Press release
Pharma Quality Assurance Services Market to Reach $697.5 Million by 2034 as Drug Recalls and GMP Compliance Drive Digital Transformation
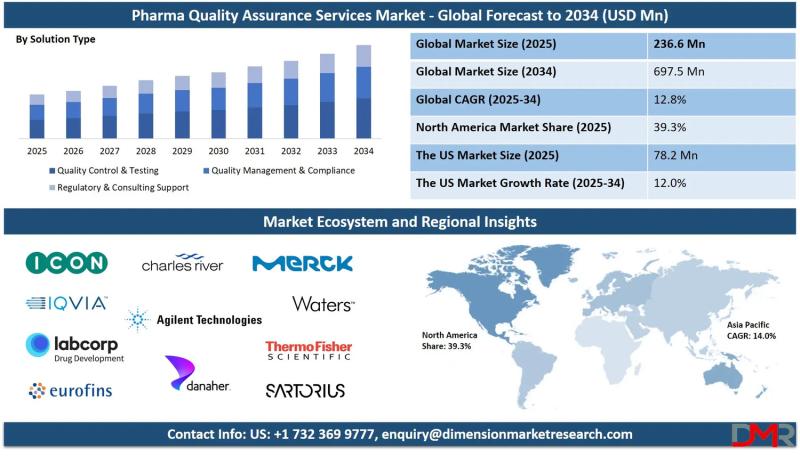
The global Pharma Quality Assurance Services Market is on a steady growth trajectory, with market valuation projected to rise from an estimated USD 236.6 million in 2025 to USD 697.5 million by 2034, registering a compound annual growth rate (CAGR) of 12.8%. According to Dimension Market Research, this expansion is being fundamentally driven by three converging forces: escalating drug recalls and regulatory scrutiny across major markets, the rapid expansion of pharmaceutical manufacturing in emerging economies, and the increasing complexity of biologics and personalized medicine requiring specialized QA protocols.Pharma quality assurance services-encompassing quality control testing, compliance management, regulatory consulting, and data integrity verification-have become indispensable as regulatory bodies worldwide intensify inspections and enforcement actions. According to Dimension Market Research, the U.S. market alone is projected to reach USD 78.2 million in 2025 and grow to USD 217.1 million by 2034 at a CAGR of 12.0% , driven by the FDA's 6,086 drug quality inspections conducted globally in 2023 and 113 warning letters issued for QA-related violations. With Japan's market growing at 13.0% (reaching USD 14.2 million in 2025) and Europe at 9.0% (USD 41.6 million), the sector is witnessing a global acceleration that positions QA as a strategic imperative rather than a regulatory burden.
📄 Get Your Sample Report Today → https://dimensionmarketresearch.com/request-sample/pharma-quality-assurance-services-market/
🔷 The News Angle: From Compliance Burden to Competitive Advantage-The Digital QA Transformation
The dominant narrative reshaping the pharma quality assurance services market is the transition from reactive compliance checklists to proactive, data-driven quality management systems powered by artificial intelligence, blockchain, and real-time analytics. This shift is not incremental-it represents a fundamental re-architecting of how pharmaceutical companies ensure product safety and integrity.
Escalating drug recalls are the most powerful catalyst. According to the report, the number of FDA drug recalls due to quality issues rose by nearly 20% between 2020 and 2023. The World Health Organization estimates that 1 in 10 medical products in low- and middle-income countries is substandard or falsified, emphasizing the need for robust QA systems globally. In 2023 alone, the FDA issued 113 warning letters to pharmaceutical firms for violations primarily related to QA issues. These incidents inflict severe reputational and financial damage, compelling both large and mid-sized manufacturers to make substantial investments in upgrading QA protocols, systems, and personnel.
Digital transformation is accelerating QA evolution. Advanced technologies-AI, machine learning, blockchain, and Internet of Things (IoT)-are increasingly being adopted to strengthen quality controls, compliance monitoring, and real-time analytics. AI is used to detect anomalies in data patterns, monitor deviations, and automate audit trails, reducing human error. Blockchain ensures data immutability and traceability across complex global supply chains, vital for authenticating drug quality from source to shelf. IoT-enabled sensors embedded in manufacturing and storage environments help maintain optimal temperature, humidity, and sterility conditions. This trend is supported by regulatory encouragement, including the U.S. FDA's "Pharmaceutical Quality for the 21st Century" initiative, which promotes risk-based and science-based QA practices using emerging technologies.
Global regulatory harmonization is another critical trend. Discrepancies in country-specific GMP (Good Manufacturing Practices), audit protocols, and documentation requirements have historically posed challenges for multinational pharmaceutical firms. The International Council for Harmonisation (ICH), the Pharmaceutical Inspection Co-operation Scheme (PIC/S), and mutual recognition agreements between agencies (EMA and FDA) are fostering greater collaboration. Standardized QA practices improve supply chain reliability, reduce non-compliance risks, and ensure consistent product quality regardless of origin.
🔷 Key Insights: Data Points Defining the Quality Assurance Revolution
North America Leads (39.3% Share in 2025): Advanced healthcare infrastructure, high R&D spending, early digital technology adoption, and stringent FDA/Health Canada standards drive regional dominance.
Quality Control & Testing Dominates Solution Type: Essential for batch release, stability testing, raw material inspection, and validation, with high adoption due to direct impact on product safety.
On-Premise Dominates Deployment Mode: Data security, compliance requirements (HIPAA, GxP, 21 CFR Part 11), and integration with legacy LIMS, ERP, and MES systems favor on-premise solutions.
Clinical Trials Lead Drug Development Stage: Pivotal role in validating safety and efficacy, generating massive data volumes requiring advanced analytics for patient recruitment, site selection, and adverse event monitoring.
Biopharmaceutical Production Leads Application: Complexity of biologics (monoclonal antibodies, gene therapies) demands advanced analytics for Process Analytical Technology (PAT), critical process parameters, and real-time monitoring.
Asia-Pacific Fastest-Growing Region: Rapid industrialization, expanding healthcare ecosystems, and increased investment in biotech research across China, India, South Korea, and Singapore drive highest CAGR.
FDA Oversight Scale: In 2023, the FDA conducted 6,086 drug quality inspections globally and issued 113 warning letters for QA-related violations.
Global Manufacturing Footprint: Over 75% of active pharmaceutical ingredients (APIs) used in U.S. drugs are manufactured outside the United States, highlighting global QA challenges.
Aging Population Demand: By 2030, more than 20% of the U.S. population and nearly 30% of Japan's population will be 65 or older, intensifying demand for quality-assured medications.
🔷 Market Dynamics: Drivers, Restraints, and Strategic Opportunities
Drivers: Drug Recalls & Emerging Market Expansion
The primary driver is the sharp increase in drug recalls due to contamination, mislabeling, and non-compliance with manufacturing standards. According to the FDA, thousands of pharmaceutical product recalls are issued annually, with many attributed to QA lapses. These incidents pose serious health risks and inflict severe reputational and financial damage. Regulatory bodies including the EMA, PMDA (Japan), CDSCO (India), and FDA have intensified inspections and enforcement actions, with increasing focus on data integrity, batch record reviews, and deviation management.
Simultaneously, the rapid growth of pharmaceutical manufacturing hubs in emerging economies-India, China, Brazil, and Southeast Asian nations-is driving increased demand for standardized QA systems. These countries have become vital players in global supply chains, particularly in API, generic drug, and vaccine component production. In response, manufacturers and governments are investing heavily in upgrading QA infrastructure to align with U.S. FDA, EMA, and WHO GMP guidelines. India's pharmaceutical sector has initiated nationwide QA reform programs, including mandatory training, digital documentation, and independent quality audits.
Restraints: High Costs & Fragmented Regulations
Despite momentum, significant barriers remain. The capital and operational expenses associated with implementing full-fledged QA systems act as a major restraint, especially for small and mid-sized enterprises (SMEs). Modern QA systems involve automated inspection tools, electronic batch recording software, LIMS, environmental monitoring devices, and skilled personnel. Upfront costs can range from hundreds of thousands to millions of dollars, with maintenance expenses including software updates, equipment calibration, regulatory audits, and staff retraining.
Additionally, navigating the global regulatory environment poses formidable challenges. Despite harmonization efforts, significant variations persist across national regulatory agencies in documentation requirements, inspection frequency, and GMP interpretations. The U.S. FDA emphasizes data integrity and real-time analytics, while the EMA may place greater weight on batch consistency and environmental controls. This fragmented landscape forces manufacturers to maintain multiple QA systems or adapt processes for each regulatory jurisdiction.
Opportunities: Personalized Medicine & Government Capacity Building
The surge in development of personalized medicine and biologics opens new growth avenues. Personalized therapies targeting specific genetic, biomarker, or molecular profiles demand highly precise, batch-specific manufacturing under tightly controlled QA conditions. Biologics-monoclonal antibodies, gene therapies, vaccines-are inherently more complex and sensitive than traditional small-molecule drugs, requiring stringent QA frameworks including real-time monitoring and predictive quality models.
Government-led capacity-building initiatives in low- and middle-income countries (LMICs) present another major opportunity. WHO's Global Benchmarking Tool (GBT) and the African Medicines Regulatory Harmonization (AMRH) initiative aim to strengthen regulatory systems and promote GMP-compliant manufacturing across Africa and parts of Asia. Donor-funded programs are supporting modernization of national drug control laboratories and adoption of digital QA platforms, enabling local manufacturers to qualify for WHO prequalification and export certifications.
📄 Get the Insights You Need to Drive Real Impact → https://dimensionmarketresearch.com/request-sample/pharma-quality-assurance-services-market/
🔷 Selective Segmentation: Where the Growth is Concentrated
By Solution Type (Quality Control & Testing-Dominant Share): Quality control and testing dominate due to their critical role in ensuring product safety, efficacy, and compliance. The pharmaceutical industry operates under rigorous regulatory frameworks mandating comprehensive testing at every stage of drug development and manufacturing. This segment sees high adoption due to its direct impact on batch release, stability testing, raw material inspection, and validation of production processes. Analytical tools including chromatography, spectroscopy, and molecular assays enable real-time monitoring, minimizing product recalls. The surge in outsourcing QC activities to specialized CROs and CDMOs underscores expanding demand for sophisticated quality testing solutions.
By Deployment Mode (On-Premise-Dominant Share): On-premise deployment dominates due to the industry's high sensitivity to data security, compliance requirements, and integration complexity with in-house systems. Pharmaceutical companies manage vast amounts of proprietary data-clinical trial outcomes, molecular research, regulatory filings-making data protection paramount. On-premise solutions provide greater control over data access, storage, and encryption, crucial for mitigating cybersecurity threats and ensuring regulatory adherence (HIPAA, GxP, 21 CFR Part 11). Integration with lab information management systems (LIMS), enterprise resource planning (ERP), and manufacturing execution systems (MES) is more seamless and secure when hosted internally. Cloud-based solutions are the fastest-growing segment, gaining traction for scalability and flexibility among smaller firms.
By Stage of Drug Development (Clinical Trials-Dominant Share): Clinical trials (Phases I, II, III) dominate due to their pivotal role in validating safety, efficacy, and therapeutic value. These phases are resource-intensive and data-rich, generating massive volumes of complex data requiring advanced analytics for processing, interpretation, and regulatory submission. Analytics play a crucial role in patient recruitment optimization, site selection, adverse event monitoring, and trial protocol compliance. Phase II and III trials demand real-time data monitoring, adaptive trial designs, and predictive modeling. With growing prevalence of personalized medicine and biologics, clinical trials are becoming increasingly complex, necessitating robust analytics for stratified patient population analysis and biomarker validation.
By Application (Biopharmaceutical Production-Dominant Share): Biopharmaceutical production dominates due to the complexity and high sensitivity involved in manufacturing biologics-monoclonal antibodies, recombinant proteins, cell and gene therapies. Unlike traditional small-molecule drugs, biopharmaceuticals are produced using living organisms, making manufacturing processes far more intricate, variable, and prone to inconsistencies. This necessitates advanced analytics to monitor, control, and optimize production parameters. Real-time process analytics enabled by Process Analytical Technology (PAT) is increasingly utilized to maintain product quality and comply with FDA cGMP guidelines. The growth in demand for biologics, driven by rising incidences of chronic diseases, cancer, and rare diseases, has led to increased investments in biomanufacturing infrastructure globally.
🔷 Regional Analysis: North America Leads, Asia-Pacific Emerges as Fastest-Growing
North America (39.3% Revenue Share in 2025): North America is projected to dominate the pharma quality assurance services market, primarily due to its advanced healthcare infrastructure, high R&D spending, and early adoption of digital technologies. The United States is home to a vast number of pharmaceutical giants, biotechnology firms, and contract research organizations (CROs) that heavily invest in data-driven drug development, clinical trials, and manufacturing optimization. Regulatory bodies including the FDA and Health Canada impose stringent quality and compliance standards, compelling companies to utilize sophisticated analytics. The presence of robust electronic health records (EHR) systems, integration with real-world data (RWD), and advancements in AI and machine learning further support market expansion. North American companies lead in precision medicine and personalized therapeutics, both demanding granular data analysis capabilities.
The U.S. Market (USD 78.2 million in 2025, 12.0% CAGR): The U.S. market is driven by a highly regulated environment, robust healthcare infrastructure, and a large pharmaceutical manufacturing base. The FDA enforces strict cGMP requirements, necessitating comprehensive QA systems across all production and supply chain stages. According to the FDA's inspection classification database, more than 6,000 inspections were conducted in 2023 alone, with quality-related compliance actions constituting a significant portion of enforcement letters. The U.S. Census Bureau projects that by 2030, more than 20% of the population will be 65 or older, increasing demand for medications and emphasis on product quality and safety. The Pharmaceutical Research and Manufacturers of America (PhRMA) notes that U.S.-based companies lead global drug innovation, heightening the importance of QA protocols that support clinical trial integrity and post-market surveillance.
Europe (USD 41.6 million in 2025, 9.0% CAGR): Europe's market benefits from a harmonized regulatory structure enforced by the European Medicines Agency (EMA) and local competent authorities under EU GMP guidelines, ensuring consistency in product quality, safety, and traceability. The European Directorate for the Quality of Medicines & Healthcare (EDQM) supports quality through the European Pharmacopoeia, which outlines critical specifications for drug substances and preparations. According to Eurostat, nearly 21% of the EU population was aged 65 or older in 2023, increasing demand for chronic disease medications, including complex biologics and generics. Brexit has led to a realignment of regulatory oversight, increasing QA complexity for companies operating in both the UK and EU, spurring demand for integrated quality solutions.
Asia-Pacific (Fastest-Growing Region-Japan at 13.0% CAGR): Asia-Pacific is projected to register the highest CAGR, driven by rapid industrialization, expanding healthcare ecosystems, and increased investment in biotech research. China, India, South Korea, and Singapore are emerging as global hubs for pharmaceutical manufacturing and clinical research outsourcing, thanks to cost efficiencies and a growing talent pool in data science and biostatistics. Governments across the region are investing in digital health infrastructure and policy frameworks to support innovation and regulatory harmonization. China's "Healthy China 2030" initiative and India's National Digital Health Mission (NDHM) have accelerated analytics adoption in drug development and patient care. Japan's market, growing at 13.0% CAGR, is deeply rooted in precision and regulation, with the PMDA overseeing strict GMP compliance and Japan's population among the oldest in the world (29.1% aged 65 or older in 2023), intensifying demand for high-quality medications.
📄 Get the Full Premium Report Now- https://dimensionmarketresearch.com/checkout/pharma-quality-assurance-services-market/
🔷 Competitive Landscape: Technology Providers, CROs, and Specialized QA Firms
The pharma quality assurance services market is moderately fragmented, with a mix of established technology providers, CROs, and pharmaceutical analytics specialists competing for market share.
Analytical Instrumentation and Lab Service Leaders: Thermo Fisher Scientific, Danaher Corporation, Sartorius AG, Merck KGaA, Waters Corporation, and Agilent Technologies provide the analytical tools-chromatography, spectroscopy, mass spectrometry-essential for quality control testing. These companies have built strong market positions through comprehensive instrument portfolios and regulatory expertise.
CRO and Contract Service Specialists: Charles River Laboratories, Eurofins Scientific, Labcorp Drug Development, Parexel International, ICON plc, PRA Health Sciences, Almac Group, IQVIA, Covance, Wuxi AppTec, and Medpace Holdings dominate the outsourced QA testing and compliance services market. IQVIA leverages its large data assets and advanced analytics to support evidence-based research and patient stratification. Wuxi AppTec provides integrated QA solutions for biologics and cell/gene therapies.
Compliance and Consulting Providers: TÜV SÜD, UL Solutions, and Pace Analytical Services offer GMP/GDP consulting, pharmaceutical quality audits, and regulatory compliance support, helping manufacturers navigate fragmented global regulatory environments.
Recent Developments Highlighting Market Momentum:
January 2025: Johnson & Johnson initiated a proposed acquisition of Intra-Cellular Therapies to strengthen its drug development pipeline with enhanced quality assurance oversight.
April 2025: The 5th Annual Pharma GMP & Quality Management Conference in Mumbai, India, addressed GMP modernization, digital quality systems, risk-based validation, and global harmonization.
May 2025: The GCC Quality Conference 2025 in Dubai focuses on enhancing pharmaceutical manufacturing quality through digital transformation and AI.
August 2024: The FDA Compounding Quality Center of Excellence Annual Conference focused on evolving best practices in compounding pharmacies and sterile product QA compliance.
🔷 The Road Ahead: What Decision-Makers Need to Know
For B2B decision-makers-pharmaceutical quality directors, manufacturing executives, regulatory affairs leaders, CRO partners, and investors-the strategic imperative is clear: pharma quality assurance has moved from a compliance necessity to a competitive differentiator. The 12.8% CAGR reflects sustained demand driven by regulatory intensity, manufacturing globalization, and biologic complexity.
Key strategic imperatives include:
Invest in digital QA platforms (AI, blockchain, IoT). Real-time analytics, predictive quality models, and immutable traceability are no longer optional for firms seeking to reduce recall risk and accelerate time-to-market.
Prioritize QC testing and biopharmaceutical production capabilities. These segments show the strongest demand and highest regulatory stakes, with biologics requiring specialized analytics.
Expand emerging market QA capacity strategically. India, China, Brazil, and Southeast Asia offer growth opportunities, but success requires alignment with WHO GMP and FDA/EMA standards for export qualification.
Address the QA talent shortage through training and automation. A shortage of trained QA professionals persists globally. AI-powered automation and digital documentation reduce reliance on manual processes.
Monitor global harmonization trends (ICH, PIC/S). Companies that adopt harmonized QA protocols across jurisdictions will reduce duplication costs and accelerate multi-market approvals.
The full report from Dimension Market Research provides granular segmentation by solution type (quality control & testing, quality management & compliance, regulatory & consulting support), deployment mode (on-premise, cloud-based, hybrid), stage of drug development (preclinical, clinical trials, manufacturing), application (biopharmaceutical production, sterile manufacturing, non-sterile manufacturing, API manufacturing, contract manufacturing quality audits, packaging & labeling compliance), and 20+ regional markets, offering actionable intelligence for strategic planning.
📄 Explore the Report with TOC → https://dimensionmarketresearch.com/report/pharma-quality-assurance-services-market/
For Sales or Inquiries, Contact
Robert John
957 Route 33, Suite 12 #308 Hamilton Square, NJ-08690 USA
Email: enquiry@dimensionmarketresearch.com
United States: (+1 732 369 9777)
Tel No: +91 88267 74855
Dimension Market Research (DMR) is a market research and consulting firm based in India & US, with its headquarters located in the USA. The company believes in providing the best and most valuable data to its customers using the best resources and analysts to work on, to create unmatchable insights into the industries and markets while offering in-depth results of over 30 industries, and all major regions across the world. We also believe that our clients don't always want what they see, so we provide customized reports as well, as per their specific requirements, to create the best possible outcomes for them and enhance their business through our data and insights in every possible way.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Pharma Quality Assurance Services Market to Reach $697.5 Million by 2034 as Drug Recalls and GMP Compliance Drive Digital Transformation here
News-ID: 4502445 • Views: …
More Releases from Dimension Market Research
Voice Recognition Security Market to Surge from 20.7 billion in 2025 to 199.2 Bi …
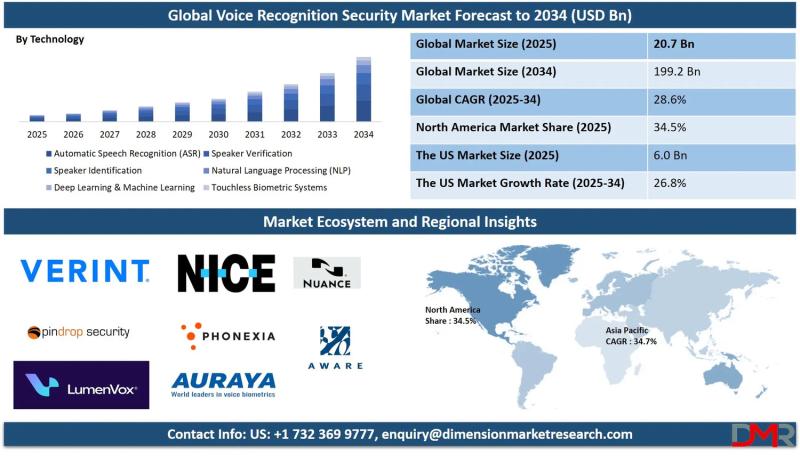
The global Voice Recognition Security Market is poised for explosive growth, with market valuation projected to surge from an estimated USD 20.7 billion in 2025 to USD 199.2 billion by 2034, registering a remarkable compound annual growth rate (CAGR) of 28.6%. According to Dimension Market Research, this extraordinary expansion is being driven by three converging forces: escalating cyber threats and identity fraud across BFSI and telecommunications, the post-pandemic surge in…
Smart Glasses for Augmented Reality Market to Reach $111.2 Billion by 2034 as En …
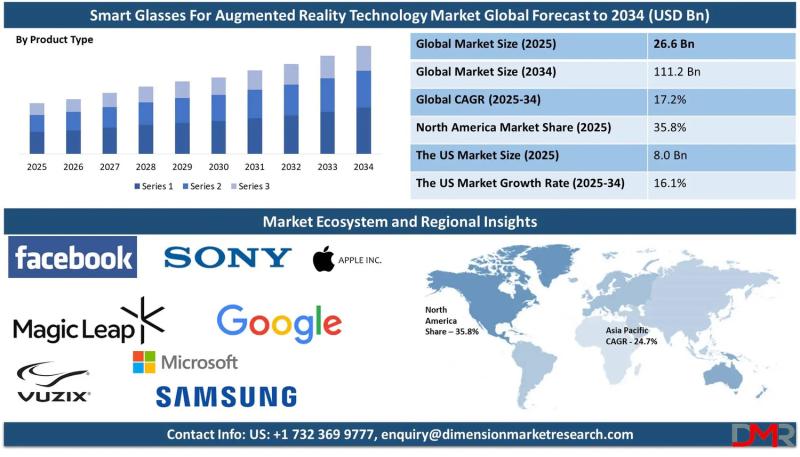
The global Smart Glasses for Augmented Reality (AR) Technology market is poised for remarkable growth, with market valuation projected to surge from an estimated USD 26.6 billion in 2025 to USD 111.2 billion by 2034, registering a strong compound annual growth rate (CAGR) of 17.2%. According to Dimension Market Research, this expansion is being driven by three transformative forces: rapid advancements in microdisplay and waveguide optics, surging enterprise adoption across…
Data Center Semiconductor Market to Surge from 252.2 billion in 2025 to 1.6 Tril …
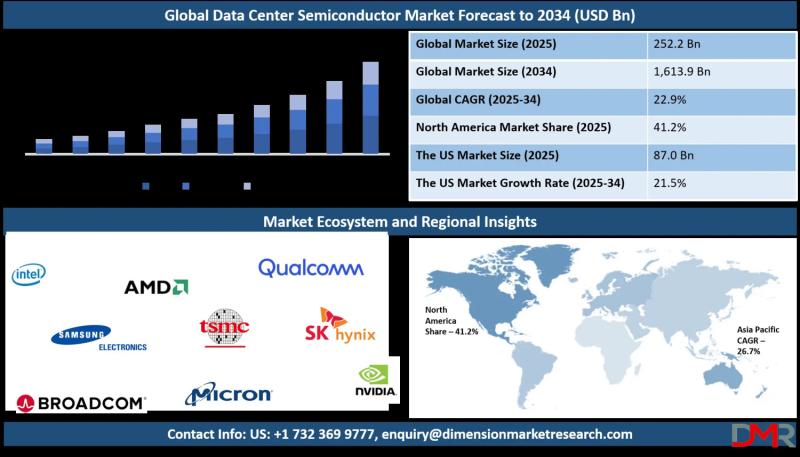
The global Data Center Semiconductor Market is poised for extraordinary growth, with market valuation projected to surge from an estimated USD 252.2 billion in 2025 to a staggering USD 1,613.9 billion by 2034, registering a powerful compound annual growth rate (CAGR) of 22.9%. According to Dimension Market Research, this unprecedented expansion is being driven by three transformative forces: the explosive growth of generative AI and large-scale inference workloads, the accelerating…

Automated Cooking Systems Market to Skyrocket from 5.0 Billion in 2025 to 34.1 B …
The global Automated Cooking Systems Market is poised for explosive growth, with market valuation projected to surge from an estimated USD 5.0 billion in 2025 to USD 34.1 billion by 2034, registering a remarkable compound annual growth rate (CAGR) of 23.8%. According to Dimension Market Research, this extraordinary expansion is being driven by three converging forces: acute labor shortages across the global foodservice industry, rapid advancements in AI-powered robotics and…
More Releases for FDA
FDA vs Non-FDA Freeze-Dried Fruit Suppliers: Risk and Market Access Differences
FDA Freeze-Dried Fruit Suppliers vs Non-FDA Approved Producers: Risk and Market Access Differences
Fujian Lixing Food Co., Ltd. - an internationally acclaimed provider of FDA freeze dried fruits and a pioneer in vacuum sublimation technology - today provided an insightful analysis that illuminated critical differences between FDA-registered suppliers and non-approved producers; with rising consumer demand for "clean label" healthy snacks a must, this study illustrates why regulatory compliance must be adhered…
DreaMed receives 5th FDA Clearance
TEL AVIV, Israel: DreaMed Diabetes LTD. ("DreaMed" or the "Company"), developer of the endo.digital Clinical Decision Support System announced today that it has received its 5th U.S Food and Drug Administration (FDA) clearance that expands the scope of AI enhanced treatment recommendations to patients on fixed meal insulin regimens. endo.digital is the first decision support system that has been cleared to assist healthcare providers in the management of diabetes…
FDA Compliant Blood Storage and Preservation
Accsense Monitoring System Automates Data Archive and Alarming
CAS DataLoggers provided the temperature alarming and monitoring system to a hospital blood bank looking to replace their old paper chart recorders as they became unreliable and spare parts were harder to find. For proper blood storage and preservation, the lab’s medical units needed to maintain storage temperatures between 2°C to 6°C (36°F to 43°F), given the perishability of blood components. The facility…
New FDA Design Control Training Courses
Salt Lake City, Utah - February 23 2017 - Procenius Consulting is a medical device consulting firm specializing solely in medical device design controls regulation (21 CFR 820.30).
Announcing New Design Control Training Courses
Procenius Consulting has just launched two new training courses covering basic and advanced topics of medical device design control regulation. These courses focus on compliance, practical implementation and industry best practices techniques for developing or improving a…
fda online training
GRC Training Solutions provides end-to-end FDA compliance solutions for those companies who want to maximize security, minimize operational costs, improve staff productivity and stay on top of all their compliance documentation.
GRC Training Solutions boasts a team of experts and specialists who have a proven track record in working with the biotechnology, medical device, diagnostic and pharmaceutical fields. Our team will work with you closely and develop solutions that meet…
FDA online training
Description:
Device firms, establishments or facilities that are involved in the production and distribution of medical devices intended for use in the U.S are required to register annually. Most establishments that are required to register with the FDA are also required to list the devices that are made there and the activities that are performed on those devices. Initially, FDA issued a 28-page Proposed Rule that would amend its regulations regarding…