Press release
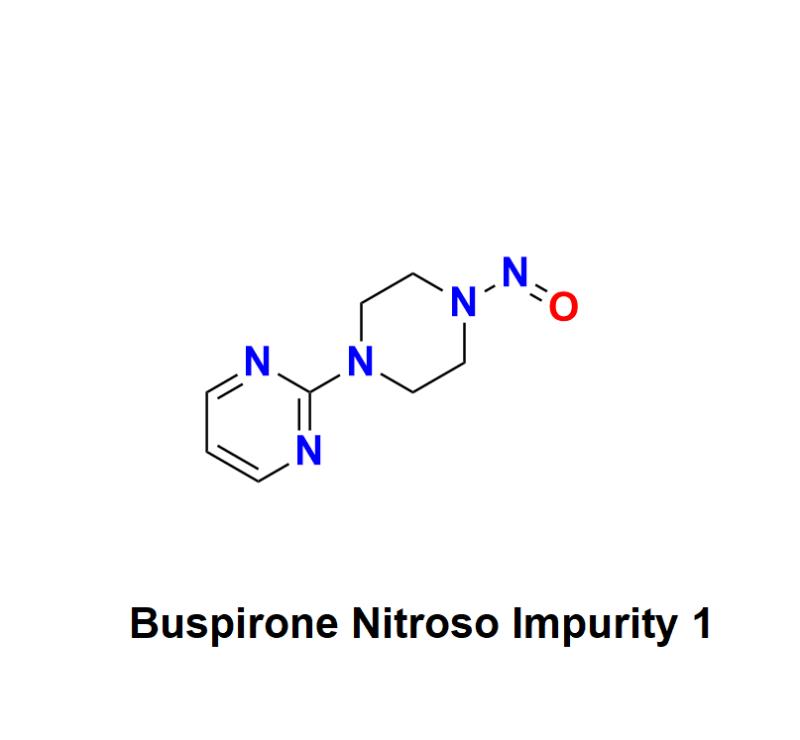
Buspirone Nitroso Impurity 1: A Growing Concern for Patient Safety, Health Risks, and Regulatory Oversight in the Pharmaceutical Industry's Efforts to Ensure the Safety, Efficacy, and Quality of Mental Health Medications like Buspirone
Buspirone Nitroso Impurity 1 has recently emerged as a critical concern within the pharmaceutical landscape, particularly about mental health medications. As a widely prescribed anxiolytic, Buspirone has offered relief to millions of patients dealing with anxiety disorders. However, the detection of this impurity raises important questions about its safety, efficacy, and the potential risks associated with its use.Buspirone is primarily used to treat anxiety disorders and is often favored for its non-sedative properties. While the drug has proven effective for many patients, the presence of Buspirone Nitroso Impurity 1 poses serious health risks, including the potential for carcinogenic effects. Nitrosamines, such as this impurity, have been linked to various types of cancer, making it essential for healthcare providers, regulatory agencies, and pharmaceutical manufacturers to closely monitor and address these risks.
Learn more about Buspirone Nitroso Impurity 1: https://aquigenbio.com/product/buspirone-nitroso-impurity-1-2/
"Effectively managing NBCEA and other nitrosamine impurities is essential for pharmaceutical companies to uphold the safety and quality of their drug products," states the CEO of Aquigen Bio Sciences. "At Aquigen, we have developed specialized expertise in identifying, characterizing, and controlling these impurities, empowering our clients to navigate the complex regulatory environment and guarantee that their medications comply with the highest standards of safety and purity."
Key Implications For Patient Safety:
1. Health Risks: The identification of Buspirone Nitroso Impurity 1 presents potential health risks that cannot be ignored. Patients taking Buspirone may face increased exposure to harmful substances, raising concerns about long-term safety and the overall impact on their health.
2. Regulatory Oversight: Regulatory bodies such as the U.S. Food and Drug Administration (FDA) are now tasked with scrutinizing the presence of Buspirone Nitroso Impurity 1 in Buspirone. The situation necessitates immediate action to implement stricter testing protocols and guidelines to ensure that all pharmaceuticals are safe for public consumption.
3. Treatment Adherence: Concerns over medication safety can lead to decreased patient adherence to prescribed treatments. If patients are aware of potential impurities in their medications, they may become reluctant to continue their treatment, which could adversely affect their mental health outcomes. It is vital for healthcare providers to address these concerns and reassure patients about the steps being taken to ensure their safety.
4. Research and Development Needs: The emergence of Buspirone Nitroso Impurity 1 underscores the need for ongoing research into the formation and mitigation of nitrosamine impurities in pharmaceuticals. Pharmaceutical manufacturers must invest in refining their production processes to eliminate such impurities and enhance the safety of their products.
Get in Touch with Us - https://aquigenbio.com/contact-us/
What Should Stakeholders Do?
The implications of Buspirone Nitroso Impurity 1 necessitate a unified response from all stakeholders within the pharmaceutical industry. It is crucial to enhance testing methodologies and elevate regulatory standards while prioritizing patient safety at every stage of drug development and distribution.
Healthcare providers must engage in transparent discussions with patients regarding their medications and associated risks. Patients should feel empowered to voice their concerns, and healthcare professionals must be prepared to provide clear and accurate information about the safety of their treatments. This openness is vital for sustaining trust in the healthcare system.
Pharmaceutical manufacturers are also responsible for implementing rigorous quality control measures to identify and eliminate impurities in their products. By prioritizing safety and investing in research and development, these companies can ensure that their medications adhere to the highest standards of efficacy and safety. This collaborative effort will ultimately protect patient health and enhance the credibility of the pharmaceutical sector.
Conclusion:
As the pharmaceutical industry navigates the challenges posed by Buspirone Nitroso Impurity 1, maintaining a focus on the safety and efficacy of mental health medications is essential. Proactively addressing the concerns surrounding this impurity will not only protect patient health but also enhance public trust in the pharmaceutical sector. By fostering collaboration among stakeholders - regulatory bodies, manufacturers, and healthcare providers - the industry can ensure that patients receive safe and effective treatments. This commitment to quality and safety will ultimately reinforce the credibility of the entire pharmaceutical landscape and contribute to better health outcomes for individuals relying on these vital medications.
Similar Trending Products:
1) N-Nitrosomorpholine - https://aquigenbio.com/product/n-nitrosomorpholine/
2) N-Nitroso-thiazolidine - https://aquigenbio.com/product/n-nitroso-thiazolidine/
3) 1-Methyl-4-(nitroso)piperazine - https://aquigenbio.com/product/1-methyl-4-nitrosopiperazine/
Contact Us:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences:
Aquigen Bio Sciences is a leading contract research organization specializing in impurity standards, including Buspirone Nitroso Impurity 1. The company is dedicated to supporting pharmaceutical firms in ensuring the safety and integrity of their products. With a commitment to excellence in analytical testing and risk management, Aquigen Bio Sciences plays a pivotal role in helping manufacturers navigate the complexities of impurities and regulatory compliance. By providing reliable data and innovative solutions, the organization contributes to the development of safer medications, ultimately benefiting public health and trust in the pharmaceutical industry.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Buspirone Nitroso Impurity 1: A Growing Concern for Patient Safety, Health Risks, and Regulatory Oversight in the Pharmaceutical Industry's Efforts to Ensure the Safety, Efficacy, and Quality of Mental Health Medications like Buspirone here
News-ID: 3703590 • Views: …
More Releases from Aquigen Biosciences
Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and …
In the ever-evolving field of targeted cancer therapy, Abemaciclib has emerged as a pivotal agent in the treatment of hormone receptor-positive (HR+), HER2-negative advanced or metastatic breast cancer. As researchers and pharmaceutical developers continue to innovate in oncology, the importance of impurity profiling and the availability of reliable Abemaciclib impurity standards has never been greater.
At the forefront of pharmaceutical impurity standards, Aquigen Bio is proud to support global manufacturers, CROs,…
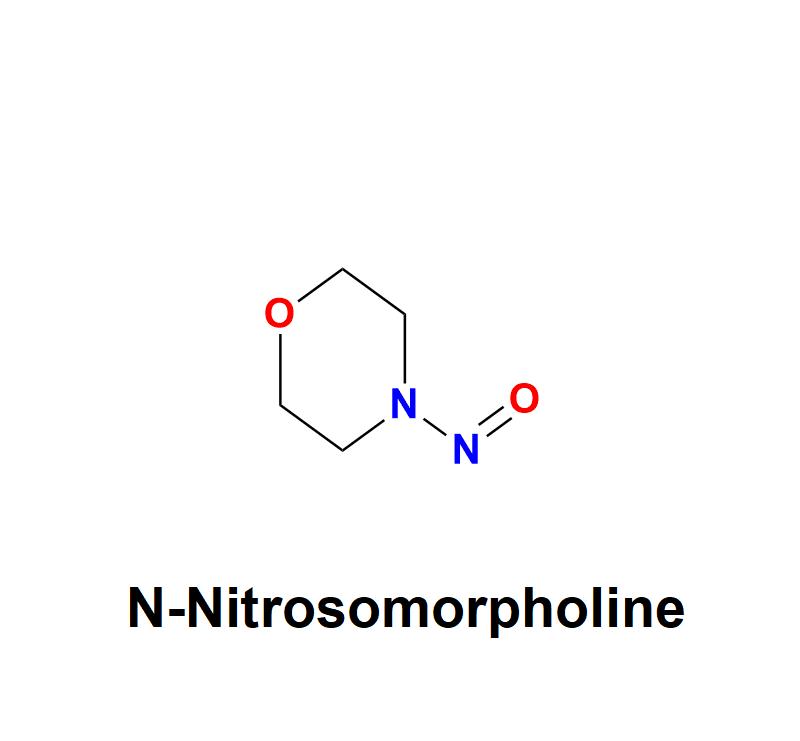
N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bi …
N-Nitrosomorpholine, a compound belonging to the nitrosamine family, has garnered significant attention in the pharmaceutical and healthcare industries due to its potential carcinogenic risks. This chemical impurity, often found as a byproduct in manufacturing processes, poses serious challenges to drug safety and human health, necessitating stringent monitoring and control measures from pharmaceutical companies.
Learn more about N-Nitrosomorpholine: https://aquigenbio.com/product/n-nitrosomorpholine/
Understanding N-Nitrosomorpholine:
N-Nitrosomorpholine is a nitrosamine impurity characterized by its chemical structure, which includes…
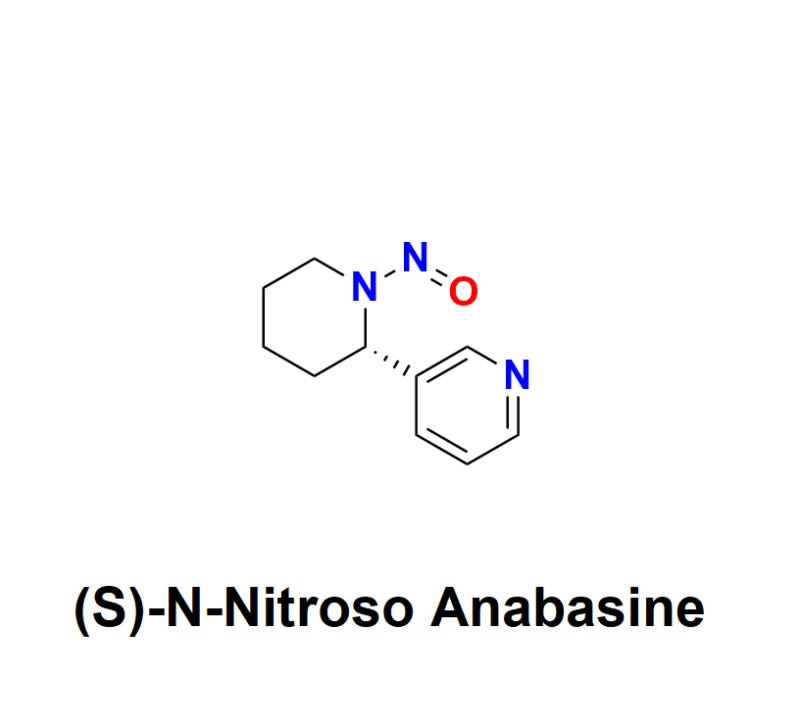
(S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How …
(S)-N-Nitroso Anabasine, a nitrosamine impurity, has raised significant safety concerns within the pharmaceutical industry. Recognized as a probable human carcinogen, this impurity has become a focal point for global regulatory agencies and manufacturers alike, urging a renewed emphasis on detection, prevention, and management.
Learn more about (S)-N-Nitroso Anabasine: https://aquigenbio.com/product/s-n-nitroso-anabasine/
What Is (S)-N-Nitroso Anabasine?
(S)-N-Nitroso Anabasine belongs to the family of nitrosamines, compounds formed through a chemical reaction known as nitrosation. This…
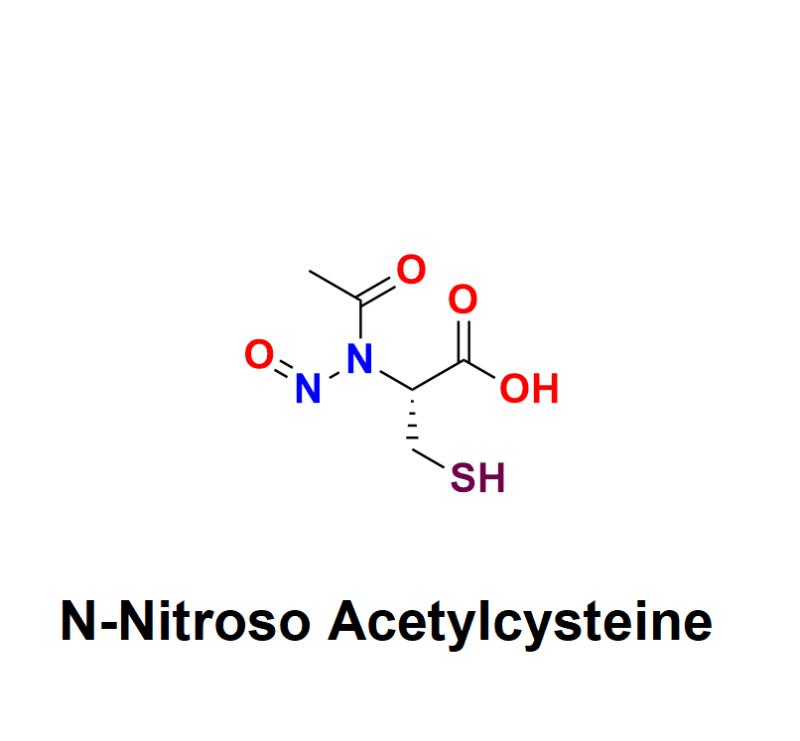
N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Haza …
N-Nitroso Acetylcysteine has emerged as a critical topic of concern in the pharmaceutical industry. As a member of the nitrosamine family, it is a potential impurity that poses significant health risks, including carcinogenicity, even in trace amounts. With increasing regulatory scrutiny on nitrosamine impurities, pharmaceutical manufacturers must address the presence of compounds like N-Nitroso Acetylcysteine to protect public health and ensure compliance with global standards.
Learn more about N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/…
More Releases for Buspirone
Analyzing the Global Buspirone Market: Size, Share & COVID-19 Impact Analysis
𝐈𝐧𝐭𝐫𝐨𝐝𝐮𝐜𝐭𝐢𝐨𝐧:
Buspirone, a medication primarily used to treat anxiety disorders, has garnered significant attention in the pharmaceutical industry. In this blog, we delve into the dynamics of the global Buspirone market, analyzing its size, share, and the impact of the COVID-19 pandemic on its trajectory. Additionally, we explore the market based on resin type, application, technology, end-use industry, and regional forecasts spanning from 2023 to 2031.
…
Buspirone Market Introducing New Industry Dynamics Through Swot Analysis 2021| B …
Los Angeles, United State, February 2021, – – QY Research offers an overarching research and analysis-based study on the global Buspirone market, covering growth prospects, market development potential, profitability, supply and demand, and other important subjects. The report presented here comes out as a highly reliable source of information and data on the global Buspirone market. The researchers and analysts who have prepared the report used an advanced research methodology…
Buspirone Market Size, Share, Development by 2025
LP INFORMATION recently released a research report on the Buspirone market analysis and elaborate the industry coverage, current market competitive status, and market outlook and forecast by 2025. Moreover, it categorizes the global Buspirone market by key players, product type, applications and regions,etc.
The main objective of this market research is to help the readers understand the structure of Buspirone market, market definition, overview, industry opportunities and trends, investment strategy…
Things You Will Like To Know About Buspirone Market| Bristol Myers Squibb, Apote …
"The new report has been added by qyresearch.com to provide detailed insight into the global Buspirone market. The study will help to get a better understanding about the Buspirone industry competitors, a channel for the distribution, Buspirone growth potential, potentially disruptive trends, Buspirone industry product innovations, market size value/volume (regional/country level, Buspirone industry segments), market share of top players/products.
Due to the pandemic, we have included a special section on the…
Buspirone Market Study for 2020 to 2026 Providing Information on Key Players, Gr …
Complete study of the global Buspirone market is carried out by the analysts in this report, taking into consideration key factors like drivers, challenges, recent trends, opportunities, advancements, and competitive landscape. This report offers a clear understanding of the present as well as future scenario of the global Buspirone industry. Research techniques like PESTLE and Porter's Five Forces analysis have been deployed by the researchers. They have also provided accurate…
Global Buspirone Hydrochloride Industry Market Analysis & Forecast 2018-2023
Market Research Report Store offers a latest published report on Buspirone Hydrochloride Industry Market Analysis and Forecast 2019-2025 delivering key insights and providing a competitive advantage to clients through a detailed report. This report focuses on the key global Buspirone Hydrochloride Industry players, to define, describe and analyze the value, market share, market competition landscape, SWOT analysis and development plans in next few years.To analyze the Buspirone Hydrochloride Industry with…