Press release
Global Orphan Drugs Market Shares Include: Europe, North America, Asia Pacific and South America
The Global Orphan Drugs Market accounted to USD 113.8 billion in 2016 growing at a CAGR of 7.23% during the forecast period of 2017 to 2024. The upcoming market report contains data for historic years 2015, the base year of calculation is 2016 and the forecast period is 2017 to 2024.The rarest diseases are known as orphan diseases. To treat these types of diseases drugs available are termed as Orphan drugs. These kinds of diseases are usually different and its occurrence rate is very less. Orphan drugs are impacts on small group of the population, as it is mostly genetic in nature.
Major Market Drivers and Restraints:
• Rising prevalence of rare diseases
• Rise in R&D investments
• Expansion of product pipeline by launching new drugs
• Impact of developments in pharmacogenomics
• Government initiatives for the development
• High prices are associated with these drug development
Click & Get Free Sample Report of Global Orphan Drugs Market Visit @ http://databridgemarketresearch.com/request-a-sample/?dbmr=global-orphan-drugs-market
Competitive Analysis:
The orphan drugs market is highly fragmented and is based on new product launches and clinical results of products. Hence the major players have used various strategies such as new product launches, clinical trials, market initiatives, high expense on research and development, agreements, joint ventures, partnerships, acquisitions, and others to increase their footprints in this market. The report includes market shares of orphan drugs market for global, Europe, North America, Asia Pacific and South America.
Market Segmentation:
On The Basis Of Product Based: biologics orphan drugs and non-biologics orphan drugs. Biologics orphan drugs is sub segmented into monoclonal antibodies, growth hormones, interferons or interleukins and plasma products.
Have any Query Regarding this Report? Contact us Visit @: http://databridgemarketresearch.com/speak-to-analyst/?dbmr=global-orphan-drugs-market
On The Basis Of Disease Type: oncologic, metabolic, hematologic & immunologic, infectious and neurologic.
On The Basis Of Indication: non-Hodgkin lymphoma, acute myeloid leukemia, cystic fibrosis, glioma, pancreatic cancer, ovarian cancer, multiple myeloma, Duchenne muscular dystrophy, graft vs host disease and renal cell carcinoma.
On The Basis Of Geography: Orphan Drugs Market Report covers data points for 28 countries across multiple geographies such as North America & South America, Europe, Asia-Pacific, and Middle East & Africa. Some of the major countries covered in this report are U.S., Canada, Germany, France, U.K., Netherlands, Switzerland, Turkey, Russia, China, India, South Korea, Japan, Australia, Singapore, Saudi Arabia, South Africa, and Brazil among others. In 2017, North America is expected to dominate the market.
Major Market Competitors:
Some of the major players operating in orphan drugs market are F. Hoffmann-La Roche Ltd., Novartis AG, Pfizer Inc., AbbVie, AstraZeneca, Baxter, Eli Lilly and Company, Johnson & Johnson Services Inc., Merck KGaA, Novo Nordisk A/S, Sanofi, Bristol-Myers Squibb Company, CELGENE CORPORATION, Alexion, Alnylam Pharmaceuticals, Inc., Amgen Inc., Biogen, Celldex Therapeutics, GlaxoSmithKline plc., Eisai Co., Ltd., Intercept Pharmaceuticals, Inc., Northwest Biotherapeutics, Inc., NewLink Genetics Corporation, Shire, Takeda Pharmaceutical Company Limited, Vertex Pharmaceuticals Incorporated and XOMA among others.
For more information about this report visit @ http://databridgemarketresearch.com/reports/global-orphan-drugs-market/
Related Report
Global Ophthalmic Suspension Market – Industry Trends and Forecast to 2024
Global Ophthalmic Suspension Market by Product (Antibiotic, Antifungal, Antibacterial, Steroids, Non-Steroidal Anti-Inflammatory Drugs), By Treatment (Bacterial Infections, Retinal Disorders, Glaucoma, Allergies, Diabetic Eye Disease), By End User (Hospitals, Eye Clinics, Clinics), By Geography – Industry Trends and Forecast to 2024
Read More: http://databridgemarketresearch.com/reports/global-ophthalmic-suspension-market/
About Data Bridge Market Research:
Data Bridge Market Research set forth itself as an unconventional and neoteric Market research and consulting firm with unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market. Data Bridge endeavors to provide appropriate solutions to the complex business challenges and initiates an effortless decision-making process.
Contact:
Vishal Dixit
Data Bridge Market Research
Tel: +1-888-387-2818
Email: Sales@databridgemarketresearch.com
Investor: investors@databridgemarketresearch.com
Visit Blog http://databridgemarketresearch.com/blog/
Follow us on LinkedIn: https://www.linkedin.com/company/data-bridge-market-research
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Orphan Drugs Market Shares Include: Europe, North America, Asia Pacific and South America here
News-ID: 800090 • Views: …
More Releases from Data Bridge Market Research
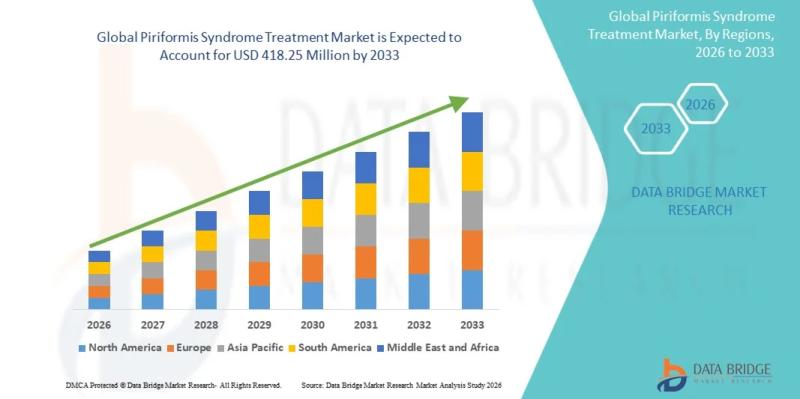
Piriformis Syndrome Treatment Market Size to Reach USD 418.25 million by 2033, G …
As per Data Bridge Market Research analysis, the Piriformis Syndrome Treatment Market was estimated at USD 0.26 billion in 2025. The market is expected to grow from USD 262.42 million in 2025 to USD 418.25 million by 2033, at a CAGR of 6.00% during the forecast period with driven by the rising demand for minimally invasive pain management therapies, increasing prevalence of musculoskeletal disorders, advancements in diagnostic imaging, and growing…
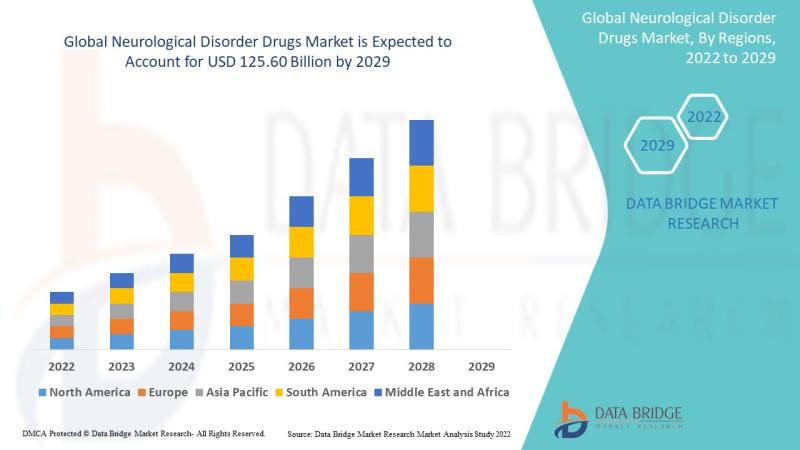
Neurological Disorder Drugs Market Outlook (2026-2032): 5.9% CAGR Driving Growth …
"As per Data Bridge Market Research analysis, the Neurological Disorder Drugs Market was estimated at USD 99.86 billion in 2025. The market is expected to grow from USD 105.76 billion in 2026 to USD 149.17 billion in 2032, at a CAGR of 5.9% during the forecast period with driven by the rising demand for advanced neurotherapeutics, increasing prevalence of neurological diseases, and expanding adoption of targeted biologics and precision medicine…
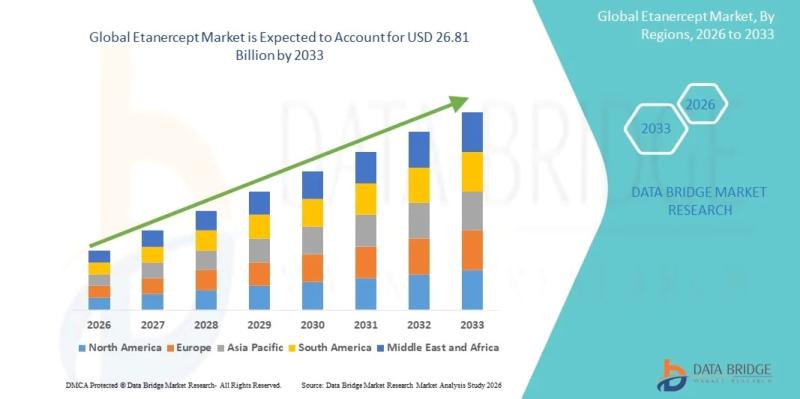
Etanercept Market Size, Share & Industry Trends Analysis Report, 2026-2033: Mark …
As per Data Bridge Market Research analysis, the Etanercept Market was estimated at USD 20.05 billion in 2025. The market is expected to grow from USD 20.79 billion in 2026 to USD 26.81 billion in 2033, at a CAGR of 3.7% during the forecast period with driven by the rising demand for biologic therapies for autoimmune disorders, expanding biosimilar adoption, increasing prevalence of rheumatoid arthritis and psoriasis, and continuous advancements…
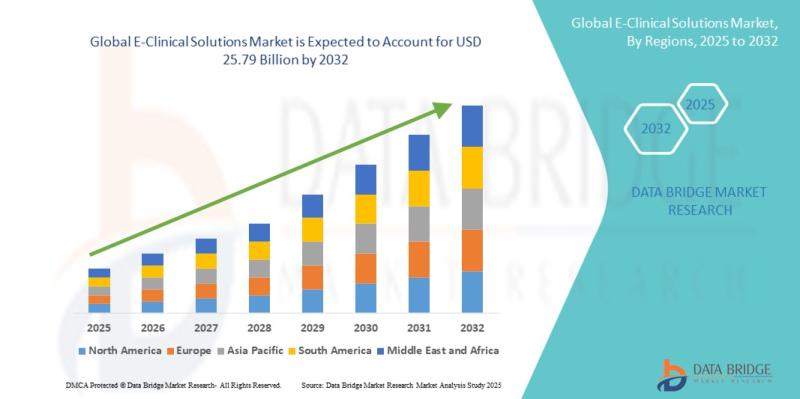
E-Clinical Solutions Market Size, Share & Industry Analysis, By Product, Deploym …
As per Data Bridge Market Research analysis, the E-Clinical Solutions Market was estimated at USD 10.62 billion in 2025. The market is expected to grow from USD 12.06 billion in 2026 to USD 25.79 billion in 2032, at a CAGR of 13.50% during the forecast period, driven by the rising demand for decentralized clinical trials, increasing adoption of cloud-based clinical data management platforms, growing pharmaceutical R&D investments, and expanding integration…
More Releases for Orphan
Acquired Orphan Blood Disease Market
Acquired Orphan Blood Disease Market to reach over USD 18.93 billion by the year 2031 - Exclusive Report by InsightAce Analytic
According to a new report by InsightAce Analytic, the "Acquired Orphan Blood Disease Market" in terms of revenue was estimated to be worth $8.65 billion in 2023 and is poised to reach $18.93 billion by 2031, growing at a CAGR of 10.47% from 2024 to 2031.
Get Free Access to…
Orphan Drugs Market Size to Hit $3199.3 Billion by 2028 | Orphan Drugs Industry …
Market Overview:
According to our experience research team, Orphan Drugs Market was valued at USD 112.36 Billion in 2021, and the global Orphan Drugs industry is projected to reach a value of USD 3199.3 Billion by 2028, at a CAGR of 7.4% during the forecast period 2022-2028
Vantage Market Research is a collection of market research studies on several industries, such as Chemicals, semiconductors & Electronics, Food & Beverages Technology, Energy &…
Orphan Drug Pipeline & Regulatory Insight 2025
Orphan Drug Pipeline & Regulatory Insight 2025" Report Highlights:
Global Orphan Drug Market Overview: US$ 260 Billion Opportunity
Global Orphan Drug market by Segment
Regulatory Landscape: USA, Europe & Asia
Orphan Drug Designation & Reimbursement Policy: USA, Europe & Asia
Global Orphan Drug Clinical Pipeline Insight: 973 Drugs
Marketed Orphan Drug Clinical Insight: 366 Drugs
The global orphan drug market opportunity is expected to surpass US$ 260 Billion by 2025 as per…
US Orphan Drug Pipeline Analysis
In recent years, the pharmaceutical industry has been experiencing a paradigm shift. While a large pool of patients was considered as a major source of revenue for pharma companies in the past, the focus is now gradually shifting to small sections of patients suffering from rare disease. In US, this pool of patients is gradually growing and orphan drugs are becoming an extremely attractive business proposition for the pharmaceuticals industry.…
Europe Orphan Drugs Pipeline Analysis
“Europe Orphan Drugs Pipeline Analysis” by PNS Pharma gives comprehensive insight on the various drug profiles under Orphan Drugs status in Europe. Research report covers all the ongoing drug development in various phases. Each drug profiles include detailed information like: Originator, Owner, Collaborator, Technology Provider, Licensee, Development Phase, Development Indications, Mechanism of Action, Chemical Formula, Country of Development and detailed analysis on the development process. The information for particular drug…
Global Orphan Drug Pipeline Analysis
In recent years, the pharmaceutical industry has been experiencing a paradigm shift. While a large pool of patients was considered as a major source of revenue for pharma companies in the past, the focus is now gradually shifting to small sections of patients suffering from rare disease. In US & Europe, this pool of patients is gradually growing and orphan drugs are becoming an extremely attractive business proposition for the…
