Press release
Movement Disorder Market Outlook with Ongoing Trends, Top Players Profile Analysis and Forecast 2017-2022
Growing prevalence of the neurological diseases is expected to contribute towards growth of the global movement disorder market. As manufacturers are increasingly focusing on development and innovation of products, growth of the global market is expected to remain positive. This Fact.MR report analyzes the expansion of global movement disorder market till date, and provides key insights on the growth of the market during the forecast period, 2017-2026.Request Free Sample Report- https://www.factmr.com/connectus/sample?flag=S&rep_id=329
The next section offers an overview of the global movement disorder market. This comprises an introduction to the market, along with a standard definition of the product – movement disorder. In this section, market value and year-over-year growth is offered to the readers. Year-over-year growth provides readers with a broader view of growth patterns over the forecast period.
The report’s succeeding section focuses on drivers, restraints and key trends from macroeconomic, demand, and supply perspectives. Impact analysis of weighted average model-based growth drivers is contained in the report for better provision of decision-making insights to clients.
In order to offer readers with up-to-date information about the latest advancements in the global movement disorder market, the report provides updates about market opportunities, which can benefit leading manufacturers of movement disorder. With continuous evolution of the healthcare sector, keeping a record of latest developments and trends is fundamental for movement disorder manufacturers to formulate key business strategies. Detailed insights about raw material sourcing, supply chain, regulatory approvals, pricing analysis, list of distributors, and cost structure are provided in this section.
FDA Approval to Boost Sales of Medications
Medications and treatments approved by FDA is also expected to impact growth of the global movement disorder market. Xadago (safinamide) tablets and Ingrezza capsules are two drugs that have recently cleared the FDA pipeline. Ingrezza capsules have recently received approval for treatment of dyskinesia and Xadago (safinamide) tablets have received approval of Parkinson’s disease. In addition, the FDA has also approved implantation of brain to reduce the tremor symptoms. These factors are further expected to boost growth of the global movement disorders market.
Advanced Features to Impact Growth of the Global Market
In order to gain a competitive edge, various companies operating in the global movement disorder market are focusing on product innovation and developments. Manufacturers are mainly concentrating on incorporating advanced technology in the medical devices for faster recovery and improved results. Equipped with the advanced features such as sensors, 360-degree stabilizing solutions and cloud technology. Parkinson’s spoon equipped with advanced technological features enable the end users to retrieve and store information regarding the status of the unwanted tremors and generates the algorithm of tremor patterns for optimal stabilization. Moreover, the manufacturers operating in the global market are offering the electronic stabilizing handle, which comprises everyday spoon, spork, soup spoon and a fork. Bound to these factors, the global movement disorders market is expected to witness significant growth during the forecast period.
Browse Full Report with TOC-https://www.factmr.com/report/329/movement-disorder-market
On the other hand, various factors continue to impact growth of the global movement disorders market negatively. Lack of investment in research and development will continue to inhibit growth of the global movement disorder market. In addition, slow approval of the medications developed is further likely to impact growth of the global market negatively. Moreover, poor method of therapeutics conducted continues to pose significant challenges towards growth of the global market.
Parkinson’s disease to Represent a Leading Segment
Growing need for medical devices with the uninterrupted functioning for treatment of the neurological disorders has led to an upsurge in adoption of rechargeable deep brain stimulator devices. On the basis of product type, the rechargeable deep brain stimulator devices segment is expected to generate significant revenues, accounting for a value of over US$ 500 Mn by the end of 2026. However, the non-rechargeable deep brain stimulator devices product type segment is expected to register the highest CAGR during the forecast period.
Based on end user, the hospitals segment is expected to represent the highest revenue growth, recording a value of less than US$ 100 Mn by the end of 2017. On the other hand, the clinics end user segment is expected to register a robust through 2026.
By application, the Parkinson’s disease segment is expected to represent a significant revenue growth, accounting for a value of over US$ 400 Mn by the end of 2026. In contrary to this, the dystonia application segment is expected to register a healthy CAGR throughout the forecast period.
Competition Tracking
Leading market players operating in the global movement disorder market include Medtronic Plc, Abbott Laboratories, Boston Scientific Corporation and Mayo Clinic.
Check Discount- https://www.factmr.com/connectus/sample?flag=D&rep_id=329
About Fact.MR
Fact.MR is a fast-growing market research firm that offers the most comprehensive suite of syndicated and customized Healthcare market research reports. We believe transformative intelligence can educate and inspire businesses to make smarter decisions. We know the limitations of the one-size-fits-all approach; that's why we publish multi-industry global, regional, and country-specific research reports.
Contact Us
Fact.MR
Suite 9884
27 Upper Pembroke Street,
Dublin 2, Ireland
Telephone: +353-1-6111-593
Email: sales@factmr.com/
Web: https://www.factmr.com/
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Movement Disorder Market Outlook with Ongoing Trends, Top Players Profile Analysis and Forecast 2017-2022 here
News-ID: 921725 • Views: …
More Releases from Fact.MR

Organic Rice Syrup Market is forecasted to increase at a CAGR of 5.1% and US$ 1. …
The global Organic Rice Syrup Market is projected to expand steadily over the coming decade, driven by rising consumer demand for natural, clean-label sweeteners and growing awareness of health and wellness trends. Industry analysts estimate that the organic rice syrup market, valued at approximately USD 450 million in 2025, is expected to reach nearly USD 880 million by 2035, registering a compound annual growth rate (CAGR) of about 7.1% during…
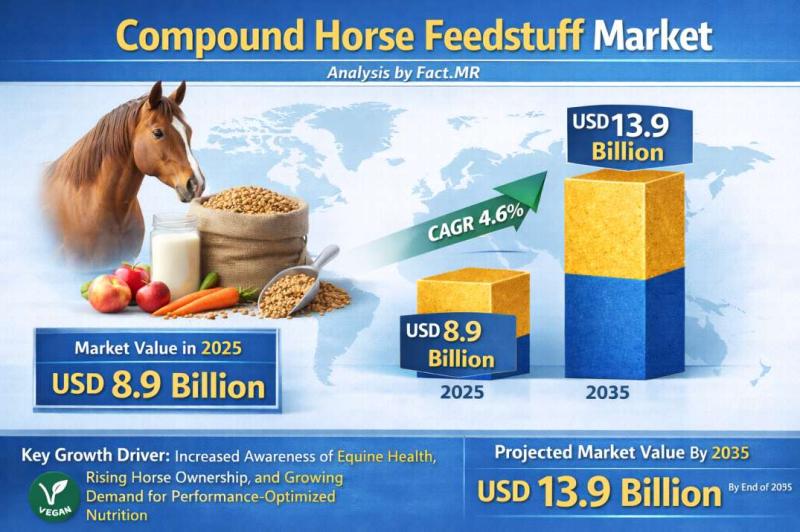
Compound Horse Feedstuff Market is Estimated to Grow at a CAGR of 4.6%, Reaching …
The global compound horse feedstuff market is galloping toward steady growth, projected to expand from a valuation of USD 3.8 billion in 2026 to approximately USD 5.4 billion by 2036. This represents a compound annual growth rate (CAGR) of 3.6% over the ten-year forecast period.
The market is being driven by the "humanization" of equine companions, the professionalization of equestrian sports, and a significant shift toward specialized performance nutrition that…

Nutritional Yeast Market Forecasted CAGR is 7.1% by 2035 | Fact.MR Report
The global nutritional yeast market is experiencing a significant surge in demand, projected to grow from a valuation of USD 515.2 million in 2026 to approximately USD 1.2 billion by 2036. This represents a robust compound annual growth rate (CAGR) of 8.8% over the ten-year forecast period.
The market is being propelled by the global explosion of plant-based diets and the "clean-label" movement, with nutritional yeast emerging as the primary…
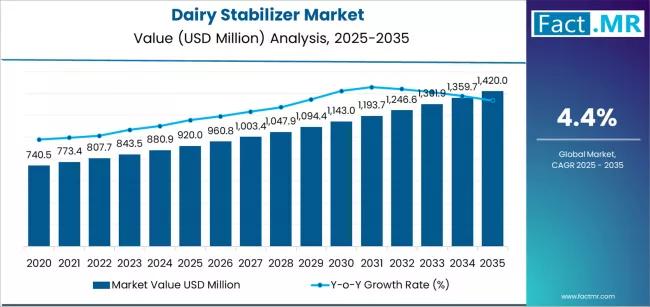
Dairy Stabilizer Market is Expected to Reach USD 1,420.0 million by 2035 | Resea …
The global Dairy Stabilizers Market is projected to sustain solid growth over the next decade as consumer demand for high-quality dairy and dairy-based products continues to expand across foodservice and retail sectors. Industry analysts estimate that the dairy stabilizers market, valued at approximately USD 2.4 billion in 2025, is expected to reach around USD 4.3 billion by 2035, registering a compound annual growth rate (CAGR) of about 6.5% during the…
More Releases for FDA
DreaMed receives 5th FDA Clearance
TEL AVIV, Israel: DreaMed Diabetes LTD. ("DreaMed" or the "Company"), developer of the endo.digital Clinical Decision Support System announced today that it has received its 5th U.S Food and Drug Administration (FDA) clearance that expands the scope of AI enhanced treatment recommendations to patients on fixed meal insulin regimens. endo.digital is the first decision support system that has been cleared to assist healthcare providers in the management of diabetes…
FDA Compliant Blood Storage and Preservation
Accsense Monitoring System Automates Data Archive and Alarming
CAS DataLoggers provided the temperature alarming and monitoring system to a hospital blood bank looking to replace their old paper chart recorders as they became unreliable and spare parts were harder to find. For proper blood storage and preservation, the lab’s medical units needed to maintain storage temperatures between 2°C to 6°C (36°F to 43°F), given the perishability of blood components. The facility…
FDA grants orphan drug status to Vicore
US Food and Drug Administration has awarded Vicore Pharmaceuticals with orphan Drug designation for the treatment of Idiopathic Pulmonary Fibrosis (IPF). FDA’s Orphan Drug Designation program provides certain incentives for companies developing therapeutics to treat rare diseases or conditions, defined as those affecting less than 200,000 individuals in the U.S. A drug candidate and its sponsor must meet several key criteria in order to qualify for, and obtain, orphan drug…
New FDA Design Control Training Courses
Salt Lake City, Utah - February 23 2017 - Procenius Consulting is a medical device consulting firm specializing solely in medical device design controls regulation (21 CFR 820.30).
Announcing New Design Control Training Courses
Procenius Consulting has just launched two new training courses covering basic and advanced topics of medical device design control regulation. These courses focus on compliance, practical implementation and industry best practices techniques for developing or improving a…
fda online training
GRC Training Solutions provides end-to-end FDA compliance solutions for those companies who want to maximize security, minimize operational costs, improve staff productivity and stay on top of all their compliance documentation.
GRC Training Solutions boasts a team of experts and specialists who have a proven track record in working with the biotechnology, medical device, diagnostic and pharmaceutical fields. Our team will work with you closely and develop solutions that meet…
FDA online training
Description:
Device firms, establishments or facilities that are involved in the production and distribution of medical devices intended for use in the U.S are required to register annually. Most establishments that are required to register with the FDA are also required to list the devices that are made there and the activities that are performed on those devices. Initially, FDA issued a 28-page Proposed Rule that would amend its regulations regarding…
