Press release
Global Flu Vaccine Market Size, Share & Forecast: Rising Seasonal Immunization Demand Drives Industry Expansion Across the U.S., Europe, and Asia-Pacific
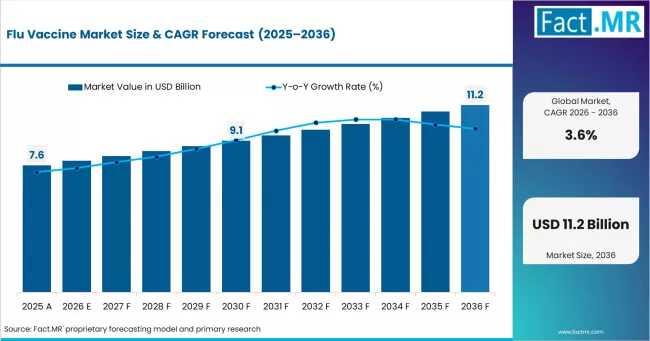
According to Fact.MR analysis, the global flu vaccine market is valued at approximately USD 7.6 billion in 2025 and is projected to reach USD 11.2 billion by 2036, expanding at a CAGR of 3.6% between 2026 and 2036. While growth rates remain moderate compared with pandemic-era vaccine markets, the category continues to attract sustained investment due to its recurring demand profile, strategic public health importance, and evolving technology landscape.Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=B&rep_id=9389
Flu Vaccine Market Enters a New Strategic Phase as Emerging Economies and Next-Generation Platforms Reshape Global Demand
The global flu vaccine market is entering a period of structural transition rather than explosive expansion. Long anchored by annual seasonal immunization cycles and government procurement programs, the sector is now being reshaped by two parallel forces: rising vaccination adoption in emerging economies and accelerating innovation in vaccine manufacturing technologies.
Quick Stats
Global market value (2025): USD 7.6 billion
Estimated market size (2026): USD 7.9 billion
Forecast market size (2036): USD 11.2 billion
Forecast CAGR (2026-2036): 3.6%
Absolute dollar opportunity: USD 3.3 billion
Leading product segment (2026): Inactivated Influenza Vaccines with 41.8% share
Leading end-user segment (2026): Public Immunization Programs with 52.1% share
Fastest-growing markets: India (5.4% CAGR) and China (4.7% CAGR)
Seasonal Vaccination Is Becoming a Strategic Public Health Infrastructure Market
Unlike discretionary pharmaceutical categories, influenza vaccines operate within a recurring annual demand cycle that is heavily reinforced by government-funded immunization programs. This creates what industry analysts increasingly describe as a "non-discretionary procurement floor" across major healthcare systems.
That demand stability is especially important at a time when healthcare procurement teams are facing budget scrutiny, supply-chain recalibration, and post-pandemic reassessment of vaccine manufacturing resilience.
The market's growth trajectory is being sustained less by dramatic price escalation and more by widening vaccination coverage, particularly in countries where seasonal influenza immunization has historically remained limited.
India, China, and Brazil are emerging as the most strategically important expansion markets because flu vaccination penetration remains relatively low compared with mature Western healthcare systems. As public health agencies expand outreach programs and institutional vaccination adoption increases, annual dose volumes are expected to rise steadily over the next decade.
At the same time, mature markets such as the United States, Germany, Japan, and South Korea are shifting toward premium vaccine formulations designed for aging populations and higher-risk patient groups.
Technology Platforms Are Quietly Redefining Competitive Advantage
The most consequential transformation underway in the flu vaccine market may not be geographic expansion, but manufacturing modernization.
Traditional egg-based production still dominates global supply, supported by decades of regulatory familiarity and established production infrastructure. However, manufacturers are increasingly investing in cell-based, recombinant, and mRNA technologies that promise faster production timelines, improved strain matching, and stronger pandemic responsiveness.
The strategic significance of these platforms extends beyond seasonal flu prevention.
Manufacturers capable of rapidly adjusting vaccine strains and scaling production capacity are likely to gain stronger positioning in future pandemic preparedness frameworks being developed by governments worldwide.
mRNA-based influenza vaccines, in particular, represent a potentially disruptive force over the long term. Developers including Moderna and Pfizer are advancing flu-focused mRNA programs and combination flu-COVID vaccine candidates through clinical development pipelines.
These technologies could significantly reduce production lead times compared with conventional egg-based methods while also simplifying annual immunization schedules through combined respiratory vaccines.
For procurement leaders and healthcare systems, the shift raises broader questions around manufacturing flexibility, cold-chain readiness, and future vaccine portfolio strategies.
Inactivated Vaccines Maintain Dominance Despite Innovation Pressure
Despite increasing attention on next-generation platforms, inactivated influenza vaccines are expected to retain the market's leading position through the forecast period.
The segment is projected to account for 41.8% of global market share in 2026, supported by broad regulatory approval across age groups, proven safety performance, and entrenched integration into national vaccination programs.
The continued dominance of inactivated vaccines illustrates an important reality within vaccine markets: innovation adoption is often gradual due to regulatory complexity, procurement conservatism, and healthcare infrastructure dependencies.
For many public immunization systems, reliability, manufacturing consistency, and supply security remain more important than rapid technological transition.
Nevertheless, premium formulations are emerging as a critical value-growth segment. High-dose and adjuvanted vaccines targeted at elderly and immunocompromised populations are generating higher-margin opportunities for manufacturers facing pricing pressure in tender-driven markets.
This dynamic is particularly visible in North America, Japan, and parts of Europe where aging demographics are increasing demand for specialized influenza protection.
Public Immunization Programs Continue to Control Market Economics
Public immunization programs are expected to account for 52.1% of end-user demand in 2026, reinforcing the sector's dependence on institutional procurement cycles.
Government purchasing frameworks remain the primary determinant of global flu vaccine volumes, pricing stability, and supplier market share.
This creates a structurally different competitive environment compared with many commercial pharmaceutical markets.
Winning large-scale public contracts often depends on manufacturing scale, supply reliability, distribution infrastructure, and long-term procurement relationships rather than consumer marketing.
As a result, the competitive landscape remains highly concentrated among a relatively small group of multinational vaccine manufacturers.
Companies including Sanofi Pasteur, GlaxoSmithKline, and CSL Limited through its Seqirus division continue to dominate global procurement volumes through extensive manufacturing capacity and established government relationships.
However, emerging-market vaccine producers are gaining relevance in cost-sensitive regions.
Indian manufacturers such as Bharat Biotech and Serum Institute of India are expanding production capabilities and strengthening their position in regional immunization supply chains.
Chinese producers including Sinovac Biotech are also increasing competitive pressure through domestic supply expansion and cost-efficient production models.
Asia-Pacific Is Becoming the Industry's Most Important Growth Engine
Asia-Pacific is forecast to remain the fastest-growing regional market throughout the next decade, driven primarily by healthcare infrastructure expansion and rising awareness of preventive immunization.
India leads projected growth globally with a CAGR of 5.4% through 2036.
Several structural factors support this momentum:
Expansion of public immunization infrastructure
Growth in domestic vaccine manufacturing capacity
Increasing seasonal flu awareness
Rising private-market vaccination adoption through hospitals and pharmacy networks
China follows closely with projected growth of 4.7%, supported by government investment in seasonal influenza prevention and sustained post-pandemic public health awareness.
Brazil also stands out among high-growth markets, benefiting from established national vaccination campaigns and expanding pharmacy-based immunization channels.
Meanwhile, mature markets are entering a more replacement-driven growth phase.
The United States is expected to grow at 3.4%, supported largely by premium vaccine adoption and strong retail pharmacy accessibility. Germany and Japan are projected to expand at 2.9% and 2.6% respectively, reflecting stable but mature vaccination ecosystems.
Margin Pressure and Supply Complexity Remain Persistent Challenges
Despite dependable demand patterns, the flu vaccine market faces several structural constraints.
Tender-based procurement systems continue to compress manufacturer margins in many developed markets, limiting revenue acceleration even as vaccination volumes remain stable.
At the same time, annual strain variability presents ongoing commercial and reputational risk. Vaccine effectiveness can fluctuate significantly between seasons depending on how accurately manufacturers predict circulating influenza strains.
Lower-than-expected efficacy seasons may weaken public confidence and reduce voluntary vaccination uptake in subsequent years.
Cold-chain infrastructure also remains a major operational bottleneck in developing economies, particularly in rural and underserved regions where healthcare logistics networks remain fragmented.
For manufacturers seeking expansion in emerging markets, distribution capability is becoming nearly as important as manufacturing scale itself.
Competitive Positioning Is Increasingly Defined by Platform Flexibility
The next decade may ultimately reward companies capable of balancing three priorities simultaneously:
Large-scale seasonal vaccine supply
Premium formulation development
Flexible next-generation manufacturing platforms
This balancing act explains the industry's growing investment in cell-based and recombinant production technologies, which offer dual-use advantages for both seasonal influenza and pandemic preparedness.
Recent developments underscore this shift.
Moderna advanced its combined flu-COVID mRNA vaccine candidate through Phase 3 trials in 2025, with regulatory submissions anticipated in 2026. Meanwhile, Sanofi Pasteur expanded cell-based manufacturing capacity in the United States and Europe, and Seqirus introduced updated adjuvanted influenza products targeting elderly populations.
For investors and healthcare strategists, these moves indicate that the future competitive landscape may be shaped less by vaccine volume alone and more by manufacturing adaptability, speed-to-market capability, and portfolio diversification.
Executive Takeaways
The flu vaccine market remains fundamentally resilient because annual immunization cycles create recurring institutional demand.
Emerging economies, particularly India and China, are becoming the industry's most important long-term volume growth drivers.
Inactivated vaccines will continue dominating global supply, but cell-based, recombinant, and mRNA technologies are steadily reshaping future competition.
Premium formulations targeting elderly and high-risk populations represent one of the strongest margin-expansion opportunities in mature markets.
Government procurement structures continue to favor large-scale manufacturers with proven supply reliability and regulatory credibility.
Pandemic preparedness investments are accelerating infrastructure modernization across vaccine manufacturing ecosystems globally.
Outlook: Stability With Strategic Transformation
The global flu vaccine industry is unlikely to experience the hypergrowth dynamics associated with pandemic-era vaccine markets. Instead, its evolution is expected to be defined by gradual but strategically meaningful transformation.
Recurring seasonal demand provides unusual market stability for manufacturers and investors, while emerging-market immunization expansion creates long-term volume upside. At the same time, manufacturing modernization and mRNA platform development could fundamentally alter production economics and competitive positioning over the next decade.
For healthcare systems, vaccine producers, and institutional investors alike, the market is increasingly less about seasonal flu alone and more about broader preparedness infrastructure, platform flexibility, and long-term resilience in global respiratory disease management.
Unlock 360° insights for strategic decision making and investment planning:
https://www.factmr.com/checkout/9389
Browse Full Report : https://www.factmr.com/connectus/sample?flag=B&rep_id=9389
To View Related Report:
Fluoroscopy Equipment Market https://www.factmr.com/report/267/fluoroscopy-equipment-market
Fluidized-Bed Bioreactors Market https://www.factmr.com/report/fluidized-bed-bioreactors-market
Fluorescent Tracers Market https://www.factmr.com/report/fluorescent-tracers-market
Fluid Management Market https://www.factmr.com/report/4763/fluid-management-market
- Contact Us -
11140 Rockville Pike, Suite 400, Rockville,
MD 20852, United States
Tel: +1 (628) 251-1583 | sales@factmr.com
ABOUT US
Fact.MR is a global market research and consulting firm, trusted by Fortune 500 companies and emerging businesses for reliable insights and strategic intelligence. With a presence across the U.S., UK, India, and Dubai, we deliver data-driven research and tailored consulting solutions across 30+ industries and 1,000+ markets. Backed by deep expertise and advanced analytics, Fact.MR helps organizations uncover opportunities, reduce risks, and make informed decisions for sustainable growth.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Flu Vaccine Market Size, Share & Forecast: Rising Seasonal Immunization Demand Drives Industry Expansion Across the U.S., Europe, and Asia-Pacific here
News-ID: 4508655 • Views: …
More Releases from Fact.MR

Rainwater Harvesting Systems Become Strategic Infrastructure as Water Stress Int …
The global rainwater harvesting system market is steadily moving from a niche sustainability category into a critical component of water infrastructure planning. As industries, cities, and agricultural producers face mounting pressure from water scarcity, climate variability, and rising utility costs, rainwater collection systems are increasingly being adopted to improve long-term water security and operational resilience.
Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=9667
What was once viewed primarily as…
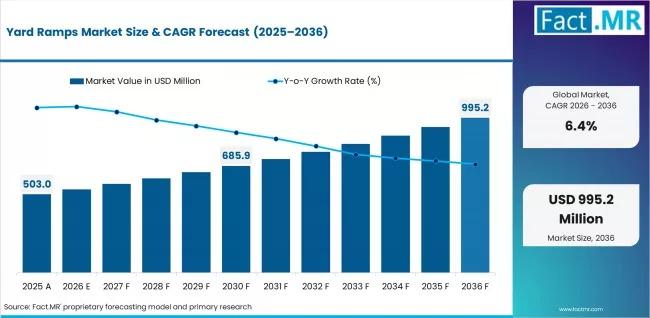
Yard Ramps Market Forecast to Grow at 6.4% CAGR, Creating USD 995 Million Opport …
In an era where supply chain agility is the ultimate competitive moat, the "permanent dock" is losing its status as a prerequisite for industrial operations. As e-commerce giants and third-party logistics (3PL) providers race to commission facilities in record time, a once-overlooked piece of equipment the yard ramp has emerged as a strategic linchpin. No longer viewed as a temporary workaround, these mobile loading solutions are becoming foundational to the…
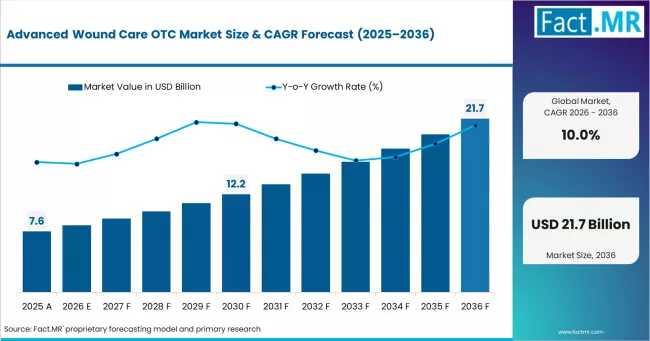
Advanced Wound Care OTC Market Expands Globally as India, China & U.S. Drive Dem …
According to Fact.MR estimates, the global advanced wound care otc Market was valued at USD 7.6 billion in 2025. The market is projected to reach USD 8.4 billion in 2026 and is expected to grow to USD 21.7 billion by 2036, expanding at a CAGR of 10.0%. Advanced Wound Dressings is anticipated to account for 48.0% of the product segment in 2026, while Chronic Wound Management is expected to remain…
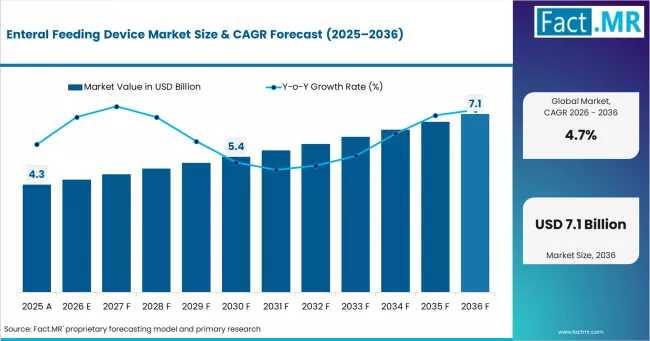
Global Enteral Feeding Device Market Size, Share & Forecast: Rising Demand for H …
According to Fact.MR estimates, the global enteral feeding device Market was valued at USD 4.3 billion in 2025. The market is projected to reach USD 4.5 billion in 2026 and is expected to grow to USD 7.1 billion by 2036, expanding at a CAGR of 4.7%. Feeding Pumps is anticipated to account for 40.0% of the product segment in 2026, while Adult is expected to remain the leading application with…
More Releases for Flu
Bird Flu Market Safeguarding Flocks, Securing Health: The Bird Flu Challenge in …
Bird Flu Market to Record an Exponential CAGR by 2031 - Exclusive Report by InsightAce Analytic Pvt. Ltd.
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Bird Flu Market - (By Virus Type (H5N1, H7N9, H5N6, H5N8, H10N8, H9N2, Others), By Test Type (PCR Tests, ELISA Tests, Hemagglutination Assays, Virus Isolation Tests, Next Generation Sequencing, Others), By Treatment Type (Vaccines, Antiviral Medication, Immunomodulators, Others),…
Microneedle Flu Vaccine Market New develop microneedle patch for flu vaccination
Drug delivery through the skin by transdermal patches has always been effective against viral infection and flu. Drug delivery through transdermal route is mainly effective due to passive diffusion of the drug and followed by drug development in transdermal delivery systems through different mechanism of action such as iontophorresis and sonophoresis. Moreover, pharmaceutical companies are focusing on development of micro-needle vaccines due to its immense utility in transdermal delivery of…
Flu Vaccine (Influenza Vaccine) Market: A look into the future of the “Univers …
Latest update on Flu Vaccine (Influenza Vaccine) Market Analysis Report published with an SMI, the industry growth analysis, and Projection by 2021-2028. This report is highly predictive as it holds the overall market analysis of the topmost companies in the Flu Vaccine (Influenza Vaccine) industry. With the classified Flu Vaccine (Influenza Vaccine) market research based on various growing regions, this report provides leading players' portfolios along with sales, growth, market…
Researchers Develop Microneedle Flu Vaccine for flu vaccination
New research funding to develop commercially available micro needle flu vaccine, which would pace up the launch of these products. In 2015, the Georgia Institute of Technology and Micron Biomedical were awarded US$ 2.5 million in grants from the Bill & Melinda Gates Foundation to advance the development of dissolvable microneedle patches for polio immunization. This would also widen up the disease profile for which microneedle vaccine could be developed.
Growing…
Canine Flu Therapeutics Market 2019| To Develop Therapeutics For The Flu To Cont …
"Canine Flu Therapeutics Market: Global Industry Analysis 2013-2017 and Opportunity Assessment 2018-2028" is the latest addition to MarketResearchReports.Biz industry research reports collection.
Canine flu (also known as dog flu) is a contagious respiratory disease in dogs mainly caused by Type A influenza/canine influenza virus. They are two various kinds of influenza viruses namely H3N8 virus and H3N2. Canine flu originated in horses, spread to dogs, and now spreads in between dogs…
Benefits of a Workplace Flu Shot Program
It's that time of year again — we're at the start of the annual epidemic of seasonal influenza, which (in North America) peaks between now and April. As a business owner or office manager, you may well know the tremendous disruption that influenza, commonly known as the flu, can cause when it spreads from one employee to the next across your organization. According to a recent review of the scientific…