Press release
N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Hazards, Regulatory Challenges, and How Aquigen Bio Sciences Offers Advanced Solutions for Impurity Detection, Risk Mitigation, and Ensuring Compliance with Global Standards
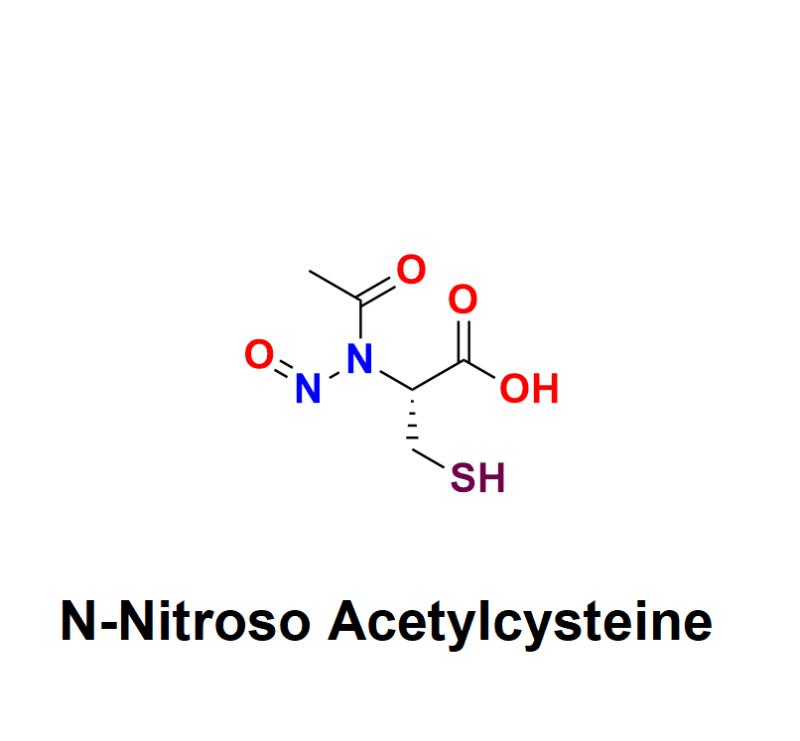
N-Nitroso Acetylcysteine has emerged as a critical topic of concern in the pharmaceutical industry. As a member of the nitrosamine family, it is a potential impurity that poses significant health risks, including carcinogenicity, even in trace amounts. With increasing regulatory scrutiny on nitrosamine impurities, pharmaceutical manufacturers must address the presence of compounds like N-Nitroso Acetylcysteine to protect public health and ensure compliance with global standards.Learn more about N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/
What is N-Nitroso Acetylcysteine?
N-Nitroso Acetylcysteine is a nitrosamine impurity that can form during the production of acetylcysteine-based medications. Acetylcysteine is widely used for its therapeutic benefits, such as treating respiratory conditions and acetaminophen overdose. However, the interaction of nitrosating agents and acidic environments during manufacturing can lead to the formation of N-Nitroso Acetylcysteine, potentially contaminating the final pharmaceutical product.
Health Hazards of N-Nitroso Acetylcysteine:
Nitrosamines, including N-Nitroso Acetylcysteine, are classified as probable human carcinogens due to their ability to cause DNA damage. Long-term exposure, even at low levels, may increase the risk of cancers such as gastric, oesophagal, and liver cancers. The stability of these compounds makes them persistent contaminants that can accumulate in the body, intensifying their harmful effects.
The presence of N-Nitroso Acetylcysteine in pharmaceutical products is especially concerning as it undermines the safety and efficacy of medications meant to improve health. Given its potential to go undetected without robust testing, the risks extend beyond patient safety to regulatory non-compliance, product recalls, and loss of consumer trust.
Regulatory Spotlight on Nitrosamine Impurities:
In response to the growing awareness of nitrosamine impurities, global regulatory bodies have implemented stringent guidelines to control and limit their presence in pharmaceuticals.
1. The U.S. FDA requires pharmaceutical companies to conduct risk assessments, implement control strategies, and ensure that nitrosamine levels in their products remain below the Acceptable Daily Intake (ADI) limits.
2. The European Medicines Agency (EMA) has issued similar guidance, emphasizing the adoption of analytical methods to detect and quantify nitrosamines, including N-Nitroso Acetylcysteine.
3. Indian regulators, too, are actively enforcing stringent monitoring of impurities in pharmaceuticals to ensure patient safety.
These frameworks underscore the urgent need for manufacturers to reassess their production processes, mitigate contamination risks, and adopt advanced testing technologies to comply with the evolving standards.
Get in Touch with Us - https://aquigenbio.com/contact-us/
Challenges in Managing N-Nitroso Acetylcysteine:
Detecting and mitigating the risks associated with N-Nitroso Acetylcysteine is not without challenges. Pharmaceutical companies face the following hurdles:
1. Complex Manufacturing Processes: The formation of nitrosamines often occurs due to specific conditions in manufacturing that may not be immediately apparent. Identifying and eliminating these conditions requires thorough process reviews.
2. Limitations of Conventional Testing Methods: Traditional analytical tools may not have the sensitivity required to detect trace levels of N-Nitroso Acetylcysteine, necessitating advanced techniques such as high-resolution mass spectrometry.
3. Supply Chain Risks: Contamination can occur at any stage, from raw material sourcing to final product formulation. Ensuring the purity of raw materials and maintaining stringent quality checks across the supply chain is essential.
4. Regulatory Complexity: The dynamic nature of regulations across regions requires pharmaceutical companies to stay updated and adapt quickly to new compliance requirements.
Why Proactive Measures are crucial:
The presence of nitrosamines like N-Nitroso Acetylcysteine is more than a regulatory challenge - it represents a fundamental responsibility of pharmaceutical companies to prioritize patient safety. Several high-profile drug recalls due to nitrosamine contamination have already demonstrated the far-reaching consequences of failing to address these impurities.
Proactive measures include:
1. Process Optimization: Reviewing and refining manufacturing methods to eliminate conditions conducive to nitrosation.
2. Comprehensive Risk Assessments: Identifying potential sources of contamination and implementing control measures.
3. Cutting-Edge Analytical Testing: Employing advanced methods such as gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-mass spectrometry (LC-MS) to detect and quantify nitrosamines accurately.
4. Collaborative Solutions: Partnering with specialized impurity analysis providers for expert guidance and reliable solutions.
Conclusion:
As the pharmaceutical industry grapples with the complexities of nitrosamine impurities, Aquigen Bio Sciences stands as a trusted ally. Specializing in impurity standards and advanced analytical solutions, Aquigen Bio Sciences provides the expertise and resources necessary to detect, analyze, and manage N-Nitroso Acetylcysteine and other nitrosamines.
With a commitment to innovation, regulatory compliance, and patient safety, Aquigen Bio Sciences empowers pharmaceutical companies to navigate the challenges of impurity control confidently. By leveraging their cutting-edge technologies and industry expertise, manufacturers can ensure the integrity of their products and uphold the trust of healthcare providers and patients alike. For more information on managing nitrosamine impurities, contact Aquigen Bio Sciences.
Similar Trending Products:
1) N-Nitroso Linagliptin Impurity: https://aquigenbio.com/product/n-nitroso-linagliptin-impurity-2/
2) N-Nitroso S-Mirabegron: https://aquigenbio.com/product/n-nitroso-s-mirabegron/
3) N-nitroso (S,S)-formoterol: https://aquigenbio.com/product/n-nitroso-ss-formoterol/
Contact Us:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences:
Aquigen Bio Sciences is a renowned contract research organization based in Pune, India, specializing in impurity standards and comprehensive solutions for the pharmaceutical industry. With a focus on advancing pharmaceutical safety and compliance, Aquigen provides expert guidance on identifying, quantifying, and managing impurities in various drug formulations. Their commitment to quality and regulatory excellence positions them as a trusted partner for manufacturers striving to meet stringent safety standards and improve patient outcomes.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Hazards, Regulatory Challenges, and How Aquigen Bio Sciences Offers Advanced Solutions for Impurity Detection, Risk Mitigation, and Ensuring Compliance with Global Standards here
News-ID: 3764233 • Views: …
More Releases from Aquigen Biosciences
Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and …
In the ever-evolving field of targeted cancer therapy, Abemaciclib has emerged as a pivotal agent in the treatment of hormone receptor-positive (HR+), HER2-negative advanced or metastatic breast cancer. As researchers and pharmaceutical developers continue to innovate in oncology, the importance of impurity profiling and the availability of reliable Abemaciclib impurity standards has never been greater.
At the forefront of pharmaceutical impurity standards, Aquigen Bio is proud to support global manufacturers, CROs,…
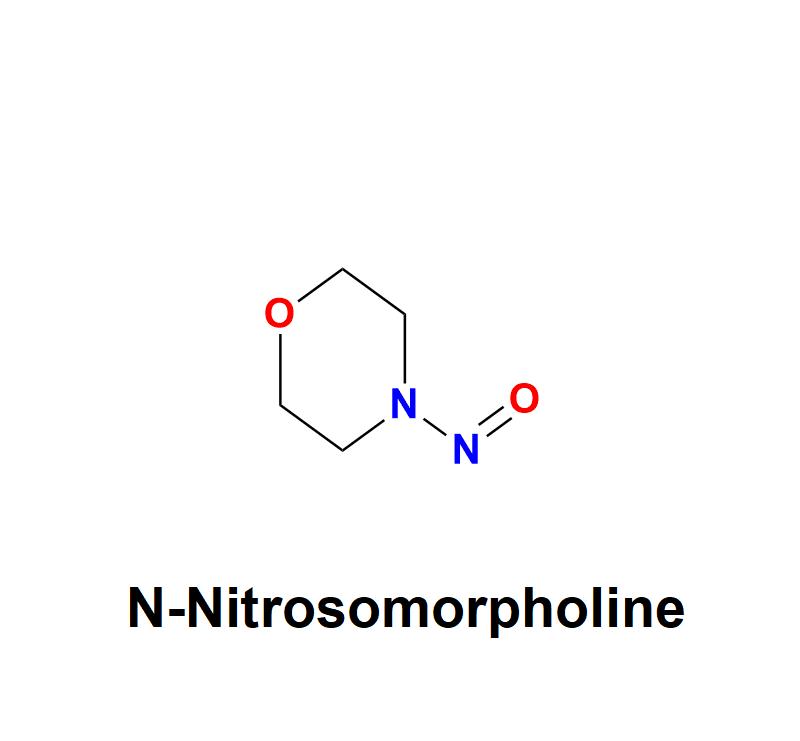
N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bi …
N-Nitrosomorpholine, a compound belonging to the nitrosamine family, has garnered significant attention in the pharmaceutical and healthcare industries due to its potential carcinogenic risks. This chemical impurity, often found as a byproduct in manufacturing processes, poses serious challenges to drug safety and human health, necessitating stringent monitoring and control measures from pharmaceutical companies.
Learn more about N-Nitrosomorpholine: https://aquigenbio.com/product/n-nitrosomorpholine/
Understanding N-Nitrosomorpholine:
N-Nitrosomorpholine is a nitrosamine impurity characterized by its chemical structure, which includes…
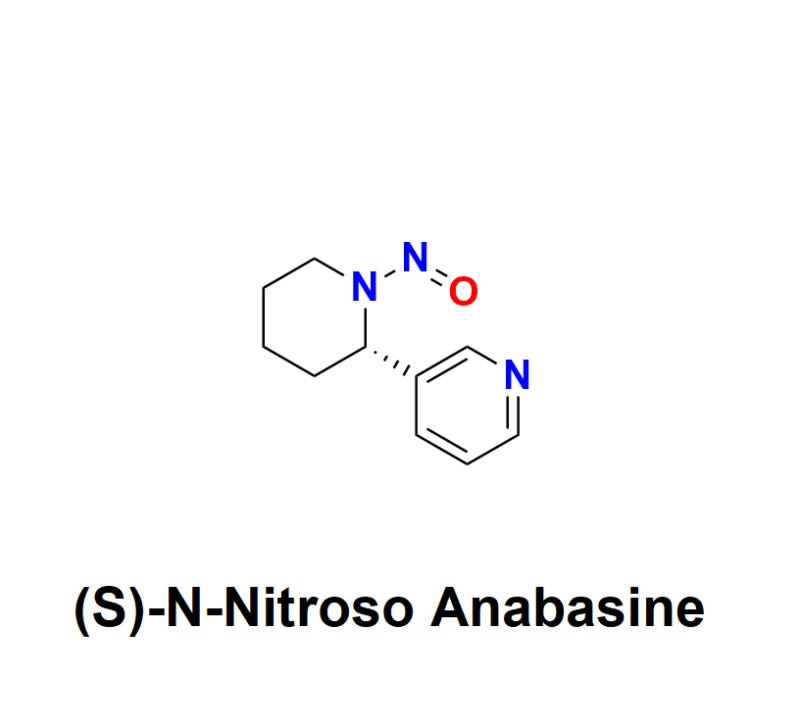
(S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How …
(S)-N-Nitroso Anabasine, a nitrosamine impurity, has raised significant safety concerns within the pharmaceutical industry. Recognized as a probable human carcinogen, this impurity has become a focal point for global regulatory agencies and manufacturers alike, urging a renewed emphasis on detection, prevention, and management.
Learn more about (S)-N-Nitroso Anabasine: https://aquigenbio.com/product/s-n-nitroso-anabasine/
What Is (S)-N-Nitroso Anabasine?
(S)-N-Nitroso Anabasine belongs to the family of nitrosamines, compounds formed through a chemical reaction known as nitrosation. This…
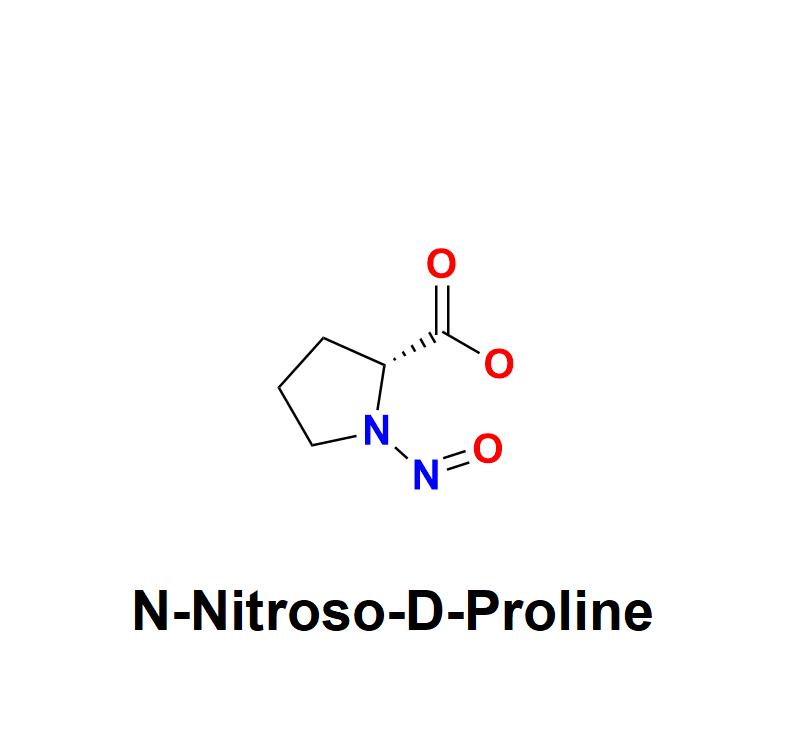
N-Nitroso-D-Proline: A Growing Concern for Pharmaceutical Safety - Explore How A …
N-Nitroso-D-Proline, a chemical impurity belonging to the nitrosamine family, has emerged as a significant concern for pharmaceutical manufacturers worldwide. Its presence in drugs, even at trace levels, poses potential risks to human health due to its genotoxic and carcinogenic properties. As regulatory bodies intensify scrutiny, the pharmaceutical industry must prioritize its detection, mitigation, and prevention to ensure the safety and efficacy of medications.
Learn more about N-Nitroso-D-Proline: https://aquigenbio.com/product/n-nitroso-d-proline/
Understanding N-Nitroso-D-Proline:
N-Nitroso-D-Proline is…
More Releases for Acetylcysteine
Acetylcysteine Market: Transforming Healthcare Through Innovation and Expanding …
Market Overview and Growth Trajectory
The global acetylcysteine market stands as one of the most rapidly expanding pharmaceutical sectors, demonstrating exceptional growth momentum driven by increasing healthcare demands and diverse therapeutic applications. The global acetylcysteine market was valued at approximately USD 1.2 billion in 2023 and is anticipated to reach USD 1.9 billion by 2033, growing at a CAGR of 4.8% from 2024 to 2033.
This remarkable growth trajectory positions acetylcysteine as…
Acetylcysteine Market Global Rising Demand & Huge Scope Till 2031
Acetylcysteine Market business research report assesses the current as well as upcoming performance of the market, also brand-new trends in the market. It provides product overview and highlights of product and application segments of the market including price, revenue, sales, sales growth rate, and market share by product. The market statistics within the report is displayed in a statistical format to offer a better understanding upon the market dynamics.
Request Sample…
N-Acetylcysteine Business Strategy, Top Manufactures And Forecast 2031
"Leading market research firm SkyQuest Technology Group recently released a study titled ' N-Acetylcysteine Market Global Size, Share, Growth, Industry Trends, Opportunity and Forecast 2024-2031,' This study N-Acetylcysteine report offers a thorough analysis of the market, as well as competitor and geographical analysis and a focus on the most recent technological developments. The research study on the N-Acetylcysteine Market extensively demonstrates existing and upcoming opportunities, profitability, revenue growth rates, pricing,…
Acetylcysteine Market Revenue Surges: Acumen Research Projection
The global acetylcysteine market is on a robust growth trajectory, with projections indicating that revenue will reach USD 9.3 billion by 2032. This remarkable growth is underpinned by a compound annual growth rate (CAGR) of 21.1% from 2023 to 2032. Several factors are contributing to this surge, including advancements in medical applications, increasing demand for pharmaceuticals, and expanding accessibility through various distribution channels. This article delves into the key highlights…
Global NAC (Acetylcysteine) Market Research Report 2024
LOS ANGELES, United States: The global NAC (Acetylcysteine) Market is comprehensively analyzed in the report with the main objective of providing accurate market data and useful recommendations to help players to gain strong growth in future. The report is compiled by subject matter experts and experienced market analysts, which makes it highly authentic and reliable. Readers are provided with deep analysis of historical and future market scenarios to get sound understanding of market…
Acetylcysteine Market Size, Share, Development by 2024
Global Info Research offers a latest published report on Acetylcysteine Market Analysis and Forecast 2019-2025 delivering key insights and providing a competitive advantage to clients through a detailed report. This report focuses on the key global Acetylcysteine players, to define, describe and analyze the value, market share, market competition landscape, SWOT analysis and development plans in next few years.
To analyze the Acetylcysteine with respect to individual growth trends, future prospects,…