Abemaciclib Impurity 5
https://aquigenbio.com/product/abemaciclib-impurity-5/
Aquigen Bio offers Abemaciclib Impurity 5 with full spectral data and batch-specific documentation. Our scientific rigor ensures you get reproducible results for method development, setting a benchmark in both R&D and QC environments.
Whether you're developing a new formulation or optimizing a synthesis pathway, incorporating Abemaciclib Impurity 5 into your analytical protocols will elevate the reliability of your data and accelerate regulatory approval timelines.
Supporting Development Pipelines with Abemaciclib Impurity 2
Often encountered in intermediate stages of drug development, Abemaciclib Impurity 2 is another essential reference standard supplied by Aquigen Bio. While typically present in low concentrations, its accurate quantification is indispensable for demonstrating process robustness and ensuring the therapeutic integrity of the final API.
To know more about Abemaciclib Impurity 2
https://aquigenbio.com/product/abemaciclib-impurity-2/
Our impurity is available in milligram to gram-scale quantities with:
Traceable documentation
Fast turnaround times for custom orders
Compatibility with compendial and non-compendial testing methods
This impurity standard aids in the validation of precision assays, helping your quality control team detect even minute fluctuations in synthesis outcomes.
Navigating Genotoxic Risk: Abemaciclib Nitroso Impurity 1
Among the most concerning categories of pharmaceutical impurities are nitroso compounds, known for their potential genotoxicity and carcinogenic risk. In light of global recalls due to nitrosamine contamination, the accurate detection of such impurities is paramount.
To know more about Abemaciclib Nitroso Impurity 1
https://aquigenbio.com/product/abemaciclib-nitroso-impurity-1/
Abemaciclib Nitroso Impurity 1 exemplifies this risk category.
Aquigen Bio provides this impurity standard with careful consideration of its sensitive handling and characterization.
Our offering includes:
Impurity profiling by high-resolution mass spectrometry
Genotoxic risk categorization support
Guidance on LOQ/LOD for nitroso impurities
Compliance with ICH M7 (R1) and regulatory expectations
We partner with pharmaceutical clients to develop robust risk assessment protocols for nitrosamine impurities, especially in oncology drugs where even nanogram-level contamination is unacceptable.
Why Aquigen Bio?
Aquigen Bio has emerged as a trusted provider of pharmaceutical impurity reference standards, catering to innovators, generics, and research organizations globally. With a product suite that now includes Abemaciclib Impurity 1, 2, 5, and Nitroso Impurity 1, we are empowering precision in analytical workflows.
Our differentiators:
In-house synthesis under GMP-analogue conditions
ISO-certified QA/QC protocols
Responsive customer support and technical documentation
Compatibility with US FDA, EMA, and CDSCO regulatory filings
Our commitment to purity, compliance, and reliability is unmatched. Whether you're looking for milligram quantities for early-stage discovery or gram-scale lots for commercial filings, Aquigen Bio is your trusted source.
Partnering for Future-Proof Oncology Development
As the oncology pharmaceutical pipeline continues to evolve, the need for advanced impurity standards will intensify. Regulatory bodies are calling for deeper impurity profiling, better documentation, and proactive risk mitigation.
By choosing Aquigen Bio as your impurity standards partner, you're investing in the future of precision drug development. Our Abemaciclib impurity range, including Impurity 1, Impurity 2, Impurity 5, and Nitroso Impurity 1, enables you to maintain quality benchmarks at every stage of the drug lifecycle-from discovery to market launch.
Let us support your journey to regulatory success and scientific excellence.
Contact:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio
Aquigen Bio is an emerging leader in the field of pharmaceutical reference standards and impurity solutions. With a robust catalog of API impurities, degradants, metabolites, and working standards, the company supports pharmaceutical and biopharmaceutical clients in meeting quality, safety, and regulatory expectations.
By combining scientific expertise, advanced analytical infrastructure, and a global mindset, Aquigen Bio enables the pharmaceutical industry to develop safer, more effective drugs with confidence and speed.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and Related Profiles here
News-ID: 4051132 • Views: …
More Releases from Aquigen Biosciences
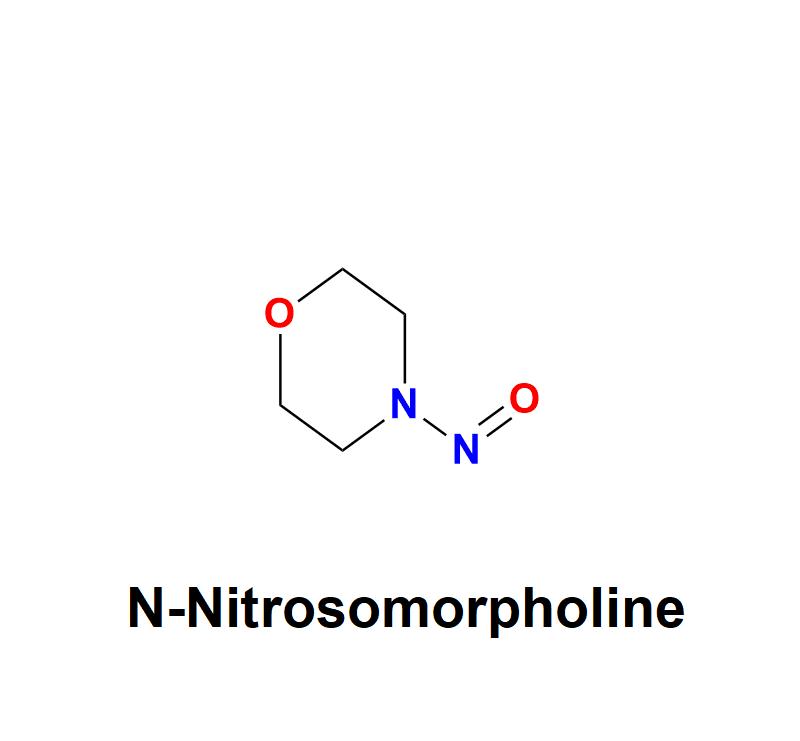
N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bi …
N-Nitrosomorpholine, a compound belonging to the nitrosamine family, has garnered significant attention in the pharmaceutical and healthcare industries due to its potential carcinogenic risks. This chemical impurity, often found as a byproduct in manufacturing processes, poses serious challenges to drug safety and human health, necessitating stringent monitoring and control measures from pharmaceutical companies.
Learn more about N-Nitrosomorpholine: https://aquigenbio.com/product/n-nitrosomorpholine/
Understanding N-Nitrosomorpholine:
N-Nitrosomorpholine is a nitrosamine impurity characterized by its chemical structure, which includes…
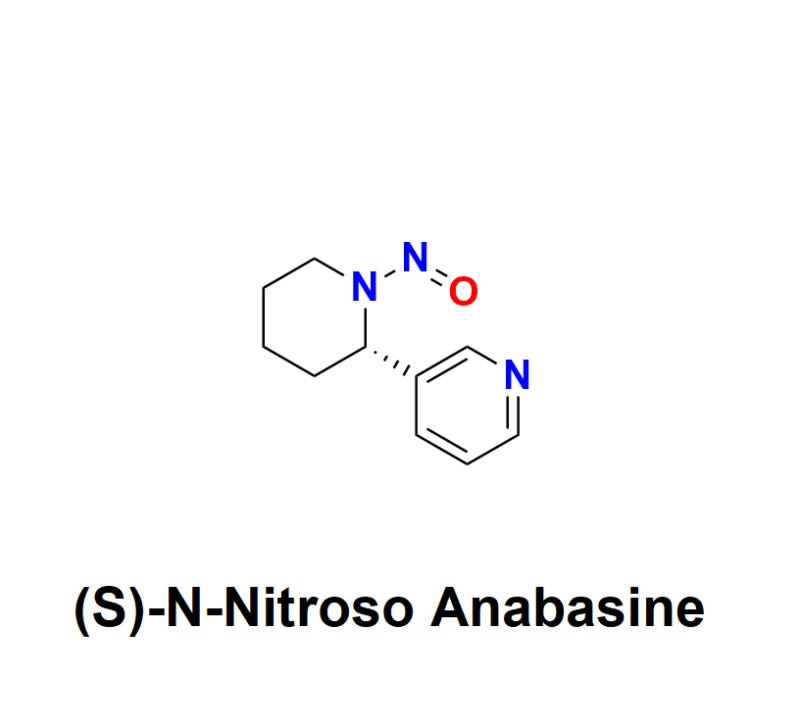
(S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How …
(S)-N-Nitroso Anabasine, a nitrosamine impurity, has raised significant safety concerns within the pharmaceutical industry. Recognized as a probable human carcinogen, this impurity has become a focal point for global regulatory agencies and manufacturers alike, urging a renewed emphasis on detection, prevention, and management.
Learn more about (S)-N-Nitroso Anabasine: https://aquigenbio.com/product/s-n-nitroso-anabasine/
What Is (S)-N-Nitroso Anabasine?
(S)-N-Nitroso Anabasine belongs to the family of nitrosamines, compounds formed through a chemical reaction known as nitrosation. This…
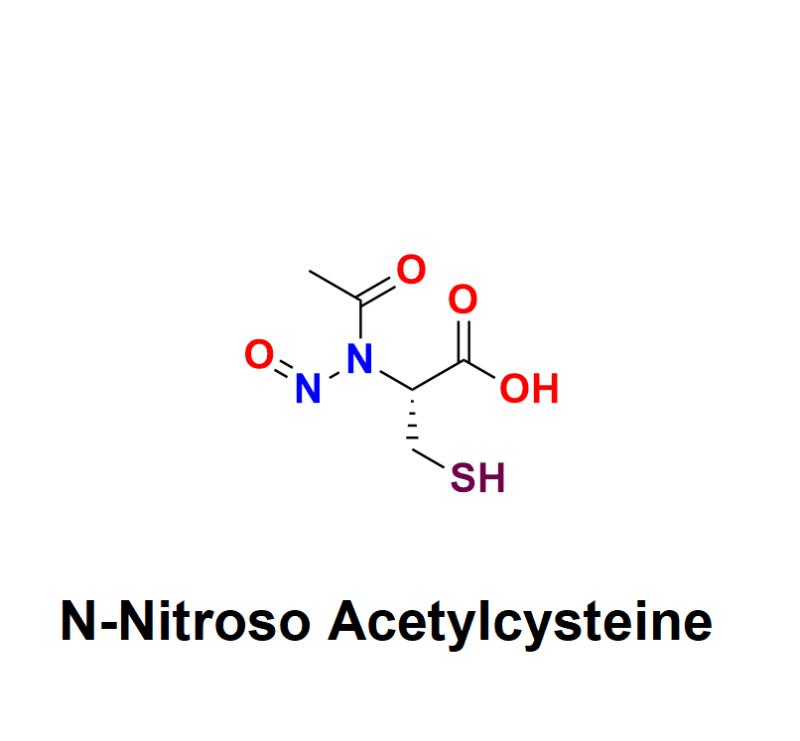
N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Haza …
N-Nitroso Acetylcysteine has emerged as a critical topic of concern in the pharmaceutical industry. As a member of the nitrosamine family, it is a potential impurity that poses significant health risks, including carcinogenicity, even in trace amounts. With increasing regulatory scrutiny on nitrosamine impurities, pharmaceutical manufacturers must address the presence of compounds like N-Nitroso Acetylcysteine to protect public health and ensure compliance with global standards.
Learn more about N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/…
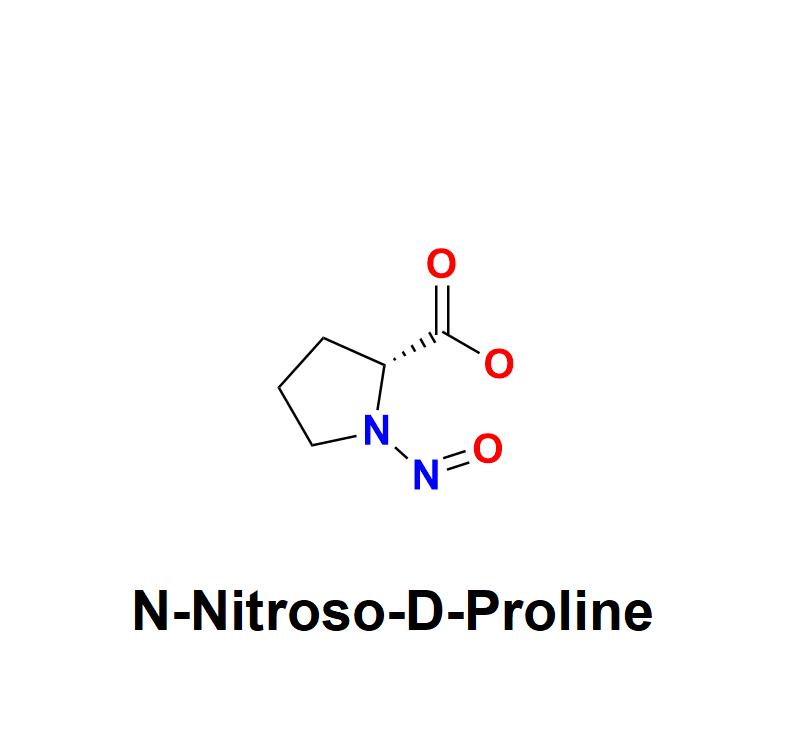
N-Nitroso-D-Proline: A Growing Concern for Pharmaceutical Safety - Explore How A …
N-Nitroso-D-Proline, a chemical impurity belonging to the nitrosamine family, has emerged as a significant concern for pharmaceutical manufacturers worldwide. Its presence in drugs, even at trace levels, poses potential risks to human health due to its genotoxic and carcinogenic properties. As regulatory bodies intensify scrutiny, the pharmaceutical industry must prioritize its detection, mitigation, and prevention to ensure the safety and efficacy of medications.
Learn more about N-Nitroso-D-Proline: https://aquigenbio.com/product/n-nitroso-d-proline/
Understanding N-Nitroso-D-Proline:
N-Nitroso-D-Proline is…
More Releases for Impurity
Tizanidine Nitroso Impurity 1 - High-Purity Reference Standard for Reliable Impu …
In the highly regulated world of pharmaceutical manufacturing, impurity profiling plays a critical role in ensuring drug safety, efficacy, and regulatory compliance. Among the wide range of impurity reference standards required during API development and validation, Tizanidine Nitroso Impurity 1 stands out as a vital compound for researchers and manufacturers working on Tizanidine-based formulations.
At Aquigen Bio, we are proud to offer Tizanidine Nitroso Impurity 1, a high-purity reference standard meticulously…
AquigenBio Launches High-Purity EP Impurity Standards for Propylthiouracil, Incl …
AquigenBio is pleased to announce the availability of two essential impurity reference standards-Propylthiouracil EP Impurity A and Propylthiouracil EP Impurity B. These impurity standards are now available to support pharmaceutical companies, contract research organizations, and analytical laboratories engaged in drug development, quality control, and regulatory compliance.
Propylthiouracil (PTU) is a well-known antithyroid medication, widely used in the treatment of hyperthyroidism. In line with pharmacopeial guidelines, it is critical to identify and…
Precision in Impurity Profiling: Aquigen Bio Introduces High-Purity Doxorubicin …
Pune, India - July 2025 - As regulatory standards tighten across the pharmaceutical industry, the need for accurate, high-purity reference standards has never been more critical. Aquigen Bio, a trusted provider of pharmaceutical reference materials, is proud to announce the availability of Doxorubicin Dimer Impurity 3, a key impurity used in the impurity profiling of the widely used chemotherapeutic agent, Doxorubicin.
This launch is part of Aquigen Bio's expanded portfolio of…
Breaking Ground in Pharmaceutical Impurity Standards: Aquigen Bio Expands Abirat …
In the evolving landscape of pharmaceutical development and quality control, the identification, synthesis, and supply of high-purity impurity standards play a pivotal role. One of the most significant advancements in recent times is the availability of specialized Abiraterone-related impurity standards-essential tools for characterizing drug formulations and ensuring regulatory compliance.
Manufactured and supplied by Aquigen BioSciences, a trusted leader in impurity synthesis and reference standards, these impurity standards offer analytical laboratories and…
Unlock Advanced Dabigatran Research with High-Purity Standards: Now Available - …
In the ever-evolving pharmaceutical landscape, precision, safety, and regulatory compliance have never been more crucial. With Dabigatran etexilate mesylate becoming a cornerstone in the management of thromboembolic disorders, the demand for high-purity standards and robust impurity profiling has surged. Recognizing this essential need, a new suite of advanced analytical tools for Dabigatran and its associated impurities has been made available to support the global scientific and quality control community.
To kno…
Aquigen Bio Expands Oncology Impurity Standards Portfolio with High-Purity Etopo …
India - 07/05/2025 - Aquigen Bio, a leading provider of pharmaceutical impurity reference standards, proudly announces the launch of its latest high-quality product: Etoposide EP Impurity A. This new addition to Aquigen's growing portfolio of oncology-related impurities is tailored to meet the stringent requirements of pharmaceutical manufacturers and analytical laboratories focused on drug safety, efficacy, and compliance.
To know more about : Etoposide EP Impurity A
https://aquigenbio.com/product/etoposide-ep-impurity-a/
A Strategic Addition to Oncology Drug…