Press release
Degenerative Disc Disease Treatment Market to Reach $66.2 Billion by 2033 as Gene Therapies and Artificial Discs Reshape Spinal Care - 90% of Adults Over 50 Now Affected
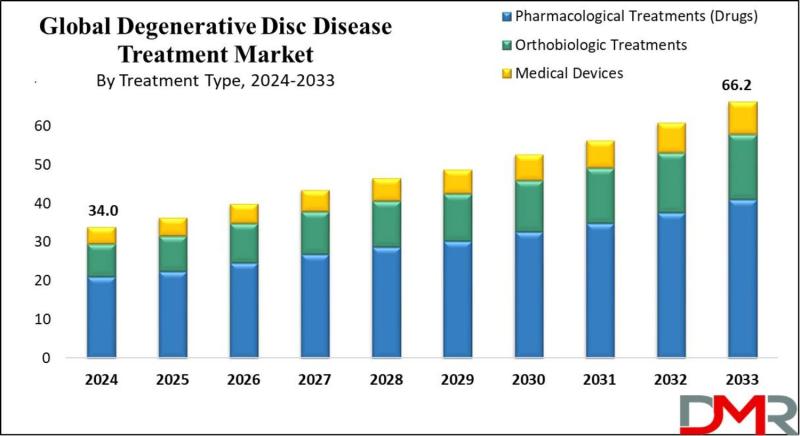
According to Dimension Market Research, the global degenerative disc disease treatment market is accelerating from USD 34.0 billion in 2024 to USD 66.2 billion by 2033, driven by an aging global population and breakthrough innovations in orthobiologics and implantable devices.A comprehensive new market analysis reveals that degenerative disc disease (DDD) is far more prevalent than previously understood. Clinical data published in PubMed, cited in the report, shows that over 90% of adults aged 50 and older exhibit disc degeneration across cervical, thoracic, or lumbar regions, with lumbar prevalence reaching 69-76% depending on sex. This near-universal occurrence among older adults is fundamentally reshaping treatment demand, moving DDD from a niche orthopedic concern to a mainstream chronic condition requiring scalable, cost-effective interventions.
According to Dimension Market Research, pharmacological treatments currently dominate with 61.9% market share, as pain relievers, NSAIDs, and muscle relaxants remain the first line of defense. However, the fastest-growing segments are orthobiologic therapies and next-generation artificial discs, with gene editing and mesenchymal stem cell (MSC) therapies positioned to address the root causes of disc degeneration rather than merely managing symptoms.
📄 Get Your Sample Report Today → https://dimensionmarketresearch.com/request-sample/degenerative-disc-disease-treatment-market/
🔷 Unique News Angle: The Regeneration Revolution - Moving Beyond Pain Management to Disease Modification
The most significant narrative shift in the degenerative disc disease treatment market is the transition from symptom-focused care to disease-modifying interventions. For decades, treatment options were limited to pain relievers, physical therapy, and ultimately spinal fusion - a procedure that eliminates motion to reduce pain but can accelerate degeneration in adjacent spinal segments. That paradigm is now cracking. Gene therapy vectors designed to replace or repair defective genes that cause disc degeneration are advancing through clinical development. Mesenchymal stem cell (MSC) therapies, including the EU-approved treatment from Mesoblast (February 2024), are demonstrating the ability to regenerate damaged disc tissue rather than simply masking pain. Tissue-engineered products combining synthetic scaffolds with bioactive molecules are also gaining traction. According to Dimension Market Research, the orthobiologic segment, while still smaller than pharmaceuticals, is growing at nearly double the rate of traditional drugs. For investors and healthcare systems, this represents a fundamental revaluation: the DDD treatment market is no longer just about managing chronic pain in an aging population - it is about potentially reversing degeneration and avoiding costly spinal surgeries altogether.
🔷 Key Insights - Data That Demands Attention
1. Pharmacological treatments lead at 61.9% share - Pain relievers, NSAIDs, muscle relaxants, and opioids remain the most widely used interventions due to low cost, ease of administration, and accessibility. Oral steroids and narcotics are prescribed for acute and chronic cases respectively.
2. Oral administration dominates at 66.7% - Tablets, capsules, and liquids are preferred by patients for their non-invasive nature, convenience, and ability to be self-administered at home without clinical supervision.
3. Mid-stage disease drives the largest treatment demand - Patients with persistent pain and reduced mobility require more aggressive management, including stronger pharmaceuticals and consideration of surgical options like spinal fusion or artificial disc replacement.
4. North America holds 37.9% global share - The US leads due to a large aging patient pool, presence of major medical device and pharmaceutical companies, advanced healthcare infrastructure, and favorable reimbursement policies.
5. The US market alone is projected to reach $20.3 billion by 2033 - Growing at 7.2% CAGR, the US market benefits from high adoption of minimally invasive surgical techniques and rapid regulatory approvals for novel orthobiologic therapies.
📄 Get the Insights You Need to Drive Real Impact → https://dimensionmarketresearch.com/request-sample/degenerative-disc-disease-treatment-market/
🔷 Market Dynamics: Drivers, Restraints & Strategic Opportunities
Drivers - Why Growth Is Accelerating
The global aging population is the primary engine. As life expectancy increases and birth rates decline in developed economies, the proportion of adults aged 60 and above continues to rise. Since disc degeneration is nearly universal in this demographic, the addressable patient population expands inexorably each year. Technological advancements in medical devices, including robotic-assisted spinal surgery and real-time imaging, have improved success rates and reduced complications, making surgical interventions more attractive. Increased healthcare spending, particularly in North America and Europe, enables broader access to expensive treatments like artificial disc replacement and cell-based therapies.
Restraints - Headwinds to Watch
High treatment costs remain the most significant barrier. Spinal fusion surgery and artificial disc replacement can cost tens of thousands of dollars, placing them out of reach for many patients, particularly in developing regions. Even newer biologic drugs carry premium price tags. Stringent regulatory requirements from the FDA and EMA slow the entry of new therapies, with approval timelines extending five to ten years from initial discovery. Insurance coverage gaps further limit access, particularly for experimental orthobiologic treatments not yet included in standard formularies.
Opportunities - Where to Invest Now
Emerging markets in Asia-Pacific and Latin America represent the largest growth runway. As healthcare infrastructure improves in China, India, and Brazil, demand for minimally invasive surgical options and orthobiologic therapies is poised to surge. Gene therapy and advanced cell-based treatments offer the potential for true disease modification, commanding premium pricing and patient loyalty. Strategic partnerships between medical device companies and academic research institutions, such as Zimmer Biomet's June 2024 collaboration with the University of California, are accelerating the pipeline of breakthrough therapies.
🔷 Segmentation Spotlight - Where the Money Is Flowing
By treatment type, pharmacological treatments dominate at 61.9% share, but orthobiologics are the high-growth segment. Medical devices, including spinal fusion devices and artificial discs, occupy the middle ground, with consistent demand driven by surgical volumes.
By route of administration, oral drugs hold 66.7% share, but injectable biologics are gaining share as MSC therapies and gene therapy vectors require clinical administration. Topical treatments remain a niche segment for localized pain management.
By disease indication, mid-stage DDD commands the largest share. Early-stage patients often manage with over-the-counter medications and lifestyle modifications. Late-stage patients with severe degeneration frequently require surgical intervention. Mid-stage patients, experiencing persistent pain and mobility limitations but not yet candidates for surgery, represent the sweet spot for prescription pharmaceuticals and emerging orthobiologic interventions.
By end user, hospitals dominate due to their ability to provide surgical procedures, multidisciplinary care, and emergency management of complications. Ambulatory surgical centers are gaining share for less complex spinal procedures, while specialty clinics focus on pain management and orthobiologic injections.
🔷 Regional Analysis - North America Leads, Asia-Pacific Emerges
North America holds 37.9% global share in 2024, driven by the US market which alone accounts for $10.8 billion. The region benefits from the highest concentration of major market players including Medtronic, Johnson & Johnson, Stryker, and Zimmer Biomet. Favorable reimbursement policies under Medicare and private insurance plans ensure broad access to both pharmacological and surgical treatments. The FDA's accelerated approval pathways for breakthrough devices and biologics have enabled rapid commercialization of innovations like real-time monitoring spinal implants (Spinal Innovations Inc., December 2023).
Europe follows as the second-largest market, with Germany, France, and the UK leading adoption of minimally invasive techniques. The EU's approval of Mesoblast's MSC therapy (February 2024) represents a regulatory milestone that could accelerate similar approvals globally.
Asia-Pacific is the fastest-growing region, driven by large aging populations in China and Japan, rising disposable incomes, and improving healthcare infrastructure. India's growing medical tourism sector for spinal surgeries is also contributing to regional growth. Local manufacturers are entering the market with lower-cost spinal devices, intensifying price competition.
🔷 Competitive Landscape - Giants, Specialists, and Biotech Disruptors
The competitive landscape features established medical device giants, pharmaceutical leaders, and emerging biotech firms specializing in orthobiologics. Medtronic remains the market leader in spinal devices, launching its next-generation spinal fusion device in August 2024 with advanced materials designed to enhance fusion rates and reduce postoperative complications. Johnson & Johnson, through its DePuy Synthes subsidiary, introduced a new generation of artificial discs in April 2024 featuring enhanced durability and biomechanics that more closely mimic natural spinal movement.
Stryker completed the acquisition of BioTech Innovations in May 2024, expanding its orthobiologic portfolio with cell-based therapies and tissue-engineered products. Zimmer Biomet partnered with the University of California in June 2024 to develop gene therapies for early-stage DDD. In the pharmaceutical space, Pfizer received FDA approval in July 2024 for a novel oral anti-inflammatory drug specifically targeting mid-stage DDD inflammation. Eli Lilly launched a new pain management drug in January 2024 designed for long-term chronic pain management. In orthobiologics, Mesoblast received EU approval for its MSC therapy in February 2024, the first cell-based treatment approved for mid-stage DDD in the region. Vericel Corporation reported positive Phase III results in March 2024 for its cell-based therapy targeting late-stage patients, demonstrating significant improvements in pain relief and disc function.
📄 Get the Full Premium Report Now → https://dimensionmarketresearch.com/checkout/degenerative-disc-disease-treatment-market/
🔷 Strategic Opportunities for Lead Generation
Throughout this release, we have withheld granular data on regional reimbursement landscapes, comparative efficacy rates of different MSC therapy protocols, FDA approval timelines for gene therapy vectors currently in development, and detailed market shares of individual players in the artificial disc segment. These insights, along with 10-year forecasts by treatment type, route of administration, disease indication, and end user, are available exclusively in the full report. Investors, healthcare procurement decision-makers, and pharmaceutical R&D leaders seeking to validate pipeline investments, identify underserved geographic markets, or benchmark competitive pricing strategies should access the complete dataset.
🔷 Why This Market Intelligence Matters Now
The degenerative disc disease treatment market is at a historic inflection point. For the first time, disease-modifying interventions - gene therapy, MSC regeneration, tissue engineering - are moving from academic research into clinical practice. The EU's approval of Mesoblast's MSC therapy in February 2024 marks the beginning of a new treatment era. Simultaneously, the near-universal prevalence of disc degeneration among adults over 50 means the addressable market is vast and growing with each aging cohort. According to Dimension Market Research, the next 36 months will see accelerated M&A activity as pharmaceutical companies acquire biotech firms with promising orthobiologic pipelines. Medical device companies will increasingly bundle spinal implants with biologic adjuvants to improve fusion rates. For hospitals, insurers, and investors, understanding which technologies deliver genuine clinical differentiation versus incremental improvement is critical for strategic positioning. The shift from pain management to regeneration is not theoretical - it is underway. Early adopters of orthobiologic technologies and gene-based interventions will define the standard of care for the next decade.
📄 Explore the Report with TOC → https://dimensionmarketresearch.com/report/degenerative-disc-disease-treatment-market/
For Sales or Inquiries, Contact
Robert John
957 Route 33, Suite 12 #308 Hamilton Square, NJ-08690 USA
Email: enquiry@dimensionmarketresearch.com
United States: (+1 732 369 9777)
Tel No: +91 88267 74855
Dimension Market Research (DMR) is a market research and consulting firm based in India & US, with its headquarters located in the USA. The company believes in providing the best and most valuable data to its customers using the best resources and analysts to work on, to create unmatchable insights into the industries and markets while offering in-depth results of over 30 industries, and all major regions across the world. We also believe that our clients don't always want what they see, so we provide customized reports as well, as per their specific requirements, to create the best possible outcomes for them and enhance their business through our data and insights in every possible way.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Degenerative Disc Disease Treatment Market to Reach $66.2 Billion by 2033 as Gene Therapies and Artificial Discs Reshape Spinal Care - 90% of Adults Over 50 Now Affected here
News-ID: 4513292 • Views: …
More Releases from Dimension Market Research
Breast Pumps Market to Reach $8.0 Billion by 2033, Growing at 10% CAGR as Wearab …
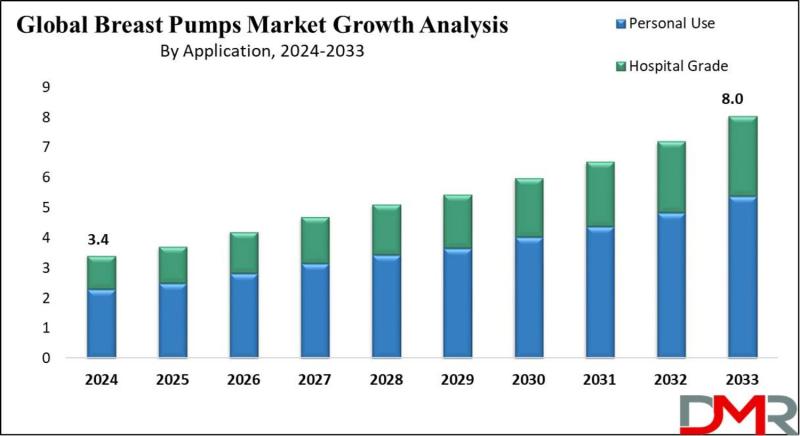
According to Dimension Market Research, the global breast pumps market is accelerating from USD 3.4 billion in 2024 to USD 8.0 billion by 2033, driven by rising female workforce participation and surging demand for discreet, tech-enabled pumping solutions.
A newly published market analysis reveals that breast pumps have evolved from basic medical devices into essential lifestyle tools for modern mothers. With nearly 67% of sales going to personal-use devices, the market…
Medical Bed Market to Reach $7.0 Billion by 2034 as AI-Integrated Smart Beds Res …
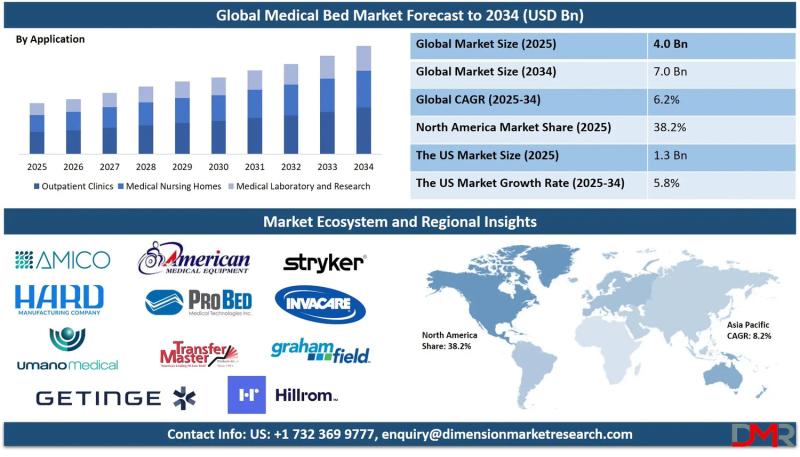
According to Dimension Market Research, the market is set to expand at a 6.2% CAGR from 2025 to 2034, with electronic beds capturing nearly half of all demand and outpatient clinics driving 74.9% of application-level revenue.
A new comprehensive analysis projects the global medical bed market will climb from USD 4.0 billion in 2025 to USD 7.0 billion by 2034, driven by an unprecedented convergence of demographic aging, chronic disease prevalence,…
Europe Heparin Market to Reach $7.1 Billion by 2033 as Anticoagulant Demand Rise …
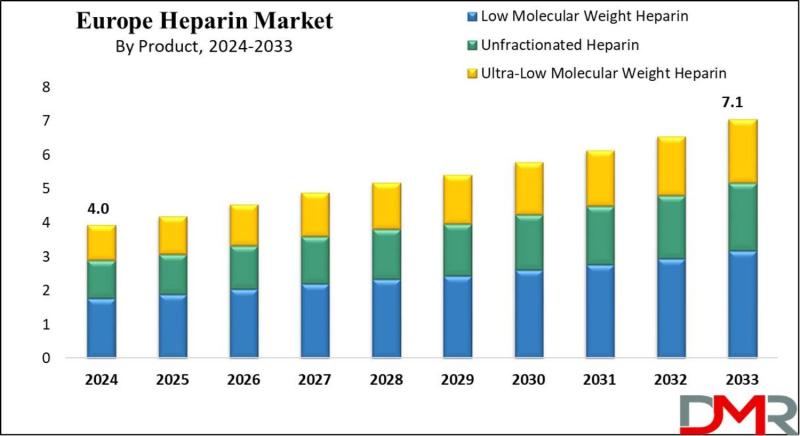
The Europe Heparin Market is on a steady growth trajectory, with market valuation projected to rise from an estimated USD 4.0 billion in 2024 to USD 7.1 billion by 2033, registering a compound annual growth rate (CAGR) of 6.6%. According to Dimension Market Research, this expansion is being driven by three converging forces: the rising prevalence of cardiovascular diseases including deep vein thrombosis, pulmonary embolism, and atrial fibrillation; the increasing…
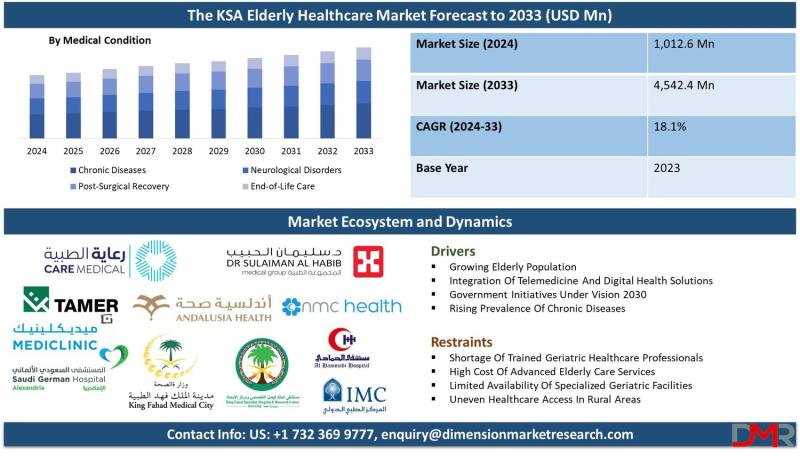
Kingdom of Saudi Arabia Elderly Healthcare Market to Reach $44,542.4 million by …
The Kingdom of Saudi Arabia (KSA) Elderly Healthcare Market is poised for explosive growth, with market valuation projected to surge from an estimated USD 1,012.6 million in 2024 to USD 4,542.4 million by 2033, registering a remarkable compound annual growth rate (CAGR) of 18.1%. According to Dimension Market Research, this extraordinary expansion is being driven by three converging forces: the rapidly aging Saudi population (over 11% expected to be above…
More Releases for MSC
Desh Bhagat University Announces MSc Nursing Entrance Exam 2026
Mandi Gobindgarh, Punjab, India - Desh Bhagat University is pleased to announce the MSc Nursing Entrance Exam for admission to its postgraduate nursing programme for the year 2026. This exam is an important step for students who want to pursue advanced studies in nursing.
The MSc Nursing Entrance Exam is designed to help students who have completed their B.Sc Nursing (Basic or Post Basic) take the next step in their…
Indian Students Choosing University of Leicester, UK for MSc Law and MSc Crimino …
Kochi, India - More and more Indian students are choosing to pursue higher studies at the University of Leicester, UK. With excellent academic programs like MSc Law and MSc Criminology, the university offers world-class education and career opportunities. As the No. 1 study abroad consultants in Kochi, Guidance Plus is proud to support students in making their study abroad dreams a reality.
Study in University of Leicester
The University of Leicester is…
Cruise Market is Booming Worldwide | Carnival , RCI , NCLH , MSC
HTF Market Intelligence released a new research report of 121 pages on title 'Global Cruise Market Report 2018' with detailed analysis, forecast and strategies. The study covers key regions that includes North America Country (United States, Canada) , South America , Asia Country (China, Japan, India, Korea) , Europe Country (Germany, UK, France, Italy) , Other Country (Middle East, Africa, GCC) and important players such as Carnival , RCI…
MSC Recognizes EAC Network at Breakfast of Champions
EAC Network received a Certificate of Appreciation from MSC at their second annual Breakfast of Champions event, which recognizes the Long Island company’s local nonprofit partners.
MSC is a leading North American distributor of maintenance, repair, and operations products and services based in Melville, NY. The company has built strong partnerships with local and national nonprofit organizations throughout its 75-year history.
MSC has supported EAC Network significantly through funding since…
FDM Honours First FDM MSc Programme Graduate
The UK’s leading IT graduate employer, FDM Group, has honoured its first graduate from the FDM MSc Programme at the Brighton Dome.
The graduation ceremony, held on 28th July, saw FDM Trainer Kevin Wright accept his Master’s in Applied Computer Science from University of Brighton. FDM fully-funded the course, which he studied via distance learning alongside his day job with the company.
Developed in partnership with University of Brighton,…
Info Session - MSc Program Engineering Management
Your key to the top engineeRing league!
On May 8, 2008 the info session for the MSc Program Engineering Management takes place at the Vienna University of Technology.
This part-time MSc Program is tailor-made for Europe`s small and medium-sized enterprises facing a growing and increasingly competitive market. It is design to prepare graduates from technical and economic universities for leadership roles in technical, corporate and national affairs.
The Academic Director Prof. DDr.…