Press release
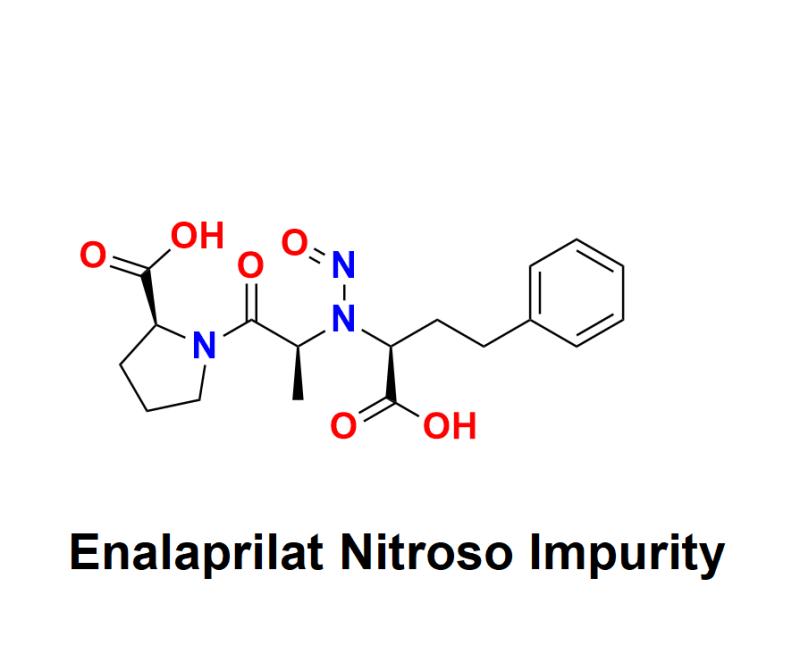
Enalaprilat Nitroso Impurity: Comprehensive Testing, Regulatory Compliance, and Quality Control Solutions By Aquigen Bio Sciences for Enhanced Drug Safety and Efficacy in Pharmaceutical Manufacturing
Enalaprilat Nitroso Impurity has emerged as a significant topic within pharmaceutical research and development, drawing attention to the crucial need for impurity standards and quality control. As an impurity found in Enalapril, a widely prescribed medication for managing high blood pressure and certain heart conditions, Enalaprilat Nitroso Impurity underscores the necessity of rigorous testing and safety protocols. Aquigen Bio Sciences, a leading contract research organization based in Pune, India, provides comprehensive resources and solutions to detect, quantify, and manage Enalaprilat Nitroso Impurity, ensuring the highest standards of drug safety.Learn more about Enalaprilat Nitroso Impurity: https://aquigenbio.com/product/enalaprilat-nitroso-impurity/
Enalaprilat Nitroso Impurity: Background and Importance
Enalapril is an ACE (angiotensin-converting enzyme) inhibitor prescribed for conditions such as hypertension and heart failure. However, during its manufacturing process, impurities such as Enalaprilat Nitroso Impurity can be introduced, posing potential health risks if left unchecked. Nitrosamines, including Enalaprilat Nitroso Impurity, are of particular concern because of their links to adverse effects when accumulated in the body over extended periods. The identification and management of these impurities are essential for regulatory compliance, drug efficacy, and patient safety, prompting both pharmaceutical manufacturers and regulatory authorities to prioritize impurity management.
In recent years, global regulatory bodies such as the U.S. FDA, European Medicines Agency (EMA), and Health Canada have heightened their scrutiny of nitrosamine impurities in pharmaceuticals. These authorities have introduced stringent guidelines for manufacturers to control and limit nitrosamine impurities, including those like Enalaprilat Nitroso Impurity, to ensure that medications remain safe and effective for patients.
Role of Enalaprilat Nitroso Impurity in Pharmaceutical Development:
The presence of impurities in pharmaceutical drugs is not uncommon. However, nitrosamines are classified as probable human carcinogens, making their identification, quantification, and control especially critical. Enalaprilat Nitroso Impurity, if present above acceptable limits, may pose risks that could compromise the quality, safety, and efficacy of Enalapril. Therefore, accurate and reliable testing is indispensable.
Pharmaceutical companies must integrate stringent quality control measures, with specialized laboratories performing sophisticated analyses to detect and quantify Enalaprilat Nitroso Impurity. These tests include advanced chromatographic and spectroscopic techniques, ensuring that even trace levels of impurities are identified and managed effectively. As drug safety requirements evolve, such as the adoption of ICH M7 guidelines, the methods for assessing genotoxic impurities like Enalaprilat Nitroso Impurity become even more refined, incorporating robust methodologies to safeguard patient health.
Functions, Uses, and Restrictions of Enalapril and Enalaprilat Nitroso Impurity:
Enalapril is known for its effectiveness in managing high blood pressure and associated cardiovascular risks, including preventing heart attacks and strokes. It works by relaxing blood vessels, enabling improved blood flow and reduced strain on the heart. However, the presence of impurities, including Enalaprilat Nitroso Impurity, highlights the necessity for rigorous quality checks to maintain the drug's efficacy without compromising patient safety.
While Enalaprilat Nitroso Impurity itself is not a usable component of Enalapril, it is an unintended byproduct that requires diligent monitoring. Due to its potential carcinogenic effects, regulatory bodies mandate strict limits on the permissible levels of such impurities, ensuring that the drug remains within safe usage parameters. As a result, manufacturers must invest in advanced technologies and adhere to guidelines that prevent unacceptable levels of Enalaprilat Nitroso Impurity in final pharmaceutical products.
Get in Touch with Us - https://aquigenbio.com/contact-us/
Aquigen Bio Sciences: The Premier Resource for Enalaprilat Nitroso Impurity Solutions
Aquigen Bio Sciences stands at the forefront of impurity testing and management, providing an exceptional range of solutions for detecting and controlling Enalaprilat Nitroso Impurity. As a contract research organization (CRO) committed to quality and regulatory compliance, Aquigen Bio Sciences offers specialized expertise, helping pharmaceutical companies meet and exceed industry standards. Their state-of-the-art laboratory facilities and team of experienced chemists enable comprehensive impurity analysis, allowing manufacturers to confidently address quality concerns related to Enalapril and other critical medications.
Aquigen Bio Sciences' services extend beyond routine impurity analysis; they provide tailored solutions, consulting support, and advanced research capabilities, helping pharmaceutical companies navigate the complexities of impurity control. With a deep understanding of regulatory requirements and advanced testing methodologies, Aquigen Bio Sciences ensures that its clients meet both global and local safety standards, safeguarding the efficacy and safety of their pharmaceutical products.
Their comprehensive range of services includes:
1. Impurity Profiling: Precise identification and quantification of impurities like Enalaprilat Nitroso Impurity, ensuring comprehensive safety and quality evaluation.
2. Custom Testing Solutions: Development of tailored analytical methods that align with each client's unique requirements and regulatory standards.
3. Regulatory Consultation: Expert guidance on global regulatory guidelines for impurity levels.
4. Analytical Method Development and Validation: Creation and validation of testing protocols that enhance the detection and quantification of impurities, ensuring consistent reliability and accuracy.
Similar Trending Products:
1) Nitroso sitagliptin STG-19: https://aquigenbio.com/product/nitroso-sitagliptin-stg-19/
2) N-Nitrosodiisobutylamine: https://aquigenbio.com/product/n-nitrosodiisobutylamine/
3) 4-Chloro-N-nitrosopiperidine: https://aquigenbio.com/product/4-chloro-n-nitrosopiperidine/
Conclusion:
Enalaprilat Nitroso Impurity highlights the growing focus on impurity control in the pharmaceutical industry, emphasizing the need for dedicated solutions to ensure drug safety and efficacy. Aquigen Bio Sciences has proven itself as a premier partner in impurity testing, helping pharmaceutical manufacturers meet the highest standards of quality and regulatory compliance. By offering precise analysis, expert consultation, and regulatory alignment, Aquigen Bio Sciences empowers its clients to deliver safe and effective medications.
For companies seeking a trusted resource for managing Enalaprilat Nitroso Impurity and other critical impurities, Aquigen Bio Sciences stands out as an industry leader, supporting quality-driven pharmaceutical development. Their commitment to innovation, regulatory insight, and tailored solutions makes them the ideal partner for pharmaceutical companies aiming to bring safe, high-quality products to market.
Contact Us:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences:
Aquigen Bio Sciences is a research organization based in Pune, India, specializing in a wide range of scientific research and development services. With a focus on innovation and excellence, Aquigen Bio Sciences delivers high-quality solutions to meet the complex needs of the pharmaceutical, environmental, and industrial sectors. The company is dedicated to advancing scientific knowledge and improving safety through cutting-edge research and technology.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Enalaprilat Nitroso Impurity: Comprehensive Testing, Regulatory Compliance, and Quality Control Solutions By Aquigen Bio Sciences for Enhanced Drug Safety and Efficacy in Pharmaceutical Manufacturing here
News-ID: 3718588 • Views: …
More Releases from Aquigen Biosciences
Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and …
In the ever-evolving field of targeted cancer therapy, Abemaciclib has emerged as a pivotal agent in the treatment of hormone receptor-positive (HR+), HER2-negative advanced or metastatic breast cancer. As researchers and pharmaceutical developers continue to innovate in oncology, the importance of impurity profiling and the availability of reliable Abemaciclib impurity standards has never been greater.
At the forefront of pharmaceutical impurity standards, Aquigen Bio is proud to support global manufacturers, CROs,…
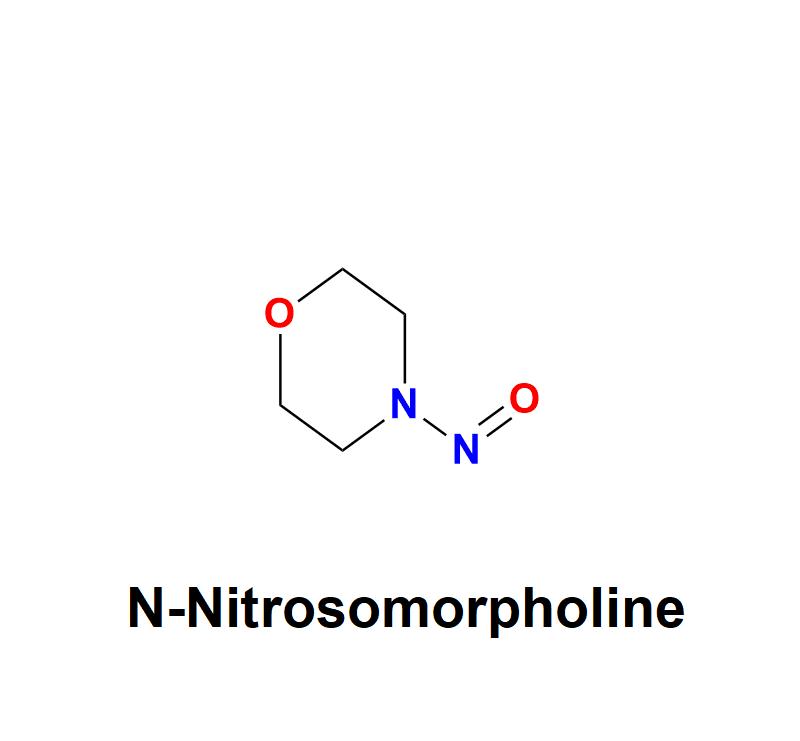
N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bi …
N-Nitrosomorpholine, a compound belonging to the nitrosamine family, has garnered significant attention in the pharmaceutical and healthcare industries due to its potential carcinogenic risks. This chemical impurity, often found as a byproduct in manufacturing processes, poses serious challenges to drug safety and human health, necessitating stringent monitoring and control measures from pharmaceutical companies.
Learn more about N-Nitrosomorpholine: https://aquigenbio.com/product/n-nitrosomorpholine/
Understanding N-Nitrosomorpholine:
N-Nitrosomorpholine is a nitrosamine impurity characterized by its chemical structure, which includes…
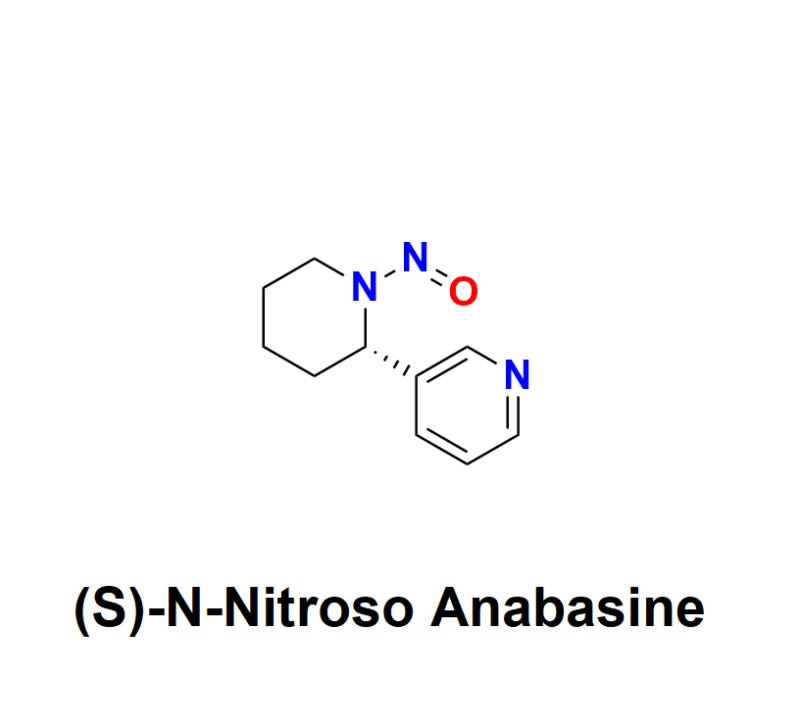
(S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How …
(S)-N-Nitroso Anabasine, a nitrosamine impurity, has raised significant safety concerns within the pharmaceutical industry. Recognized as a probable human carcinogen, this impurity has become a focal point for global regulatory agencies and manufacturers alike, urging a renewed emphasis on detection, prevention, and management.
Learn more about (S)-N-Nitroso Anabasine: https://aquigenbio.com/product/s-n-nitroso-anabasine/
What Is (S)-N-Nitroso Anabasine?
(S)-N-Nitroso Anabasine belongs to the family of nitrosamines, compounds formed through a chemical reaction known as nitrosation. This…
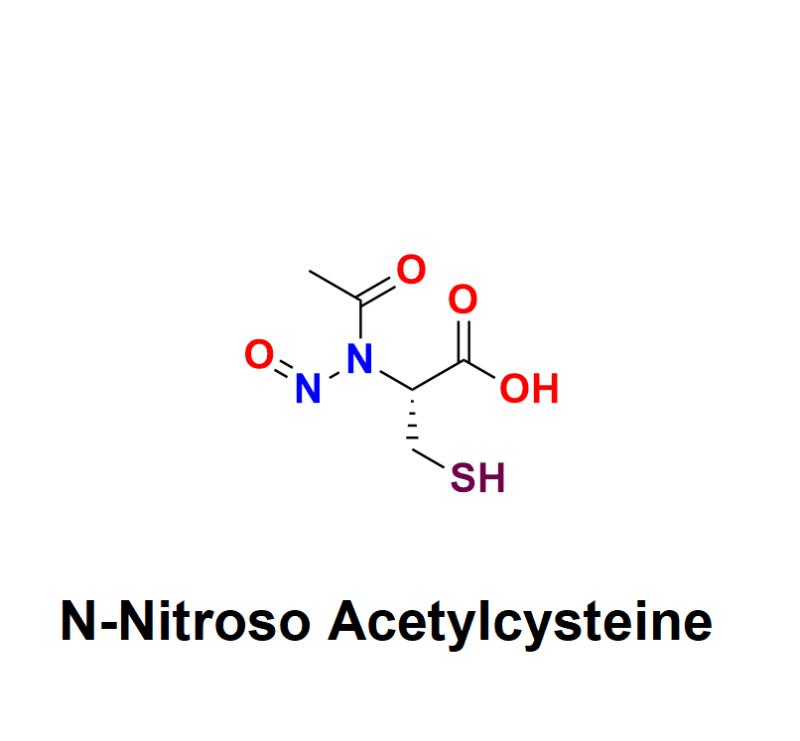
N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Haza …
N-Nitroso Acetylcysteine has emerged as a critical topic of concern in the pharmaceutical industry. As a member of the nitrosamine family, it is a potential impurity that poses significant health risks, including carcinogenicity, even in trace amounts. With increasing regulatory scrutiny on nitrosamine impurities, pharmaceutical manufacturers must address the presence of compounds like N-Nitroso Acetylcysteine to protect public health and ensure compliance with global standards.
Learn more about N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/…
More Releases for Enalapril
Enalapril Market Growth Reach USD 3.9 Billion CAGR at a 4.15% 2024 To 2032 | Tev …
In 2024, the Enalapril market growth is growing steadily with exponential rate and a rising adoption of strategies by top industry players, over the projected horizon 2024 to 2032. The high adoption of advanced technology and the presence of large players in this region are likely to create ample growth opportunities for the Enalapril market. Technological innovation and advancement will also optimize the performance of the product, making it more…
Global Enalapril Maleate Market Research Report 2023-2029
Global Leading Market Research Publisher QYResearch announces the release of its lastest report "Global Enalapril Maleate Market Report, History and Forecast 2018-2029, Breakdown Data by Manufacturers, Key Regions, Types and Application". Based on historical analysis (2018-2022) and forecast calculations (2023-2029), this report provides a comprehensive analysis of the global Enalapril Maleate market, including market size, share, demand, industry development status, and forecasts for the next few years. Provides advanced statistics…
Enalapril Maleate Market Disclosing Latest Advancement 2023 to 2028 Russo-Ukrain …
About Global Market Monitor
Global Market Monitor is a professional modern consulting company, engaged in three major business categories such as market research services, business advisory, technology consulting.Our team is composed of highly qualified professionals with decades of experience in a variety of sectors, from finance and accounting to operations, marketing, IT and human resources. We make sure that each of our consultants has an in-depth understanding of the industry…
Enalapril Maleate API market: Gross Margin, Cost, and Revenue Forecasts for 2023 …
"The Enalapril Maleate API global market is thoroughly researched in this report, noting important aspects like market competition, global and regional growth, market segmentation and market structure. The report author analysts have estimated the size of the global market in terms of value and volume using the latest research tools and techniques. The report also includes estimates for market share, revenue, production, consumption, gross profit margin, CAGR, and other key…
Enalapril Maleate Market Size is Likely to Experience a Huge Growth of by 2030
Global Enalapril Maleate Market 2022-2028 - Global Industry Size, Supply Analysis, Price Analysis, Consumption and Production, Supplier and Cost Structure Analysis
Enalapril Maleate Market Overview
The recent market intelligence study by the research analysts at Market Business Insights (MBI) takes a deep dive into inner operations of the global Enalapril Maleate Market. It thoroughly examines the market landscape to give the readers an accurate insight into the current, as well as the…
Enalapril Market Insights on Trends, Drivers and Opportunities until the End of …
This report is an extensive analysis of current and potential trends in the Enalapril market. The report provides an in-depth quantitative analysis of the current and future market estimations which helps to recognize the prevailing market opportunities. Key market players within the market are profiled in this report and their strategies are analysed completely which help to understand the competitive outlook 2020-2025.
Global Enalapril Market Overview:
The report also reveals in-depth details…