Press release
N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bio Sciences - Advanced Detection, Control Strategies, and Expert Solutions for Managing Nitrosamine Impurities in Compliance with Regulatory Standards
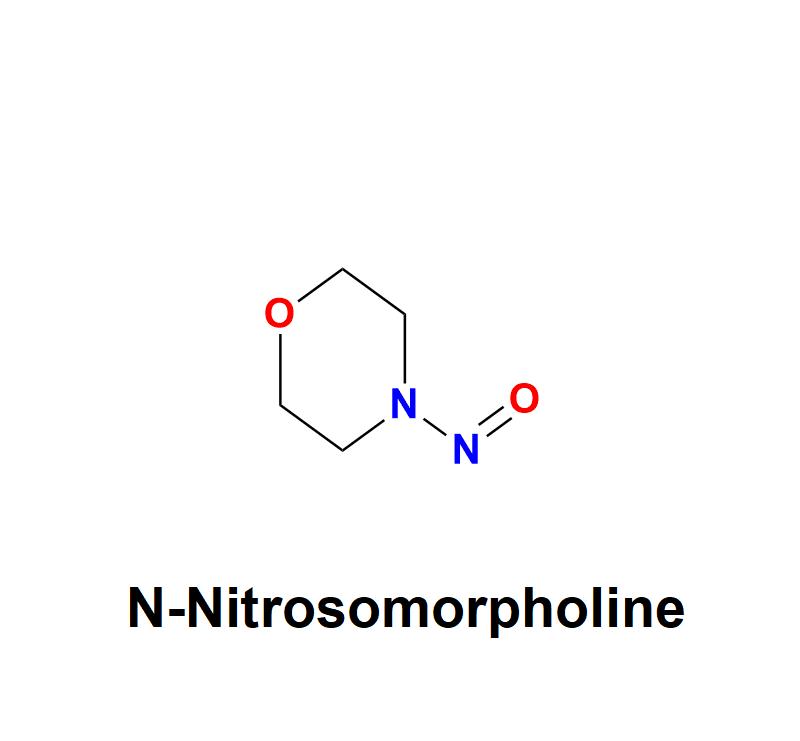
N-Nitrosomorpholine, a compound belonging to the nitrosamine family, has garnered significant attention in the pharmaceutical and healthcare industries due to its potential carcinogenic risks. This chemical impurity, often found as a byproduct in manufacturing processes, poses serious challenges to drug safety and human health, necessitating stringent monitoring and control measures from pharmaceutical companies.Learn more about N-Nitrosomorpholine: https://aquigenbio.com/product/n-nitrosomorpholine/
Understanding N-Nitrosomorpholine:
N-Nitrosomorpholine is a nitrosamine impurity characterized by its chemical structure, which includes a nitroso group (-NO) linked to morpholine, a heterocyclic compound. Nitrosamines, including N-Nitrosomorpholine, are typically formed during specific chemical reactions involving nitrites and amines under acidic or high-temperature conditions. This makes them a frequent contaminant in pharmaceuticals, agriculture, and food production industries.
In pharmaceuticals, N-Nitrosomorpholine can inadvertently form during the synthesis of active pharmaceutical ingredients (APIs), or as a result of the degradation of certain drug formulations. Its presence in medications, even at trace levels, is concerning due to its classification as a potential carcinogen. Regulatory agencies worldwide, including the FDA and EMA, have imposed strict limits on nitrosamine impurities to ensure drug safety.
Health Hazards Associated with N-Nitrosomorpholine:
The health risks associated with N-Nitrosomorpholine cannot be overstated. As a member of the nitrosamine family, it has been linked to mutagenic and carcinogenic effects in animal studies. Prolonged exposure, even at low levels, may increase the risk of liver, kidney, and gastrointestinal cancers. These alarming findings have prompted regulatory bodies to demand rigorous testing, risk assessments, and control strategies to mitigate its presence in pharmaceutical products.
Challenges for Pharmaceutical Companies:
Controlling N-Nitrosomorpholine and other nitrosamine impurities requires significant investment in technology, expertise, and compliance. Pharmaceutical companies face several challenges, including:
1. Detection Difficulties: N-Nitrosomorpholine is often present in minute quantities, making its detection and quantification a susceptible and technical task. Advanced analytical techniques like gas chromatography-mass spectrometry (GC-MS) or liquid chromatography-high resolution mass spectrometry (LC-HRMS) are required to identify its presence accurately.
2. Complex Formation Pathways: Understanding the various pathways through which N-Nitrosomorpholine forms is critical for effective prevention. This includes examining raw materials, excipients, APIs, and production environments to identify potential contamination sources.
3. Regulatory Pressures: Compliance with stringent regulations from agencies like the FDA, EMA, and ICH demands pharmaceutical companies adopt state-of-the-art impurity detection and control systems. This often necessitates revalidating manufacturing processes and adopting advanced risk assessment methodologies.
4. Consumer Trust: Any trace of nitrosamine contamination in drugs can result in product recalls, lawsuits, and damage to brand reputation. Companies must prioritize transparency and quality assurance to maintain consumer trust and comply with regulatory expectations.
Get in Touch with Us - https://aquigenbio.com/contact-us/
Proactive Measures to Mitigate Risks:
To tackle the risks posed by N-Nitrosomorpholine, pharmaceutical companies must embrace a proactive approach to detection and control, which includes:
1. Enhanced Analytical Methods: Implementing sensitive detection methods, such as GC-MS or LC-MS/MS, to ensure compliance with regulatory thresholds.
2. Process Optimization: Revisiting synthesis routes, evaluating raw materials, and employing green chemistry principles to minimize the formation of nitrosamines.
3. Comprehensive Risk Assessments: Conducting thorough risk assessments of the entire manufacturing chain, from raw material sourcing to final product formulation.
4. Regular Training and Audits: Training personnel on nitrosamine risks and conducting routine audits to ensure adherence to best practices.
Regulatory Framework and Global Collaboration:
The fight against N-Nitrosomorpholine contamination requires global collaboration among pharmaceutical manufacturers, regulatory agencies, and independent research organizations. Regulatory agencies have outlined guidelines, such as the ICH M7 guideline, which emphasizes the control of mutagenic impurities. Pharmaceutical companies must align with these frameworks while fostering innovation in detection and prevention strategies.
Conclusion: The Role of Aquigen Bio Sciences
As the pharmaceutical industry navigates the challenges posed by N-Nitrosomorpholine, reliable expertise and resources are indispensable. Aquigen Bio Sciences, a leading contract research organization, is a trusted partner for pharmaceutical companies in addressing impurity-related challenges. With state-of-the-art analytical facilities, cutting-edge technology, and a team of seasoned experts, Aquigen Bio Sciences offers comprehensive solutions for the detection, analysis, and management of N-Nitrosomorpholine and other nitrosamines.
By collaborating with Aquigen Bio Sciences, pharmaceutical companies can enhance their compliance with regulatory requirements, safeguard patient health, and protect their brand integrity. In an era where drug safety is paramount, Aquigen Bio Sciences remains committed to empowering the pharmaceutical industry with innovative tools and solutions to ensure the highest standards of quality and safety. For more information, contact Aquigen Bio Sciences today.
Similar Trending Products:
1) 2-Nitroso-1-Pentylpiperazine: https://aquigenbio.com/product/2-nitroso-1-pentylpiperazine/
2) N-Nitroso Azilsartan: https://aquigenbio.com/product/n-nitroso-azilsartan-2/
3) N-Nitroso Bromocriptine: https://aquigenbio.com/product/n-nitroso-bromocriptine/
Contact Us:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences:
Aquigen Bio Sciences is a renowned contract research organization based in Pune, India, specializing in impurity standards and comprehensive solutions for the pharmaceutical industry. With a focus on advancing pharmaceutical safety and compliance, Aquigen provides expert guidance on identifying, quantifying, and managing impurities in various drug formulations. Their commitment to quality and regulatory excellence positions them as a trusted partner for manufacturers striving to meet stringent safety standards and improve patient outcomes.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bio Sciences - Advanced Detection, Control Strategies, and Expert Solutions for Managing Nitrosamine Impurities in Compliance with Regulatory Standards here
News-ID: 3895716 • Views: …
More Releases from Aquigen Biosciences
Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and …
In the ever-evolving field of targeted cancer therapy, Abemaciclib has emerged as a pivotal agent in the treatment of hormone receptor-positive (HR+), HER2-negative advanced or metastatic breast cancer. As researchers and pharmaceutical developers continue to innovate in oncology, the importance of impurity profiling and the availability of reliable Abemaciclib impurity standards has never been greater.
At the forefront of pharmaceutical impurity standards, Aquigen Bio is proud to support global manufacturers, CROs,…
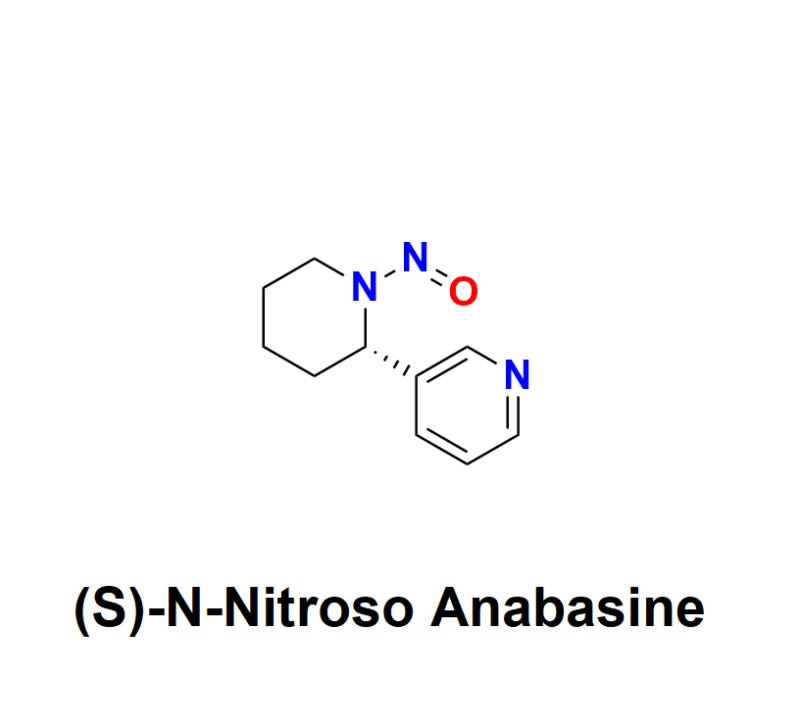
(S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How …
(S)-N-Nitroso Anabasine, a nitrosamine impurity, has raised significant safety concerns within the pharmaceutical industry. Recognized as a probable human carcinogen, this impurity has become a focal point for global regulatory agencies and manufacturers alike, urging a renewed emphasis on detection, prevention, and management.
Learn more about (S)-N-Nitroso Anabasine: https://aquigenbio.com/product/s-n-nitroso-anabasine/
What Is (S)-N-Nitroso Anabasine?
(S)-N-Nitroso Anabasine belongs to the family of nitrosamines, compounds formed through a chemical reaction known as nitrosation. This…
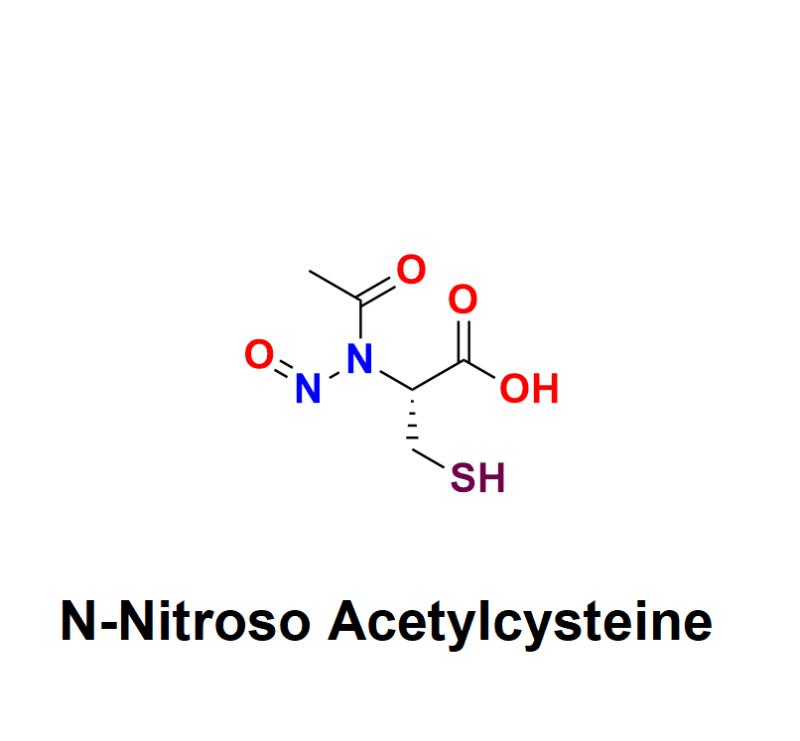
N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Haza …
N-Nitroso Acetylcysteine has emerged as a critical topic of concern in the pharmaceutical industry. As a member of the nitrosamine family, it is a potential impurity that poses significant health risks, including carcinogenicity, even in trace amounts. With increasing regulatory scrutiny on nitrosamine impurities, pharmaceutical manufacturers must address the presence of compounds like N-Nitroso Acetylcysteine to protect public health and ensure compliance with global standards.
Learn more about N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/…
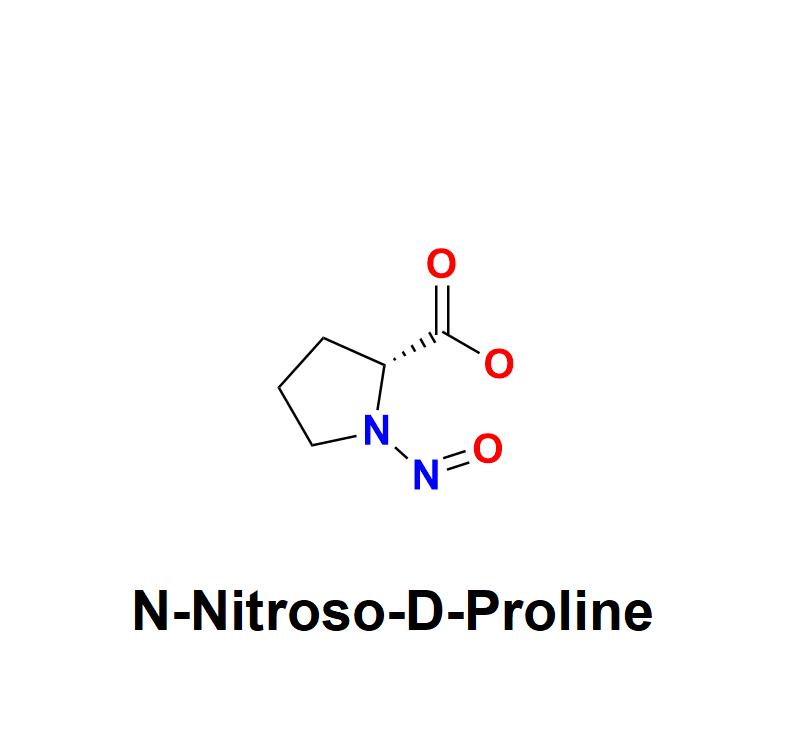
N-Nitroso-D-Proline: A Growing Concern for Pharmaceutical Safety - Explore How A …
N-Nitroso-D-Proline, a chemical impurity belonging to the nitrosamine family, has emerged as a significant concern for pharmaceutical manufacturers worldwide. Its presence in drugs, even at trace levels, poses potential risks to human health due to its genotoxic and carcinogenic properties. As regulatory bodies intensify scrutiny, the pharmaceutical industry must prioritize its detection, mitigation, and prevention to ensure the safety and efficacy of medications.
Learn more about N-Nitroso-D-Proline: https://aquigenbio.com/product/n-nitroso-d-proline/
Understanding N-Nitroso-D-Proline:
N-Nitroso-D-Proline is…
More Releases for Bio
Human Plasma Products Market 2023: Industry Future Trends | Takeda, CSL, Grifols …
The Human Plasma Products market research report is proficient and top to bottom research by specialists on the current state of the industry. This statistical surveying report gives the most up to date industry information and industry future patterns, enabling you to distinguish the items and end clients driving income development and benefit. It centres around the real drivers and restrictions for the key players and present challenge status with…
Gene Therapy Products Market | Advantagene, Avalanche Bio, Bluebird Bio, Cellado …
The global gene therapy products market report is a comprehensive report that provides a detailed analysis of the current status and future trends of the gene therapy products market worldwide. This report provides valuable information to industry stakeholders by offering an in-depth perspective on market dynamics, competitive landscape, growth opportunities, and key challenges faced by industry participants.
From the perspective of market dynamics, this report explores the factors driving the growth…
Bio-based Polymethyl Methacrylate (Bio-PMMA) Market 2023 | Detailed Report
The Bio-based Polymethyl Methacrylate (Bio-PMMA) report compiles the market information depending upon market development and growth factors, optimizing the growth path. In addition, it highlights the strategies and market share of the leading vendors in the particular market. The report follows a robust research methodology model that helps to make informed decisions. It obtains both qualitative and quantitative market information supported by primary research.
The Bio-based Polymethyl Methacrylate (Bio-PMMA) research report…
Bio Pharma Buffer Market – A comprehensive study by Key Players: Bio-Rad, Lonz …
Latest Market intelligence report released by HTF MI with title "COVID-19 Outbreak-Global Bio Pharma Buffer Industry Market Report-Development Trends, Threats, Opportunities and Competitive Landscape in 2020" is designed covering micro level of analysis by manufacturers and key business segments. The COVID-19 Outbreak-Global Bio Pharma Buffer Market survey analysis offers energetic visions to conclude and study market size, market hopes, and competitive surroundings. The research is derived through primary and secondary…
IVIG Market Anticipated a Noteworthy CAGR during 2019-2024 & Companies Included …
A report added to the rich database of Qurate Business Intelligence, titled “World IVIG Market by Product Type, Players and Regions - Forecast to 2024”, provides a 360-degree overview of the Global market. Approximations associated with the market values over the forecast period are based on empirical research and data collected through both primary and secondary sources. The authentic processes followed to exhibit various aspects of the market makes the…
Green & Bio-based Solvents Market Analysis, Demand, & Opportunities till 2023 | …
A latest research report titled as “Green & Bio-based Solvents Market for Paints & Coatings, Printing Inks, Commercial & Domestic Cleaning, Adhesives & Sealants, Pharmaceutical, Cosmetics, and Other Applications - Global Industry Analysis, Size, Share, Growth, Trends and Forecast 2015 - 2023” has been recently added to the vast portfolio of Market Research Reports Search Engine (MRRSE) online research offerings. This report is a professional and in-depth analysis on the…