Press release
Pharmacovigilance Market Size to Develop Lucratively by 2020
Global Pharmacovigilance Market: SnapshotThe global pharmacovigilance market is growing cumulatively as it is being driven by increase in awareness in the public health sector coupled with rise in preference for safe medicines. Globally, growing number of national pharmacovigilance centers is playing an imperative role to support the growth of the market in the next few years. Needless to mention, pharmacovigilance is witnessing numerous challenges to develop a better healthcare system. Unavailability of unskilled labor and information about drugs obtained via the web could be two high-impact restraints of the market. However, rigorous pharmacovigilance activities that could be made compulsory are expected to raise demand in the market. Such mandatory activities help to actively manage high-risk medicines.
The global pharmacovigilance market is anticipated to thrive at a handsome CAGR to be worth a US$6.1 billion by the end of 2020.
Obtain Report Details @ https://www.transparencymarketresearch.com/pharmacovigilance-market.html
Monitoring of Drug Safety Leads to Massive Growth in Phase III Clinical Trial
The world pharmacovigilance market could be segmented into preclinical studies, clinical trial phases I, II, III, and IV by phase of drug development. Phase IV a.k.a. post-marketing surveillance has contributed with its larger share in terms of revenue in the previous years. Safety worries relating to marketed products, growing necessity of designing systems to compare safety profiles of homologous medicinal products, and growth in public health awareness campaigns are few of the reasons which aid the growth of the segment.
Clinical trial phase III is predicted to set to rise at a higher CAGR because of escalating need for monitoring of drug safety. This phase emphasizes on drug safety and efficiency in diverse sub-groups, where the benefit-risk ratio is developed, scrutinized, and updated accordingly. In order to be more accurate on the part of drug safety, biopharmaceutical companies are focusing on creating advanced clinical trial phases.
The world pharmacovigilance market could be divided into contract outsourcing and in-house pharmacovigilance based on type of service provider. An important factor helping with the growth of contract outsourcing could be shift in focus to core business activities from that of non-core by outsourcing pharmacovigilance services. This will lead to decrease in operational and financial costs suffered from delays in product approval.
Request For Sample of Report @ https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=1729
The world pharmacovigilance market could be segregated based on five types of methods. These are intensified ADR reporting, targeted spontaneous reporting, EHR mining, spontaneous reporting, and cohort event monitoring. In a recent year, in terms of revenue, spontaneous reporting method grew as a larger segment since it is a more accurate technique of discovering new ADRs.
Strict Healthcare Regulations Result in Higher Demand in Asia Pacific
Asia Pacific is expected to gain higher growth during the forecast period. Factors supporting market growth in the region are envisaged to include high demand for strict healthcare regulations and increase in number of clinical trials being set up. Nevertheless, developed regions are projected to significantly contribute to the international pharmacovigilance market. For instance, North America’s cost benefit gained by switching to contract research organization (CRO) outsourcing from expensive in-house activities is projected to help the region to collect a larger share of the market.
Increased death rates because of adverse drug reactions (ADRs) and rise in patient fears regarding safety of medicinal products have propelled market growth of North America in the recent years. A report by Centers for Disease Control and Prevention (CDC) states that there are around 100,000 deaths caused by ADRs in the U.S. which are counted among the top ten leading causes of death in the country.
Request For Report Brochure @ https://www.transparencymarketresearch.com/sample/sample.php?flag=B&rep_id=1729
The report also provides a regional study of other important markets, viz. Europe and the Middle East and Africa. Besides this, it analyzes the competitive landscape while profiling key players such as Accenture plc, Cognizant Technology, Bristol- Myers Squibb, and Wipro Limited.
About Us
Transparency Market Research (TMR) is a global market intelligence company providing business information reports and services. The company’s exclusive blend of quantitative forecasting and trend analysis provides forward-looking insight for thousands of decision makers. TMR’s experienced team of analysts, researchers, and consultants use proprietary data sources and various tools and techniques to gather and analyze information. Our business offerings represent the latest and the most reliable information indispensable for businesses to sustain a competitive edge.
Contact Us
Transparency Market Research
State Tower,
90 State Street, Suite 700
Albany, NY 12207
United States
Tel: +1-518-618-1030
USA - Canada Toll Free: 866-552-3453
Email: sales@transparencymarketresearch.com
Website: http://www.transparencymarketresearch.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Pharmacovigilance Market Size to Develop Lucratively by 2020 here
News-ID: 1182673 • Views: …
More Releases from Transparency Market Research

North America Commercial Fuel Cards Market Growth Accelerates Toward USD 312.5 B …
The North America commercial fuel cards market is expanding steadily as businesses seek greater control over fuel expenses, fleet efficiency, and operational transparency. Commercial fuel cards are payment tools designed specifically for businesses that operate vehicle fleets, enabling them to monitor fuel usage, manage expenses, reduce fraud, and simplify accounting processes. These cards are widely used across transportation, logistics, construction, utilities, and service industries.
The North America Commercial Fuel Cards Market…
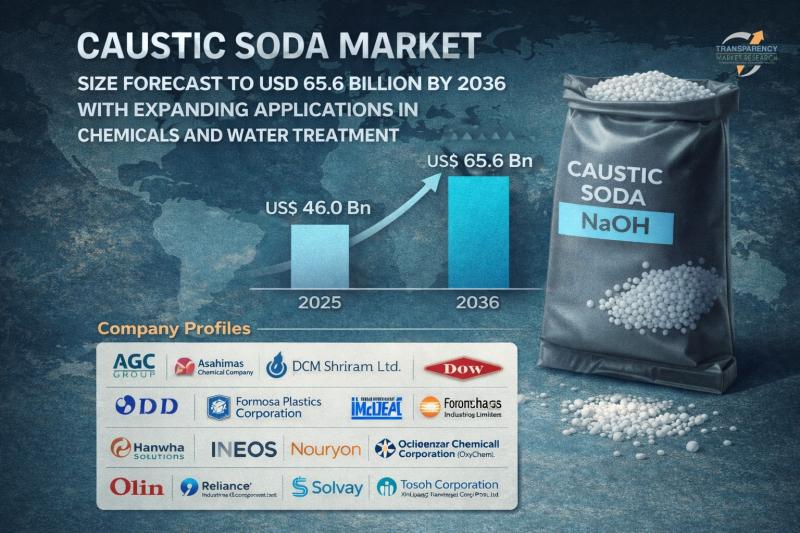
Caustic Soda Market Size Forecast to USD 65.6 Billion by 2036 with Expanding App …
Caustic Soda Market Outlook 2036
The global caustic soda market was valued at US$ 46.0 Billion in 2025 and is projected to reach US$ 65.6 Billion by 2036, expanding at a steady CAGR of 3.2% from 2026 to 2036. Market growth is driven by increasing demand from the pulp & paper industry, rising alumina production, expanding chemical manufacturing activities, and growing applications in water treatment and textiles.
👉 Get your sample market…

Global Baby Diaper Market Outlook 2036: Industry to Reach US$ 75.1 Billion by 20 …
The global baby diaper market was valued at US$ 44.5 Bn in 2025 and is projected to reach US$ 75.1 Bn by 2036, expanding at a steady CAGR of 4.9% from 2026 to 2036. This consistent upward trajectory reflects the essential nature of diapers in infant hygiene and the growing consumer preference for high-performance and convenient baby care products.
In 2025, North America accounted for 42.1% of the global revenue share,…

Rare Earth Metals Market to be Worth USD 30.9 Bn by 2036 - By Metal Type / By Ap …
The rare earth metals market has evolved from a niche industrial segment into a strategically critical global industry. In 2025, the market stood at US$ 14.1 Billion, driven primarily by increasing deployment of electric vehicles (EVs), renewable energy systems, defense electronics, and advanced industrial machinery.
Review critical insights and findings from our Report in this sample -
https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=823
By 2036, the market is expected to nearly double to US$ 30.9 Billion, supported by…
More Releases for Pharmacovigilance
Top Pharmacovigilance Companies Analysis By 2031
The Pharmacovigilance Market is expected to register a CAGR of 6.6% from 2025 to 2031, with a market size expanding from US$ XX million in 2024 to US$ XX Million by 2031.
Download PDF Copy @ https://www.theinsightpartners.com/sample/TIPRE00003127?utm_source=OpenPR&utm_medium=10379
The List of Companies
• #Accentures
• Bristol-Myers Squibb Company
• Linical Accelovance
• Cognizant
• Covance Inc.
• F. Hoffmann-La Roche Ltd.
• GlaxoSmithKline plc.
• ICON plc
• Capgemini (IGATE Corporation)
Clinical…
Pharmacovigilance - Scope and Research Methodology
The Pharmacovigilance Market is expected to register a CAGR of 6.6% from 2025 to 2031, with a market size expanding from US$ XX million in 2024 to US$ XX Million by 2031.
The Pharmacovigilance Market report covers analysis by Clinical Trial Phase (Pre-Clinical, Phase I, Phase II, Phase III, and Phase IV), Service Provider (In-House and Contract Outsourcing), Type of Method (Spontaneous Reporting, Intensified ADR Reporting, Targeted Spontaneous Reporting, Cohort Event…
Pharmacovigilance World 2025 Conference & Expo
We are delighted to welcome you to the Pharmacovigilance World 2025, and we are confident that your active participation will contribute to the advancement of drug safety practices. Together, let us strive towards a safer and more vigilant healthcare system that prioritizes patient well-being and ensures the continued benefit of medications worldwide.
As medical science advances, so does our understanding of drug safety and the need for vigilance when it comes…
Top Factor Driving Pharmacovigilance Market Growth in 2025: Research And Develop …
How Are the key drivers contributing to the expansion of the pharmacovigilance market?
The escalation in research and development undertakings stimulates growth in the pharmacovigilance market. Pharmaceutical organizations can create novel and superior drugs through enhanced safety profiles by allocating resources to R&D. The intensive testing in preclinical and clinical stages during the drug development protocol allows early recognition of potential safety issues, paving the way for adequate risk reduction approaches.…
Monitoring Medication Safety with Pharmacovigilance
Pharmacovigilance (PV) is defined as the science and activities relating to the detection, assessment, understanding and prevention of adverse effects or any other drug-related problem. Pharmacovigilance plays a significant role in pharmaceutical and biotechnological sectors in designing of drugs and their interactions. The pharmacovigilance involves collecting information from healthcare providers and patients to know about the hazards associated with medications.
Download Sample PDF at: https://www.theinsightpartners.com/sample/TIPRE00003127?utm_source=OpnePR&utm_medium=10776
Increasing cases of adverse drug reactions…
Pharmacovigilance Market Opportunity Analysis by 2028
Pharmacovigilance Market: Introduction
According to the report, the global pharmacovigilance market was valued at US$ 6.1 Bn in 2020 and is projected to expand at a CAGR of 8.8% from 2021 to 2028. Pharmacovigilance activities are defined as science used for detection, assessment, understanding, and prevention of adverse effects of drugs and vaccines. Drugs and vaccines go through rigorous testing in the clinical trials to check their safety and efficacy before…
