Press release
mRNA Therapeutics Manufacturing Platform Market Insights (2026) | Top Companies, Analysis & Forecast
mRNA Therapeutics Manufacturing Platform refers to the end‐to‐end infrastructure, technologies, processes, and quality systems that support the development and large‐scale production of messenger RNA (mRNA)‐based medicines, including vaccines and therapeutics. Unlike traditional biologics, mRNA products use a streamlined synthetic process that enables rapid design and scalability, making them ideal for vaccines, cancer therapies, rare diseases, and personalized medicine applications.The platform integrates specialized capabilities such as in vitro transcription (IVT), purification, formulation (often with lipid nanoparticles), fill‐finish and stringent quality control compliant with current Good Manufacturing Practice (cGMP) standards.
Request Custom Research: https://www.datamintelligence.com/custom-research?kbmrna
United States: Recent Industry Developments
✅ February 2026: Moderna expanded its U.S. manufacturing facility for mRNA therapeutics, increasing capacity for next-generation vaccines.
✅ January 2026: BioNTech partnered with Thermo Fisher Scientific to enhance automated mRNA production platforms for clinical and commercial applications.
✅ December 2025: Lonza scaled up its GMP mRNA manufacturing services in the U.S., focusing on rapid production for emerging infectious disease programs.
Japan: Recent Industry Developments
✅ February 2026: Takeda Pharmaceutical advanced its mRNA therapeutics platform, focusing on oncology and rare disease applications.
✅ January 2026: Fujifilm Diosynth Biotechnologies expanded its mRNA contract development and manufacturing services (CDMO) capacity in Japan.
✅ December 2025: R&D initiatives at Japanese universities collaborated with biotech firms to improve mRNA production efficiency and stability.
1. Workflow Details
The scalable GMP process involves enzymatic upstream synthesis and purification, yielding 3-5 mg/mL mRNA suitable for lipid nanoparticle (LNP) encapsulation.
Upstream Enzymatic Steps
Plasmid Linearization: Supercoiled plasmid DNA (encoding target sequence, UTRs, poly(A)) is digested with NotI-HF enzyme in IVT buffer at 37°C, 225 rpm for 2 hours; targets greater 70% supercoiled ratio, verified by single band on gel.
In Vitro Transcription (IVT): Linear DNA (0.05 mg/mL) reacts with T7 polymerase, 5 mM each NTP (ATP/CTP/GTP + pseudouridine), 4 mM CleanCap in IVT buffer at 37°C for 2 hours; produces full-length mRNA.
DNase Digestion & Quench: Add DNase I buffer/enzyme for 2 hours to remove DNA template ( less than 90 pg/mg RNA), then EDTA to quench and prevent precipitation (15 min at 37°C).
Downstream Purification (80-95% Yield)
Dilution & UFDF1: 25x dilution with nuclease-free water, then tangential flow filtration (TFF/UFDF) concentrates to 0.61 mg/mL and diafilters to remove small impurities.
Core Bead Chromatography: Flow-through on multimodal resin (prepacked, sanitized) removes enzymes/DNA; greater than 94% step yield.
UFDF2: Concentrates to 1.07 mg/mL, diafilters into 1 mM citrate pH 6.4 formulation buffer.
Final Filtration/Fill: 0.2 μm sterile filter into bulk containers; single-use, RNase-free assemblies ensure scalability from 300 mL to GMP liters.
LNPs then encapsulate for delivery: lipids stabilize mRNA, target cells, enable greater 90% encapsulation efficiency (75 ± 25 nm particles).
Latest M & A
✅ BioNTech completed its acquisition of CureVac, finalising an all‐stock transaction ($1.25 billion) and integrating CureVac's mRNA design and manufacturing capabilities into its platform (exchange offer closed Dec 18 2025; compulsory acquisition of remaining shares expected Jan 2026).
✅ Novartis agreed to acquire Avidity Biosciences in a $12 billion all‐cash deal to strengthen its RNA therapeutics pipeline and delivery platform - announced Oct 26 2025 (expected closing in H1 2026).
• Quality Control & Release
Extensive analytical testing ensures identity, purity, potency and stability.
Tests include sequencing, capping efficiency, particle size for LNPs and sterility.
Overall, mRNA manufacturing combines automated synthesis, purification, and quality systems managed under cGMP.
3. Country‐Wise Research & Manufacturing Updates
• United States: Leads with 186 trials (40%), BARDA/NIH-funded oncology/infectious disease focus; Duke's P3 platform advances rapid antibody mRNA.
• Europe: 90 trials; BioNTech/Moderna hubs drive personalized cancer vaccines; EMA PRIME accelerates.
• Asia: 184 trials; China (72) scales manufacturing for chronic diseases; Japan/South Korea innovate LNPs; India expands CDMO via Aurigene.
• Others: GCC/Australia emerging in clinical expansions.
Key Developments
✅ February 2026: Biopharma companies expanded scalable, modular mRNA manufacturing facilities to meet rising global demand for personalized vaccines and therapeutics.
✅ January 2026: Integration of continuous manufacturing technologies and single-use bioreactors enhanced production efficiency, reduced contamination risk, and shortened batch turnaround times.
✅ December 2025: Adoption of advanced lipid nanoparticle (LNP) formulation systems increased, improving mRNA stability, delivery efficiency, and therapeutic efficacy in clinical trials.
✅ November 2025: Manufacturers strengthened regulatory compliance by implementing GMP-certified automated production lines and digital batch record systems for mRNA therapeutics.
✅ October 2025: Partnerships between contract development and manufacturing organizations (CDMOs) and biotech startups accelerated access to mRNA production expertise and specialized platforms.
✅ August 2025: Industry adoption of AI-driven process optimization and real-time analytics improved yield prediction, quality control, and scalability of mRNA therapeutics.
✅ June 2025: Growing demand for mRNA-based vaccines and therapies prompted investment in regional manufacturing hubs to ensure rapid response to emerging infectious diseases and personalized medicine needs.
Germany & 🇬🇧 United Kingdom
• Europe is expanding capacity under initiatives like the EU's €1.2 billion biotechnology funding and UK government support for mRNA facilities.
• Moderna opened an mRNA vaccine facility in Oxfordshire, UK, to supply seasonal vaccines.
• BioNTech in Germany is advancing cancer vaccines and infectious disease mRNA therapies.
Japan
Strategic regulatory pathways and funding promote local mRNA R&D and manufacturing partnerships with industry players.
India
Government programs like Mission COVID Suraksha and regulatory reforms support mRNA manufacturing scale‐up in India.
Latin America & Africa
• Countries like Brazil and Mexico are developing CDMO hubs for mRNA production.
• African initiatives (e.g., Afrigen) focus on local mRNA vaccine production and technology transfer.
• Dei BioPharma in Uganda is building capacity for mRNA therapies and vaccines.
4. Top Companies & Key Developments
Some leading players and recent developments include:
• Moderna - Expanded oncology pipeline with automated LNP production for cancer and rare disease therapies.
• BioNTech - Advancing Phase III individualized vaccines; strengthening IP in oncology and infectious disease.
• Pfizer, Sanofi, GSK - Partnerships and strategic agreements to strengthen mRNA portfolios.
• CDMOs like Lonza, TriLink BioTechnologies, Samsung Biologics, and Catalent support contract manufacturing and analytical services.
• CureVac - Deployed RNA Printer® for GMP-grade automated production, enhancing vaccine scalability.
• ST Pharm - Asia's largest mRNA CDMO, producing 100 g+ batches for therapeutic applications.
• Aldevron - Upgraded plasmid/mRNA platforms supporting vaccines and gene therapy development.
5. Regulatory Affairs
Manufacturing mRNA therapeutics must align with stringent regulatory frameworks:
• Current Good Manufacturing Practice (cGMP)
Facilities must meet cGMP standards covering documentation, personnel, equipment qualification, and quality systems.
• Regulatory Approvals
Agencies such as U.S. FDA, European Medicines Agency (EMA), Japan PMDA, and others require detailed validation of the entire process - from raw materials to final product release.
Emergency use authorizations (EUAs) and full approvals have been granted for mRNA vaccines, setting precedents for future therapeutics.
• Quality & Safety
Demonstrating safety, potency, purity and stability is critical.
Regulators look closely at impurities, lipid nanoparticle formulation and cold‐chain stability - key aspects influencing both efficacy and safety.
Given the platform nature of mRNA, regulators increasingly encourage advanced analytical technologies and scalable processes that maintain product quality.
6. Requirements to Start an mRNA Manufacturing Platform
Setting up an mRNA manufacturing facility involves several major prerequisites:
• Technical & Scientific Expertise
Expertise in molecular biology, IVT processes, purification technologies, LNP formulation, and analytical development.
• Infrastructure & Equipment
Facilities with controlled cleanrooms, bioreactors for IVT, chromatography systems, sterile fill‐finish lines, cold storage and high‐end analytical instruments.
Automated systems for process control and monitoring improve quality and reproducibility.
• Supply Chain & Raw Materials
Secure supply of DNA templates, nucleotides, enzymes, lipids, plasmid production resources and consumables.
Establishing partnerships for critical inputs mitigates supply risk.
• Compliance & Quality Systems
Establish a comprehensive Quality Management System (QMS) to ensure adherence to regulatory standards (cGMP, SOPs, validation records).
• Regulatory Licensing
Obtain necessary manufacturing licenses from national regulators and approvals for product clinical trials or commercialization.
• Skilled Workforce
Hiring trained personnel in bioprocess engineering, quality assurance, regulatory affairs and research capabilities is essential.
• Funding & Partnerships
Significant capital investment is required upfront; collaborations with government, biotech firms or CDMOs can accelerate development.
Setting up such a platform can be capital‐intensive but offers strategic advantages in rapid development, scale‐up potential and future therapeutic pipelines - especially for pandemic preparedness and precision medicine.
High barriers: $50-100M initial for GMP facility; 12-18 months setup.
Facilities: ISO 6-8 cleanrooms (2,000+ sq ft, modular); -65°C storage/shipping; RNase-free single-use (RoSS.FILL/RoSS.SHIP).
Equipment: TFF (Pall/Sartorius), core bead columns, UPLC/CGE analyzers; automated filling ($5-10M).
Materials: GMP nucleotides/CleanCap (TriLink), plasmids (Aldevron); qualify for endotoxin/nuclease.
Regulatory/Team: IND submission, QA/QC experts, biosafety level 1-2; risk-assess vendors.
Challenges: Supply chain (pseudouridine shortages), scaling (300 mL to 100L), cold chain.
Conclusion
The mRNA Therapeutics Manufacturing Platform represents a transformative advance in biopharmaceutical production, enabling rapid, flexible and scalable manufacturing of next‐generation therapies and vaccines. With countries investing heavily in infrastructure and regulatory bodies adapting frameworks for these novel modalities, the industry is poised for growth across therapeutic areas from infectious disease prevention to oncology and rare diseases. Strategic partnerships, skilled talent, strong regulatory compliance and robust capital support will remain essential for organizations looking to enter and succeed in this fast‐evolving space.
Contact Us -
Company Name: DataM Intelligence
Contact Person: Sai Kiran
Email: Sai.k@datamintelligence.com
Phone: +1 877 441 4866
Website: https://www.datamintelligence.com
About Us -
DataM Intelligence is a Market Research and Consulting firm that provides end-to-end business solutions to organizations from Research to Consulting. We, at DataM Intelligence, leverage our top trademark trends, insights and developments to emancipate swift and astute solutions to clients like you. We encompass a multitude of syndicate reports and customized reports with a robust methodology.
Our research database features countless statistics and in-depth analyses across a wide range of 6300+ reports in 40+ domains creating business solutions for more than 200+ companies across 50+ countries; catering to the key business research needs that influence the growth trajectory of our vast clientele.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release mRNA Therapeutics Manufacturing Platform Market Insights (2026) | Top Companies, Analysis & Forecast here
News-ID: 4403261 • Views: …
More Releases from DataM Intelligence 4 Market Research LLP
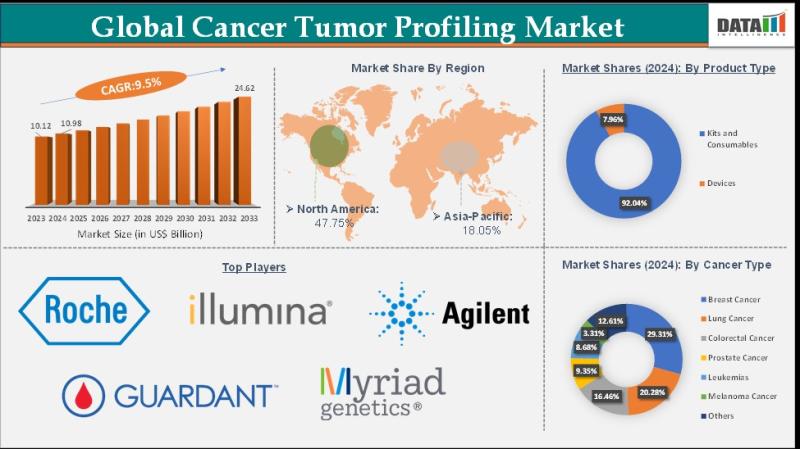
Cancer Tumor Profiling Market to Reach US$ 24.62 Billion by 2033 at 9.5% CAGR; N …
The Cancer Tumor Profiling Market reached US$ 10.98 billion in 2024, increasing from US$ 10.12 billion in 2023, and is projected to reach US$ 24.62 billion by 2033, growing at a CAGR of 9.5% during the forecast period 2025 to 2033.
Market growth is driven by the increasing adoption of precision oncology, rising prevalence of various cancers, and advancements in genomic sequencing and molecular diagnostics technologies. Tumor profiling enables comprehensive analysis…

Future of Pumped Hydro Storage Market Expansion (2026): Global Growth, Greener F …
Market Size and Growth
pumped hydro storage market reached US$ 332.12 billion in 2024 and is expected to reach US$ 666.38 billion by 2032, growing with a CAGR of 9.20% during the forecast period 2025-2032.
Request Executive Sample Report (Get Higher Priority for Corporate Email ID):- https://datamintelligence.com/download-sample/pumped-hydro-storage-market?kb
United States: Recent Industry Developments
✅ February 2026: AES Energy Storage announced a 500 MW pumped hydro project in California to support renewable grid integration.
✅ January…

Precision Nutrition Market Set for Explosive Growth to USD 22.82 Billion by 2032 …
Leander Texas -
Precision Nutrition Market size reached US$ 6.78 billion in 2024 and is expected to reach US$ 22.82 billion by 2032, growing with a CAGR of 16.38% during the forecast period 2025-2032.
The Precision Nutrition market's strong growth in the US and Japan is driven by rising demand for personalized dietary solutions, increased integration of AI and biomarker technologies into health platforms, collaborations between healthcare providers and tech firms,…

United States 3D Printed Electronics Market (2026): Flexible Circuits, Smart Dev …
Market Size and Growth
3D Printed Electronics Market reached US$ 9.35 billion in 2023 and is expected to reach US$ 28.07 billion by 2031, growing with a CAGR of 14.73% during the forecast period 2024-2031.
Request Executive Sample Report (Get Higher Priority for Corporate Email ID):- https://datamintelligence.com/download-sample/3d-printed-electronics-market?kb
United States: Recent Industry Developments
✅ February 2026: Nano Dimension unveiled a next-gen 3D printed electronics platform for rapid prototyping of PCBs and antennas.
✅ January 2026: Lockheed…
More Releases for RNA
CD Formulation Launches Custom Circular RNA Synthesis Service to Accelerate RNA …
CD Formulation introduces a customizable circRNA synthesis service, delivering high-quality, stable circRNAs for therapeutics, vaccines, and gene research, supported by advanced design and QC processes.
CD Formulation, a leading provider of advanced small nucleic acid synthesis [https://www.formulationbio.com/nucleic-acid/custom-small-nucleic-acid-synthesis.html] solutions, is proud to announce the launch of its fully customizable circular RNA (circRNA) synthesis service. This new service addresses the growing need for stable, non-immunogenic RNA molecules for therapeutic development, vaccine research, and…
Self-Amplifying RNA Synthesis Market Gains Traction as Biotech Firms Embrace Sca …
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the " Self-Amplifying RNA Synthesis Market- (By Product & Service (Products (Enzymes & Reagents, Premade saRNA, Others), Custom Synthesis Services), By Application (Therapeutics Development (Oncology, Infectious Diseases, Others), Biomedical Research), By End-User (Pharmaceutical & Biotechnology Companies, Academic & Research Institutes, Others)), Trends, Industry Competition Analysis, Revenue and Forecast To 2034."
According to the latest research by InsightAce Analytic,…
RNA Extraction and RNA Purification Market: Growth, Trends & Competitive Landsca …
The global RNA Extraction and RNA Purification Market is expected to grow at 6.3% CAGR from 2025 to 2032.
This Market Report is the result of extensive research and analysis conducted by our team of experienced market researchers through -
• 70% efforts of Primary Research
• 15% efforts of Secondary Research
• 15% efforts from the subscription to Paid database providing industry overview, macro and micro economics factors, and financials of private limited…
RNA Targeting Small Molecules Therapeutics Market: Exponential Growth with Risin …
Estimations Predict a CAGR of 29.8% by 2029 in Global RNA Targeting Small Molecules Therapeutics Market Boosted by Precision Medicine, RNA Biomarker Identification and RNA Genetic Manipulation
What Is The Projected Market Size of The Global RNA Targeting Small Molecules Therapeutics Market And Its Growth Rate?
• The market will grow from $6.1 billion in 2024 to $7.87 billion in 2025 at a compound annual growth rate (CAGR) of 28.9%.
• Expected exponential…
Global DNARNA Extraction Kit Market by Type (Cell-free DNA (cfDNA), Sequence-spe …
"DNARNA Extraction Kit Market" is segmented by Company, Region (country), By Type, Application, stakeholders and other participants. This report provides an analysis of revenue and forecast across Type and Application segments for 2023-2032.
The market for DNARNA Extraction Kits has been thoroughly researched via primary and secondary sources to produce this research study. Along with a competitive analysis of the market, segmented by application, type, and geographical trends, it offers a…
Cancer RNA Expression Market to Reap Excessive Revenues by 2028(By sequencing te …
Worldwide cancer is one of the leading cause of death and effective way of treating it still looks unaccomplished in most parts of the world. The factors which influence the successful treatment of cancer are different depending on the stage of diagnosis, treatment availability and availability of trained healthcare professionals coupled with high economic burden of the disease. The gene expression of cancerous cells varies by cancer type and may…
