Press release
European GDP Association nominates new Advisory Board Member
So far the Board of the European GDP Association comprised four members from the industry side, supported by a representative from the Finnish Medicines Agency FIMEA. Now a new Board Member has been nominated on the industry side.Dr Laura Ribeiro, who is a Responsible Person at ID Logistics (formerly Logiters) in Portugal, has recently accepted her nomination as the fifth member of the GDP Association's Board. Prior to her position at ID Logistics she was R&D and regulatory affairs manager in the pharmaceutical industry, invited Professor at Escola Superior de Tecnologias da Saúde de Coimbra and later deputy Technical Director at Logiters, being responsible for quality and GDP related activities.
"I am pleased that Laura accepted our invitation to join the ECA GDP Board" said Dr Afshin Hosseiny, GDP Association Advisory Board Chairman. "GDP is a developing subject for the pharma industry, and I am really excited that our team can have a positive influence in the future both on developing industry understanding and providing supporting guidelines for the practitioners. Laura's professional experience will be a very valuable addition to our team." "I felt honoured about the invitation to join the Association's Advisory Board" Dr Laura Ribeiro stated upon her nomination. "And I very much look forward to working with the other members to help advance the group".
Read more about the Advisory Board Members of the European GDP Association at www.good-distribution-practice-group.org.
About the GDP Association
The Good Distribution Practice Group was founded in March 2013 – then as an Interest Group. In November 2016 it was transferred to the European GDP Association and today counts close to 1.900 members from almost 80 countries. The Association represents all stakeholders e.g. from Pharmaceutical Industry, Authorities and Logistic Providers and supports them by providing them with information about the implementation of GDP. Although it has a strong focus on European GDP Regulations and their implementation into the different EU member states, it also supports harmonisation of the requirements with other regions of the world - wherever medicinal products, APIs, Excipients, etc are distributed.
The Association is a not for profit organisation under the umbrella of the ECA Foundation. Due to this support, the European GDP Association is able to offer a free membership to all individuals involved in Good Distribution Practice. To find out more please visit www.good-distribution-practice-group.org.
About the ECA Foundation
Founded as an independent not for profit organisation in 1999, the ECA Foundation is Europe’s leading association for pharmaceutical Quality Assurance and GMP compliance. Its goal is “the exchange of information between representatives of the industry, regulatory authorities and universities in the field of pharmaceutical quality assurance, especially with regard to the area of Good Manufacturing Practice (GMP).” For that purpose the organisation has set up several Interest Groups and Working Groups as well as an Academy for advanced education. Individuals can become member of the ECA Groups and in the Academy. In total more than 7.000 professionals have joined the ECA organisation. Its largest group, the European Qualified Person Association, alone counts close to 2.400 QPs. The ECA Foundation Groups developed a range of tools like several Good Practice Guides. For further information on the ECA Foundation please visit www.eca-foundation.org.
ECA Foundation
Wolfgang Heimes
Administration Manager
P.O. Box 10 21 68
69011 Heidelberg
Germany
heimes@gmp-compliance.org
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release European GDP Association nominates new Advisory Board Member here
News-ID: 623457 • Views: …
More Releases from ECA Foundation

Paul-Ehrlich-Institute and ECA Foundation jointly organise 2-day Workshop – Go …
Vaccine development and deployment are crucial to control the COVID-19 pandemic. Platform technologies have enabled rapid development of vaccines with demonstrated efficacy against COVID-19. However, large-scale manufacturing is both a prerequisite and a challenge in meeting the global demand. This implies upscaling of manufacturing, meeting Good Manufacturing Practice (GMP) requirements, and switching of manufacturing in existing facilities.
The workshop organised by the Paul-Ehrlich-Institut and the ECA Foundation will address these…

ECA Foundation added to EMA List of Stakeholder Organisations
The ECA Foundation is now listed in the European Medicines Agency's (EMA) list of eligible industry stakeholder organisations (www.ema.europa.eu/en/documents/other/list-eligible-industry-stakeholder-organisations_en.pdf). Pre-requisite for this entry is the listing in the EU's Transparency Register (http://ec.europa.eu/transparencyregister/public/consultation/searchControllerPager.do?declaration=ECA%20Foundation&search=search&locale=en#en) - where the ECA has been registered since January 2019.
The ECA Foundation is the leading European organisation with regard to pharmaceutical Quality Assurance and GMP compliance.
The ECA Foundation is a not for profit organisation. The ECA is headed…
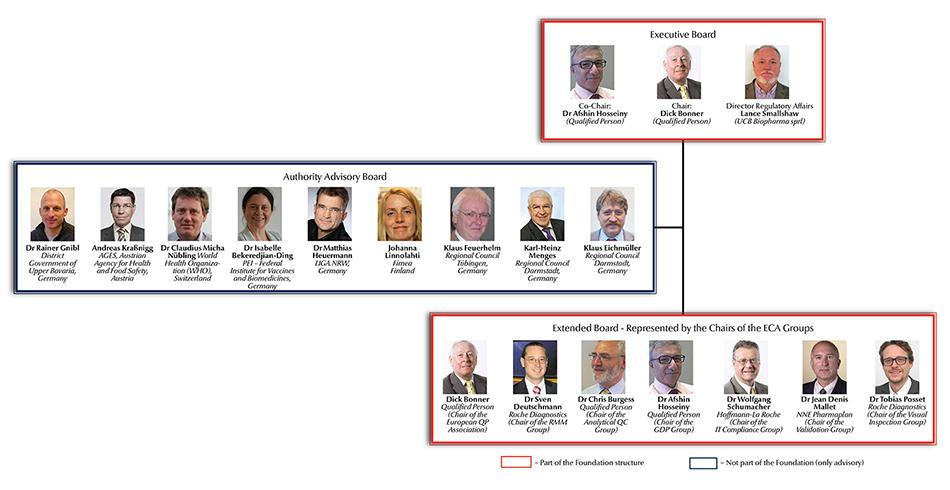
ECA Foundation announces Independent Authority Board (IAB) Members
Since June 2015 the ECA Foundation, the leading European organisation in the pharmaceutical quality assurance and GMP compliance environment, has been managed by an Executive Board and an Extended Board. In addition, the ECA set up an Advisory Committee encompassing authority representatives. Now the Foundation announced the members of the Independent Authority Board (IAB).
It is one of the ECA Foundation’s major objectives to discuss GMP developments with the stakeholders…
More Releases for GDP
Key Factors Influencing Finland's GDP Growth: Economic Indicators and Insights
Finland GDP growth in recent years has been shaped by a combination of domestic economic indicators and global market dynamics. As highlighted by Financist.fi, https://financist.fi/ Finland's economy reflects a balance between innovation-driven expansion and structural challenges such as demographic shifts and external economic pressures. Understanding the key indicators behind Finland GDP growth is essential for investors, policymakers, and researchers aiming to evaluate the country's economic direction.
Introduction to Finland GDP and…
China's GDP Surprises on Buoyant Demand
Economists are becoming more positive on this year's economic prospects.
Image: https://www.getnews.info/uploads/00fca63e211ffb8a1956472d8f5965db.jpg
In its April World Economic Outlook (WEO), the IMF now forecasts aggregate GDP growth to reach 3.2 percent this year, up 0.3 percentage points from its October projection. The Fund describes the global economy as "surprisingly resilient [https://www.imf.org/en/Publications/WEO/Issues/2024/04/16/world-economic-outlook-april-2024?cid=ca-com-homepage-SM2024]" for the way in which it has continued to grow despite the higher interest rates needed to rein in inflation.
In this vein,…
Quantta Analytics collaborates with NITI Aayog to predict GDP growth rate of Ind …
Traditionally released by the Central Statistical Office (CSO) of the Government of India, Quantta has used its extensive database on the Indian Economy to provide an advance signal to senior leaders in Government about the state of the economy.
Using data from several sectors, that drive growth, it predicted a First Quarter growth rate of 7.83% indicating that the Indian Economy is firing on all cylinders. The actual numbers released…
IATA's CEIV-Pharma Certification or HSA's GDP Certification?
Players throughout the pharmaceutical supply chain are experiencing growing pressure to gain accreditation for handling the high-value, temperature-sensitive cargo, as having a certification becomes more commonplace, rather than an exceptional feat in the industry.
There are currently 2 different measures of standards, one is IATA's CEIV-Pharma Certification and the other is HSA's GDP Certification.
There are major players on both sides of the fence which hold – GDP-compliant certificates and CEIV-pharma certifications.…
US GDP Up 1.7% Shattering Economists Predictions
Aug. 01, 2013 - HONG KONG -- On Wednesday the US Commerce Department released its highly anticipated report on GDP, stating that in the second quarter the US economy grew 1.7% driven by an increase of 1.8% in consumer spending and 9% in business investment. New home construction was another important driver of growth, with investment rising 13.4%, the fourth consecutive increase. This gain beat expectations of a 1%…
Taiwan's GDP growth to hit fastest pace in 21 years
By Audrey Wang
The Economist Intelligence Unit expects Taiwan’s gross domestic product to grow by 9.2 percent in 2010, the fastest rate in 21 years, according to the Council for Economic Planning and Development Sept. 24.
The EIU report noted that Taiwan’s economy grew by an average of 13.1 percent in the first two quarters of 2010. This stellar performance, the EIU said, “largely reflects the global economy’s return to…