Press release
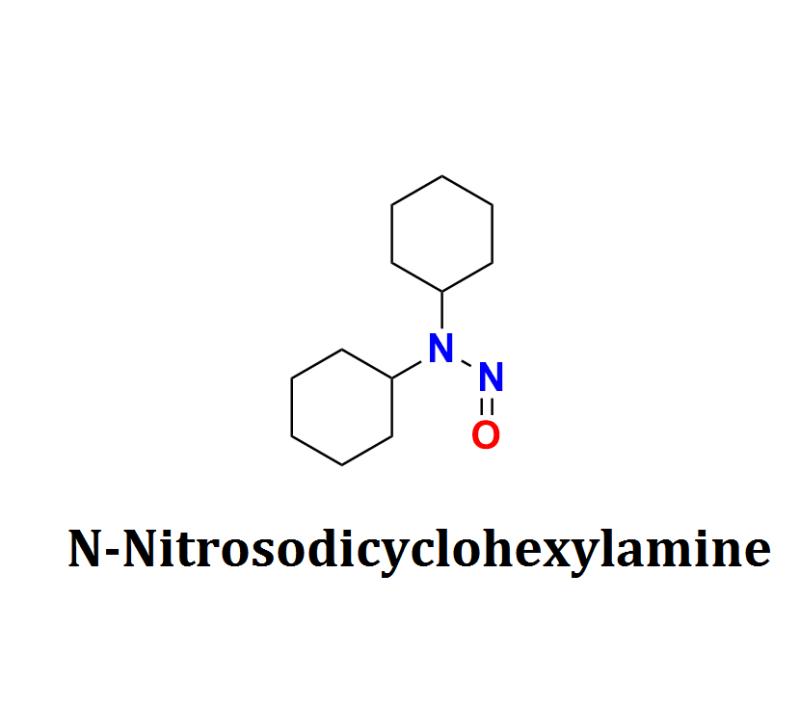
N-Nitrosodicyclohexylamine Contamination: Challenges for Pharmaceutical Manufacturers, Regulatory Compliance, and Safeguarding Patient Health in the Global Drug Supply Chain
The pharmaceutical industry is facing a new safety challenge with the detection of N-Nitrosodicyclohexylamine (NDCHA), a nitrosamine impurity found in various medications. The emergence of NDCHA raises significant concerns for both drug manufacturers and public health, prompting immediate regulatory scrutiny and safety measures.Understanding N-Nitrosodicyclohexylamine:
N-Nitrosodicyclohexylamine is a nitrosamine compound formed through the reaction of secondary or tertiary amines with nitrite under acidic conditions. Nitrosamines, in general, are classified as probable human carcinogens by the International Agency for Research on Cancer (IARC). NDCHA is particularly concerning due to its potential to form as a byproduct during pharmaceutical manufacturing or storage processes, especially in products containing amine-based compounds.
Learn more about N-Nitrosodicyclohexylamine - https://aquigenbio.com/product/n-nitrosodicyclohexylamine/
Impact on Drug Manufacturers:
The discovery of N-Nitrosodicyclohexylamine contamination in pharmaceutical products poses substantial challenges for drug manufacturers globally. Nitrosamines like NDCHA can unintentionally form during chemical synthesis or as degradation products, particularly in drug formulations that involve amines or certain solvents. Manufacturers are now under increased pressure to re-examine their entire production chain, from raw material sourcing to final product packaging, to ensure no NDCHA contamination occurs.
Manufacturers must take immediate steps to comply with the latest guidelines set forth by global regulatory bodies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and India's Central Drugs Standard Control Organization (CDSCO). These agencies have introduced strict limits on nitrosamine impurities, typically measured in nanograms, to ensure drug safety.
Regulatory Response and Compliance Requirements:
Given the carcinogenic nature of nitrosamines, regulatory authorities have set stringent safety thresholds for their presence in drugs. The FDA and EMA have issued detailed guidelines that require pharmaceutical companies to assess the risk of nitrosamine impurities, including NDCHA, in their products. Companies must identify potential sources of contamination, conduct rigorous testing, and implement control measures to mitigate risks.
Failure to meet these regulatory requirements could result in product recalls, facility inspections, penalties, or even the suspension of drug approvals. This is particularly critical for manufacturers of high-volume or life-saving medications, where N-Nitrosodicyclohexylamine contamination could disrupt global supply chains and impact patient access to essential treatments.
"The detection of N-Nitrosodicyclohexylamine in pharmaceutical products highlights the need for more advanced and rigorous impurity testing. As a leading supplier of impurity standards, Aquigen Bio Sciences is committed to supporting pharmaceutical manufacturers with the tools and expertise necessary to comply with stringent regulatory requirements. Our goal is to help ensure the highest levels of safety and quality in drug production, ultimately protecting patient health. We continue to innovate in the field of impurity standards, providing accurate solutions to detect and control even the most challenging impurities like NDCHA", said the CEO of Aquigen Bio Sciences.
Get in Touch with Us - https://aquigenbio.com/contact-us/
Steps to Address N-Nitrosodicyclohexylamine Contamination:
1. Risk Assessment and Testing: Manufacturers must conduct comprehensive risk assessments to identify potential sources of N-Nitrosodicyclohexylamine contamination throughout the manufacturing process. Advanced analytical methods, such as gas chromatography-mass spectrometry (GC-MS), should be employed to detect even trace amounts of NDCHA in drug formulations.
2. Process Optimization: Manufacturers should evaluate and, if necessary, modify their manufacturing processes to eliminate conditions that could lead to NDCHA formation. This may involve switching to alternative raw materials, refining storage conditions, or adjusting synthesis pathways.
3. Collaborating with Regulators: Proactive engagement with regulatory authorities is crucial. Manufacturers should maintain transparency and provide detailed data on their efforts to mitigate NDCHA risks.
4. Patient Communication: Establishing a communication framework that keeps patients informed about potential contamination risks and remedial measures is essential. Manufacturers must issue timely updates to patients, healthcare providers, and pharmacies to ensure public safety.
The Bottom Line:
The identification of N-Nitrosodicyclohexylamine contamination in pharmaceutical products underscores the urgent need for drug manufacturers to enhance their vigilance and ensure patient safety. By adopting robust testing and process optimization strategies, manufacturers can prevent NDCHA formation, comply with regulatory standards, and protect the health of patients worldwide.
Similar Trending Products:
1) 2-Nitroso-1-Pentylpiperazine - https://aquigenbio.com/product/2-nitroso-1-pentylpiperazine/
2) (S)-N-Nitroso Anabasine - https://aquigenbio.com/product/s-n-nitroso-anabasine/
3) N-Nitroso Phenylephrine - https://aquigenbio.com/product/n-nitroso-phenylephrine-2/
Contact Us:
Aquigen Bio Sciences
281/1, Plot No 41,
Hinjawadi - Pirangut Rd,
Kasar Amboli, Pirangut,
Pune, Maharashtra 412108
Phone: +91 7030123794
Email: bd@aquigenbio.com
Visit: www.aquigenbio.com
About Aquigen Bio Sciences:
Aquigen Bio Sciences is a research organization based in Pune, India, specializing in a wide range of scientific research and development services. With a focus on innovation and excellence, Aquigen Bio Sciences delivers high-quality solutions to meet the complex needs of the pharmaceutical, environmental, and industrial sectors. The company is dedicated to advancing scientific knowledge and improving safety through cutting-edge research and technology.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release N-Nitrosodicyclohexylamine Contamination: Challenges for Pharmaceutical Manufacturers, Regulatory Compliance, and Safeguarding Patient Health in the Global Drug Supply Chain here
News-ID: 3664556 • Views: …
More Releases from Aquigen Biosciences
Precision Standards for Oncology Research: Exploring Abemaciclib Impurity 1 and …
In the ever-evolving field of targeted cancer therapy, Abemaciclib has emerged as a pivotal agent in the treatment of hormone receptor-positive (HR+), HER2-negative advanced or metastatic breast cancer. As researchers and pharmaceutical developers continue to innovate in oncology, the importance of impurity profiling and the availability of reliable Abemaciclib impurity standards has never been greater.
At the forefront of pharmaceutical impurity standards, Aquigen Bio is proud to support global manufacturers, CROs,…
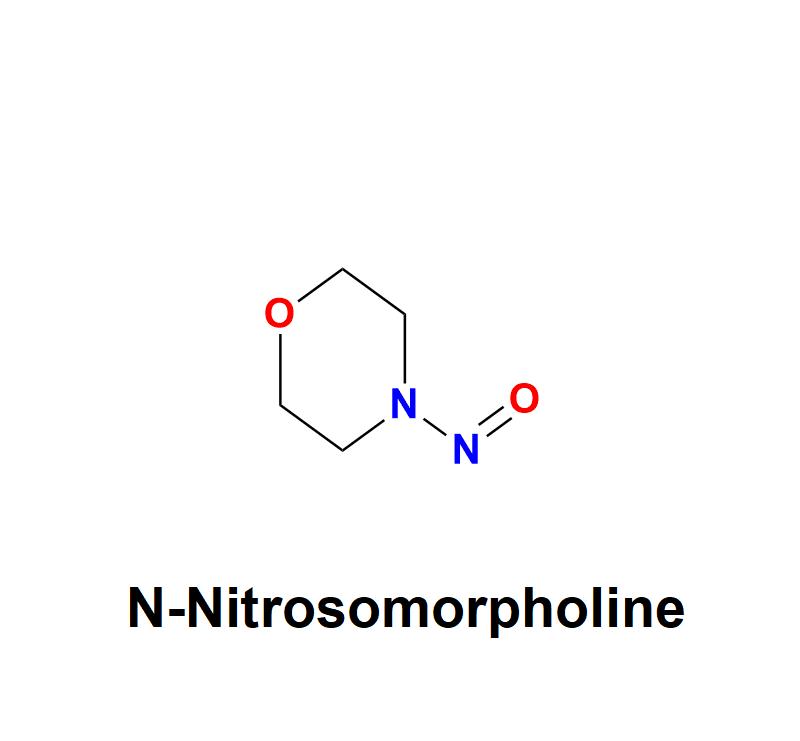
N-Nitrosomorpholine: Addressing Pharmaceutical Safety Challenges with Aquigen Bi …
N-Nitrosomorpholine, a compound belonging to the nitrosamine family, has garnered significant attention in the pharmaceutical and healthcare industries due to its potential carcinogenic risks. This chemical impurity, often found as a byproduct in manufacturing processes, poses serious challenges to drug safety and human health, necessitating stringent monitoring and control measures from pharmaceutical companies.
Learn more about N-Nitrosomorpholine: https://aquigenbio.com/product/n-nitrosomorpholine/
Understanding N-Nitrosomorpholine:
N-Nitrosomorpholine is a nitrosamine impurity characterized by its chemical structure, which includes…
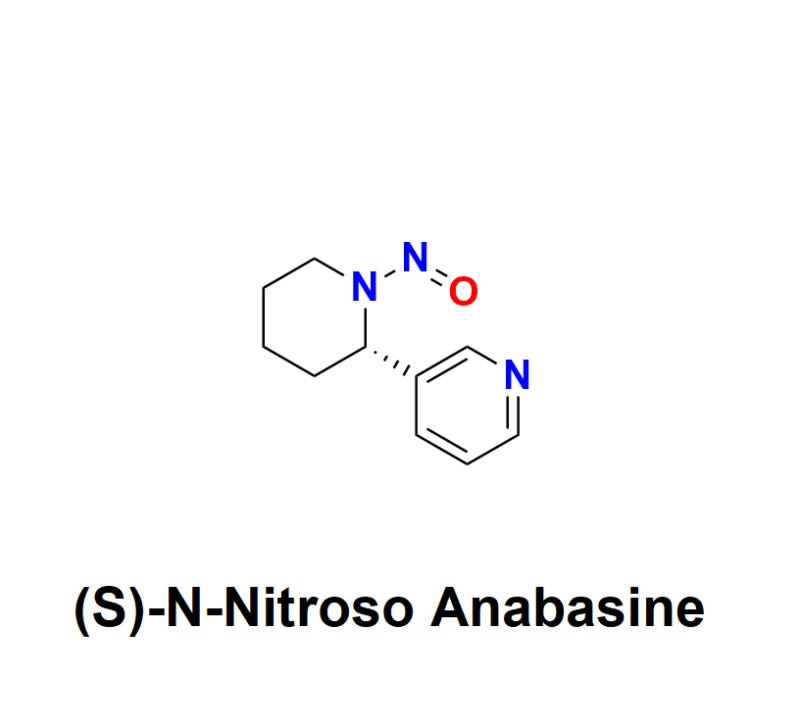
(S)-N-Nitroso Anabasine: Understanding the Risks, Regulatory Challenges, and How …
(S)-N-Nitroso Anabasine, a nitrosamine impurity, has raised significant safety concerns within the pharmaceutical industry. Recognized as a probable human carcinogen, this impurity has become a focal point for global regulatory agencies and manufacturers alike, urging a renewed emphasis on detection, prevention, and management.
Learn more about (S)-N-Nitroso Anabasine: https://aquigenbio.com/product/s-n-nitroso-anabasine/
What Is (S)-N-Nitroso Anabasine?
(S)-N-Nitroso Anabasine belongs to the family of nitrosamines, compounds formed through a chemical reaction known as nitrosation. This…
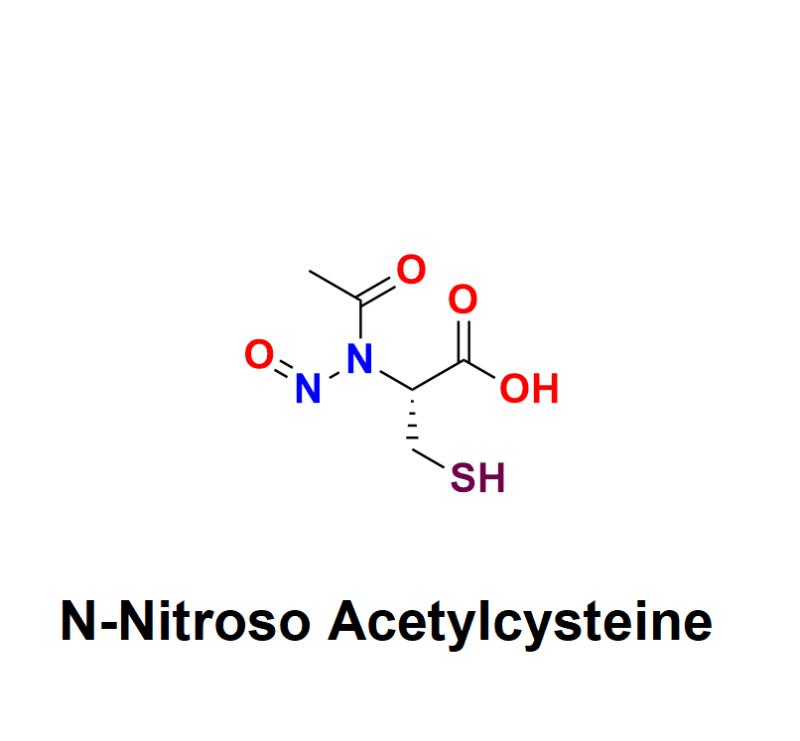
N-Nitroso Acetylcysteine: A Critical Concern in Pharmaceuticals - Exploring Haza …
N-Nitroso Acetylcysteine has emerged as a critical topic of concern in the pharmaceutical industry. As a member of the nitrosamine family, it is a potential impurity that poses significant health risks, including carcinogenicity, even in trace amounts. With increasing regulatory scrutiny on nitrosamine impurities, pharmaceutical manufacturers must address the presence of compounds like N-Nitroso Acetylcysteine to protect public health and ensure compliance with global standards.
Learn more about N-Nitroso Acetylcysteine: https://aquigenbio.com/product/n-nitroso-acetylcysteine/…