Press release
N-Nitroso-D-Proline: A Growing Concern for Pharmaceutical Safety - Explore How Aquigen Bio Sciences Leads the Way in Impurity Detection, Risk Mitigation, and Regulatory Compliance with Advanced Solutions for Safer and More Reliable Medications Worldwide
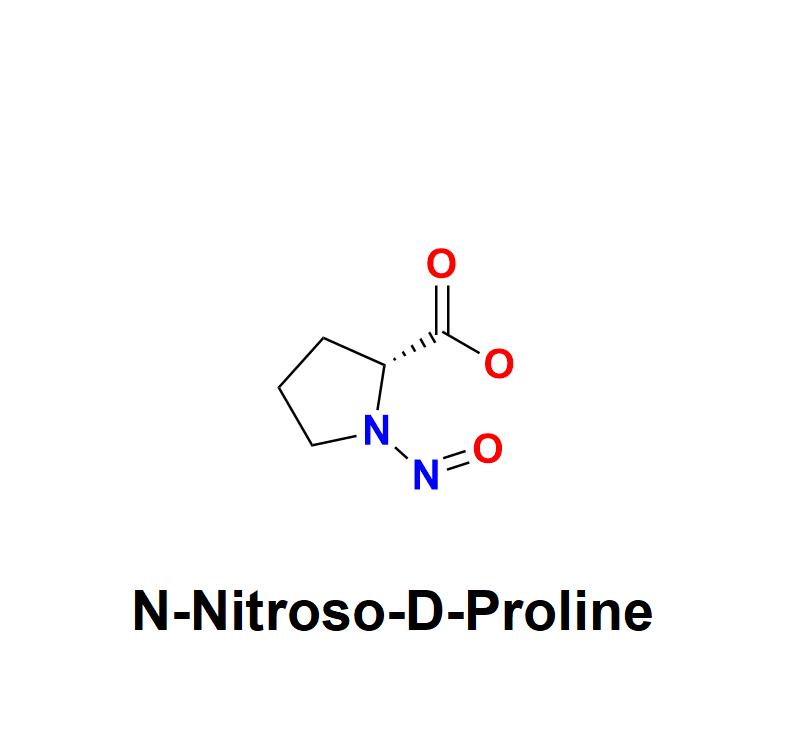
N-Nitroso-D-Proline, a chemical impurity belonging to the nitrosamine family, has emerged as a significant concern for pharmaceutical manufacturers worldwide. Its presence in drugs, even at trace levels, poses potential risks to human health due to its genotoxic and carcinogenic properties. As regulatory bodies intensify scrutiny, the pharmaceutical industry must prioritize its detection, mitigation, and prevention to ensure the safety and efficacy of medications.Learn more about N-Nitroso-D-Proline: https://aquigenbio.com/product/n-nitroso-d-proline/
Understanding N-Nitroso-D-Proline:
N-Nitroso-D-Proline is a nitrosamine impurity derived from proline, an amino acid commonly found in pharmaceutical formulations. Nitrosamines are formed when nitrites react with secondary or tertiary amines under certain conditions, such as during the manufacturing process or long-term storage of drugs. While the presence of nitrosamines like N-Nitroso-D-Proline is often inadvertent, their potential to cause harm has made them a focal point in pharmaceutical quality control.
Hazards of N-Nitroso-D-Proline:
The primary concern surrounding N-Nitroso-D-Proline lies in its carcinogenic potential. Nitrosamines are classified by the International Agency for Research on Cancer (IARC) as probable human carcinogens. Although the level of risk depends on factors such as concentration and exposure duration, even minimal amounts of N-Nitroso-D-Proline in drugs can accumulate over time, increasing the likelihood of adverse health outcomes.
Key risks associated with N-Nitroso-D-Proline include:
1. DNA Damage: The compound's genotoxic nature enables it to interact with DNA, causing mutations that may lead to cancer.
2. Organ Toxicity: Prolonged exposure can result in toxic effects on vital organs such as the liver and kidneys.
3. Regulatory Non-Compliance: Drugs contaminated with nitrosamines, including N-Nitroso-D-Proline, risk regulatory recalls, which can damage a company's reputation and result in financial losses.
Why Pharmaceutical Companies Must Pay Attention:
As public health concerns over nitrosamines intensify, regulatory authorities such as the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have introduced stringent guidelines for their control in pharmaceutical products. These include limits on permissible daily exposure levels and mandatory testing for nitrosamine impurities during drug development and manufacturing.
For pharmaceutical companies, the stakes are high. Failure to address N-Nitroso-D-Proline contamination can lead to regulatory sanctions, legal liabilities, and compromised patient trust. Moreover, as the industry transitions towards more sustainable manufacturing practices, ensuring nitrosamine-free drugs is a critical step in safeguarding public health and maintaining compliance with evolving standards.
Get in Touch with Us - https://aquigenbio.com/contact-us/
Strategies for Managing N-Nitroso-D-Proline Risks:
Proactive measures can help pharmaceutical companies mitigate the risks associated with N-Nitroso-D-Proline and similar impurities. These strategies include:
1. Enhanced Analytical Testing: Advanced analytical techniques such as liquid chromatography-mass spectrometry (LC-MS) and gas chromatography-mass spectrometry (GC-MS) are essential for detecting trace levels of N-Nitroso-D-Proline in raw materials, intermediates, and finished products.
2. Risk Assessments: Conducting thorough risk assessments during product development can identify potential sources of nitrosamine formation, allowing manufacturers to implement preventive measures.
3. Supplier Audits: Ensuring the quality and consistency of raw materials through rigorous supplier evaluations can minimize the likelihood of contamination.
4. Process Optimization: Modifying manufacturing processes to reduce nitrosating conditions, such as controlling pH and temperature, can help prevent the formation of nitrosamines.
5. Continuous Monitoring: Establishing robust quality assurance protocols and monitoring systems ensures ongoing compliance with regulatory requirements.
Collaboration and Innovation are Key:
The fight against nitrosamine impurities like N-Nitroso-D-Proline requires a collaborative effort across the pharmaceutical industry. Partnerships with regulatory agencies, research organizations, and impurity standards providers are essential for developing innovative solutions and maintaining a proactive approach to quality control.
Conclusion:
As the pharmaceutical industry navigates the complexities of nitrosamine control, N-Nitroso-D-Proline stands as a pressing challenge that demands immediate and sustained attention. Addressing this impurity is not merely a regulatory requirement but a moral obligation to ensure patient safety and uphold the integrity of healthcare products.
For companies seeking comprehensive solutions to detect, analyze, and mitigate N-Nitroso-D-Proline, Aquigen Bio Sciences emerges as a trusted partner. With its expertise in impurity standards and commitment to innovation, Aquigen Bio Sciences offers advanced analytical tools, customized solutions, and technical support tailored to meet the unique needs of pharmaceutical manufacturers. By partnering with Aquigen Bio Sciences, companies can confidently navigate regulatory complexities, enhance product quality, and contribute to a safer future for patients worldwide.
The journey to eradicating nitrosamines like N-Nitroso-D-Proline starts with informed decisions and reliable resources-steps that Aquigen Bio Sciences is proud to facilitate as a leader in impurity management solutions. For more information, contact Aquigen Bio Sciences today.
Similar Trending Products:
1) N-Nitroso Paracetamol: https://aquigenbio.com/product/n-nitroso-paracetamol-2/
2) Vonoprazan Nitroso Impurity: https://aquigenbio.com/product/vonoprazan-nitroso-impurity/
3) N-Nitroso N-Desmethyl Olanzapine: https://aquigenbio.com/product/n-nitroso-n-desmethyl-olanzapine-2/
Contact Us:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences:
Aquigen Bio Sciences is a renowned contract research organization based in Pune, India, specializing in impurity standards and comprehensive solutions for the pharmaceutical industry. With a focus on advancing pharmaceutical safety and compliance, Aquigen provides expert guidance on identifying, quantifying, and managing impurities in various drug formulations. Their commitment to quality and regulatory excellence positions them as a trusted partner for manufacturers striving to meet stringent safety standards and improve patient outcomes.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release N-Nitroso-D-Proline: A Growing Concern for Pharmaceutical Safety - Explore How Aquigen Bio Sciences Leads the Way in Impurity Detection, Risk Mitigation, and Regulatory Compliance with Advanced Solutions for Safer and More Reliable Medications Worldwide here
News-ID: 3764181 • Views: …
More Releases from Aquigen Biosciences
Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and …
In the ever-evolving field of targeted cancer therapy, Abemaciclib has emerged as a pivotal agent in the treatment of hormone receptor-positive (HR+), HER2-negative advanced or metastatic breast cancer. As researchers and pharmaceutical developers continue to innovate in oncology, the importance of impurity profiling and the availability of reliable Abemaciclib impurity standards has never been greater.
At the forefront of pharmaceutical impurity standards, Aquigen Bio is proud to support global manufacturers, CROs,…
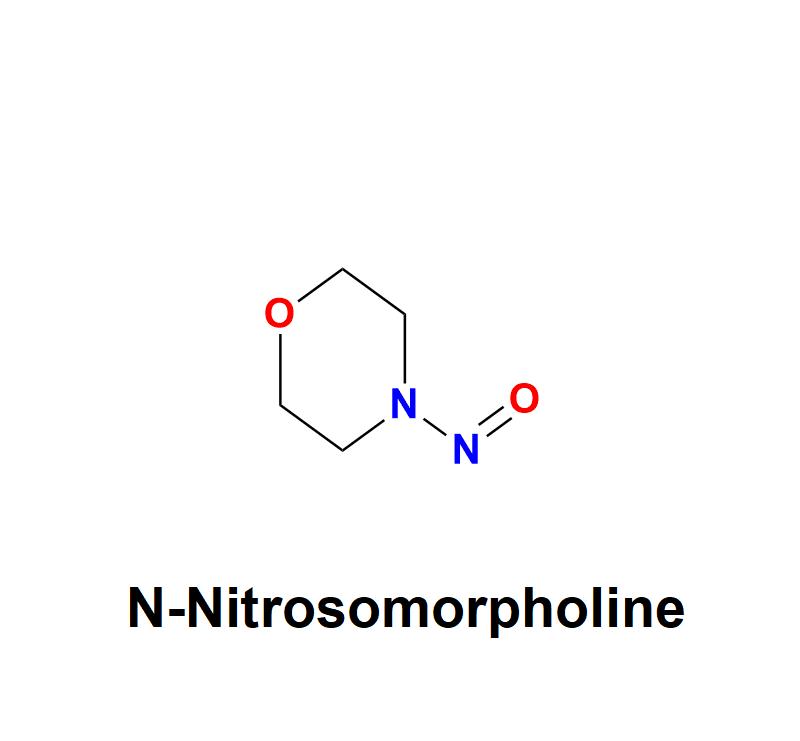
N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bi …
N-Nitrosomorpholine, a compound belonging to the nitrosamine family, has garnered significant attention in the pharmaceutical and healthcare industries due to its potential carcinogenic risks. This chemical impurity, often found as a byproduct in manufacturing processes, poses serious challenges to drug safety and human health, necessitating stringent monitoring and control measures from pharmaceutical companies.
Learn more about N-Nitrosomorpholine: https://aquigenbio.com/product/n-nitrosomorpholine/
Understanding N-Nitrosomorpholine:
N-Nitrosomorpholine is a nitrosamine impurity characterized by its chemical structure, which includes…
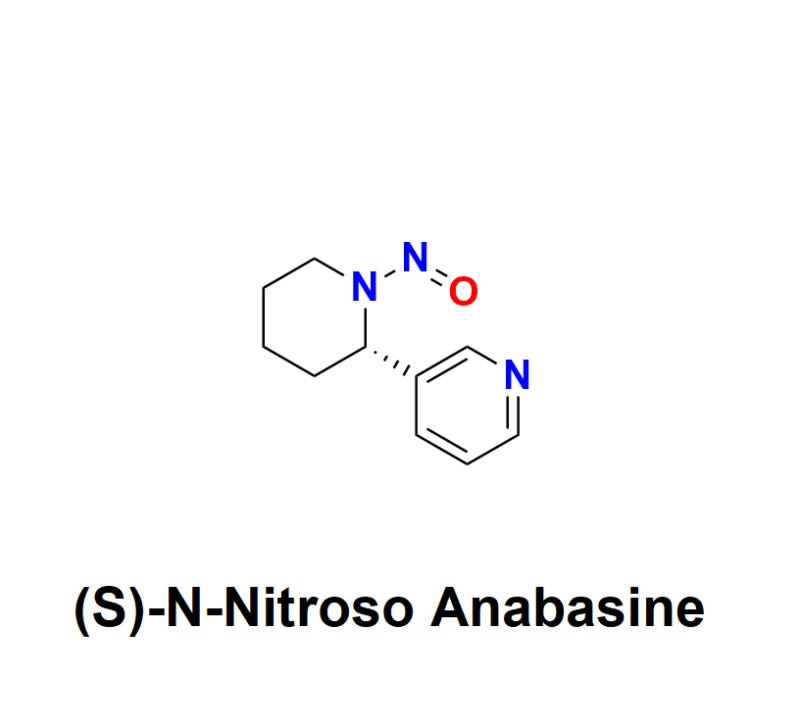
(S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How …
(S)-N-Nitroso Anabasine, a nitrosamine impurity, has raised significant safety concerns within the pharmaceutical industry. Recognized as a probable human carcinogen, this impurity has become a focal point for global regulatory agencies and manufacturers alike, urging a renewed emphasis on detection, prevention, and management.
Learn more about (S)-N-Nitroso Anabasine: https://aquigenbio.com/product/s-n-nitroso-anabasine/
What Is (S)-N-Nitroso Anabasine?
(S)-N-Nitroso Anabasine belongs to the family of nitrosamines, compounds formed through a chemical reaction known as nitrosation. This…
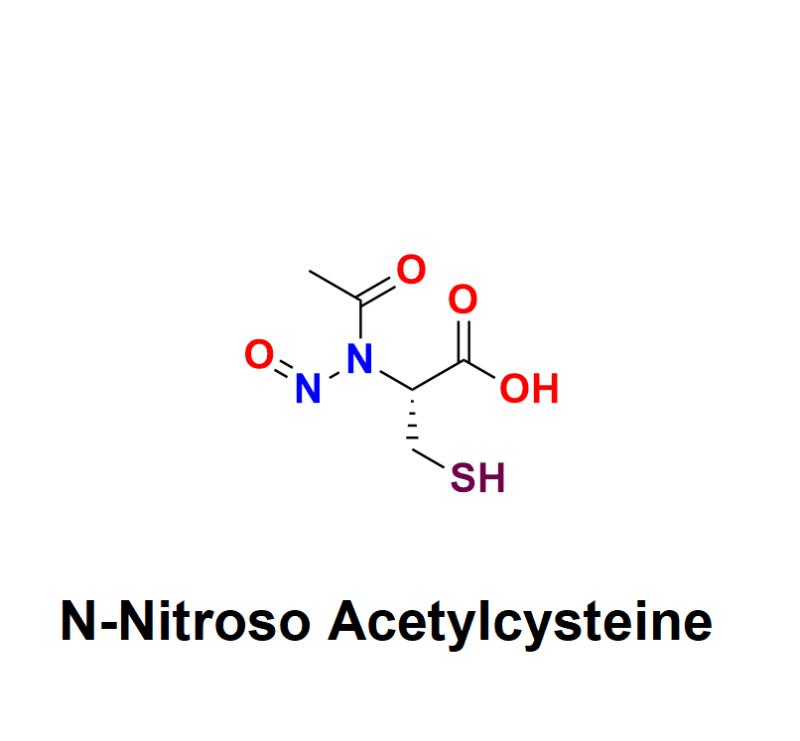
N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Haza …
N-Nitroso Acetylcysteine has emerged as a critical topic of concern in the pharmaceutical industry. As a member of the nitrosamine family, it is a potential impurity that poses significant health risks, including carcinogenicity, even in trace amounts. With increasing regulatory scrutiny on nitrosamine impurities, pharmaceutical manufacturers must address the presence of compounds like N-Nitroso Acetylcysteine to protect public health and ensure compliance with global standards.
Learn more about N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/…
More Releases for Nitroso
Riociguat N-Nitroso Des Formyl Impurity - Reliable Reference Standard for Advanc …
Riociguat N-Nitroso Des Formyl Impurity is a premium-quality analytical reference material developed to support research, testing, and quality control in the pharmaceutical industry.
As regulatory guidelines tighten worldwide, accurate detection, quantification, and control of nitroso impurities have become an essential part of drug development and manufacturing.
This compound plays a vital role in ensuring that formulations containing Riociguat meet the highest safety and compliance standards.
The Growing Importance of Nitroso Impurity…
Tizanidine Nitroso Impurity 1 - High-Purity Reference Standard for Reliable Impu …
In the highly regulated world of pharmaceutical manufacturing, impurity profiling plays a critical role in ensuring drug safety, efficacy, and regulatory compliance. Among the wide range of impurity reference standards required during API development and validation, Tizanidine Nitroso Impurity 1 stands out as a vital compound for researchers and manufacturers working on Tizanidine-based formulations.
At Aquigen Bio, we are proud to offer Tizanidine Nitroso Impurity 1, a high-purity reference standard meticulously…
Aquigen Bio Unveils N-Nitroso Felodipine: A New Benchmark in Pharmaceutical Refe …
In an era where drug safety and efficacy are paramount, the pharmaceutical industry faces increasing pressure to develop robust analytical methods that ensure the purity and quality of medicinal products. N-Nitroso Felodipine is specifically engineered to address these challenges. It serves as an indispensable tool for Analytical Method Development (AMD) and Analytical Method Validation (AMV), providing a reliable benchmark against which new and existing analytical procedures can be assessed for…
N-Nitroso Impurities: Carcinogenic Risks, Analytical Challenges, and Compliance …
N-Nitroso impurities are emerging as a significant concern in the pharmaceutical industry due to their potential carcinogenicity. These compounds, formed during the manufacturing or storage of drug products, have raised alarms among regulatory agencies across the globe. Pharmaceutical companies face mounting pressure to identify, analyze, and mitigate these impurities through stringent guidelines to ensure patient safety.
The serious risks posed by N-Nitroso compounds stem from their ability to induce genetic mutations…
Bumetanide Nitroso Impurity: Mitigating Compliance Risks and Integrating Innovat …
Bumetanide Nitroso Impurity has emerged as a significant concern in the pharmaceutical industry, particularly for companies involved in the development and manufacturing of medications used to manage edema. As awareness grows about the potential risks associated with nitroso impurities, regulatory agencies worldwide are increasing scrutiny over pharmaceutical products. This press release aims to address the challenges posed by Bumetanide Nitroso Impurity, outline the regulatory landscape, and highlight innovative practices that…
Sumatriptan Nitroso Impurity: Navigating Compliance Challenges and Implementing …
Sumatriptan Nitroso Impurity has become a pressing concern in the pharmaceutical industry, as companies grapple with significant compliance challenges related to these potentially harmful contaminants. As Sumatriptan, a widely prescribed medication for migraine relief, comes under increasing scrutiny from regulatory bodies, pharmaceutical companies must navigate a complex landscape to ensure product safety and adhere to evolving regulatory standards.
What is the issue?
Nitroso impurities have emerged as a pressing concern within the…