Press release
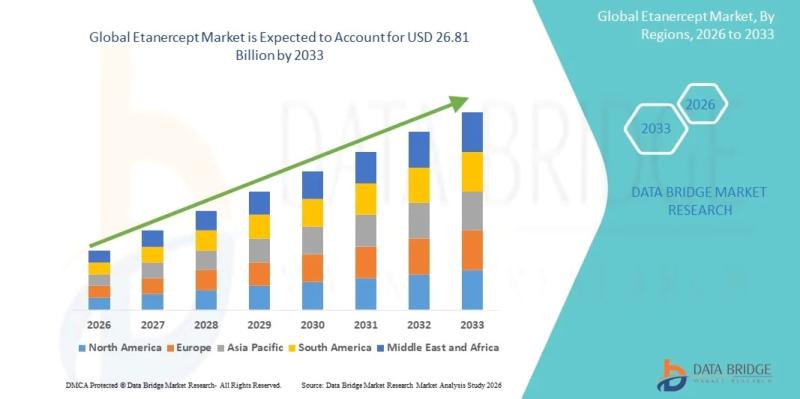
Etanercept Market Size, Share & Industry Trends Analysis Report, 2026-2033: Market Expected to Reach USD 26.81 Billion by 2033 at a CAGR of 3.7%
As per Data Bridge Market Research analysis, the Etanercept Market was estimated at USD 20.05 billion in 2025. The market is expected to grow from USD 20.79 billion in 2026 to USD 26.81 billion in 2033, at a CAGR of 3.7% during the forecast period with driven by the rising demand for biologic therapies for autoimmune disorders, expanding biosimilar adoption, increasing prevalence of rheumatoid arthritis and psoriasis, and continuous advancements in biologics manufacturing technologies.The global etanercept market is witnessing stable long-term growth due to the increasing burden of chronic inflammatory diseases, including rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, and plaque psoriasis. Rising healthcare expenditure, favorable reimbursement frameworks in developed economies, and growing physician preference for targeted biologic therapies continue to support market expansion. The growing commercialization of biosimilar etanercept products across Europe and Asia-Pacific is further improving patient accessibility and intensifying competitive pricing dynamics.
The market is also benefiting from advancements in biologics production, cold-chain logistics, and self-administration injectable devices. Regulatory agencies across North America, Europe, and Asia-Pacific are increasingly encouraging biosimilar approvals to reduce treatment costs and improve healthcare access. Strategic collaborations between pharmaceutical companies and regional distributors are accelerating market penetration across emerging economies.
Get the full PDF sample copy of the report: (Includes full table of contents, list of tables and figures, and graphs): https://www.databridgemarketresearch.com/request-a-sample/?dbmr=global-etanercept-market
Market Size & Forecast
2025 Market Size: USD 20.05 Billion
2026 Projected Market Size: USD 20.79 Billion
2033 Projected Market Size: USD 26.81 Billion
CAGR (2026-2033): 3.7%
Largest Region: North America
Fastest Growing Region: Asia-Pacific
Key Market Report Takeaways
North America accounted for the largest market share of approximately 42% in 2025 due to high biologics adoption, advanced healthcare infrastructure, and strong reimbursement coverage.
Asia-Pacific is projected to register the fastest CAGR during the forecast period owing to rising autoimmune disease prevalence, expanding healthcare access, and biosimilar commercialization.
The branded etanercept segment continues to hold the highest market share due to established clinical efficacy and strong physician confidence.
Rheumatoid arthritis remains the dominant application segment because of high global patient volume and long-term biologic treatment requirements.
Hospitals and specialty clinics represent the leading end-use segment due to higher biologic prescription rates and specialized autoimmune disease management.
Biosimilar penetration is increasing rapidly across Europe and Asia-Pacific as governments focus on reducing biologic treatment costs.
Self-injectable prefilled syringe and autoinjector formats are witnessing strong demand due to patient convenience and home-care treatment adoption.
Market Trends & Highlights
North America dominates the global etanercept market due to high biologic therapy adoption, strong reimbursement systems, advanced rheumatology treatment infrastructure, and the presence of major pharmaceutical manufacturers.
Asia-Pacific is emerging as the fastest-growing regional market driven by increasing autoimmune disease incidence, growing healthcare investments, expanding biosimilar approvals, and improving biologic accessibility in China and India.
Rheumatoid arthritis remains the leading application segment owing to rising disease prevalence, long-term treatment dependency, and increased physician preference for TNF-alpha inhibitors.
Market growth is supported by the combined impact of aging populations, increasing autoimmune disease burden, expanding biologics awareness, and higher healthcare expenditure globally.
Biosimilar innovation, advanced biologics manufacturing technologies, and patient-friendly injectable delivery systems are reshaping market competitiveness and pricing structures.
Regulatory support for biosimilars, strategic licensing agreements, and cross-border pharmaceutical partnerships are accelerating commercialization and regional market expansion.
View Report Insights: https://www.databridgemarketresearch.com/reports/global-etanercept-market
Market Dynamics
Market Drivers
Rising Prevalence of Autoimmune Disorders
The increasing global incidence of rheumatoid arthritis, psoriasis, ankylosing spondylitis, and psoriatic arthritis is a major growth driver for the etanercept market. Aging populations and changing lifestyle patterns are contributing to higher autoimmune disease diagnosis rates, particularly in North America and Europe. Long-term disease management requirements are increasing demand for biologic therapies with proven efficacy. Healthcare systems are also prioritizing early biologic intervention to reduce long-term disability and hospitalization costs.
Growing Adoption of Biologic Therapies
Biologics continue to gain strong clinical preference over conventional disease-modifying drugs due to superior therapeutic outcomes and targeted mechanisms of action. Etanercept remains widely prescribed because of its established safety profile and long-term clinical evidence. Increasing physician confidence in TNF-alpha inhibitors is supporting treatment expansion across multiple inflammatory indications. Rising awareness among patients regarding biologic treatment effectiveness is further strengthening market demand globally.
Expansion of Biosimilar Commercialization
The increasing approval and commercialization of etanercept biosimilars are significantly improving market accessibility and affordability. Europe has emerged as a leading region for biosimilar penetration due to supportive regulatory policies and reimbursement frameworks. Asia-Pacific countries are also accelerating biosimilar adoption to reduce biologic treatment costs. Competitive pricing from biosimilar manufacturers is expected to increase treatment penetration in cost-sensitive healthcare markets.
Advancements in Drug Delivery Technologies
Continuous innovation in injectable delivery systems, including autoinjectors and prefilled syringes, is improving patient adherence and convenience. Home-based biologic administration trends are encouraging pharmaceutical companies to develop user-friendly delivery devices. Technological improvements in biologics manufacturing are also enhancing production efficiency and reducing operational costs. These advancements are supporting wider adoption among chronic disease patients requiring long-term therapy.
Increasing Healthcare Expenditure and Insurance Coverage
Rising healthcare spending in developed and emerging economies is improving access to advanced biologic therapies. Public and private insurers in North America and Europe continue to expand reimbursement coverage for autoimmune disease treatment. Government-backed healthcare initiatives in Asia-Pacific are supporting biologic therapy inclusion in national healthcare programs. Improved reimbursement structures are reducing patient out-of-pocket expenses and increasing biologic therapy utilization.
Market Restraints
High Treatment Costs of Biologic Therapies
Etanercept therapies remain expensive compared to conventional anti-inflammatory medications, limiting accessibility in low- and middle-income economies. High manufacturing costs associated with biologic production continue to impact pricing structures. Even with biosimilar availability, affordability challenges persist in several developing healthcare systems. Cost pressures also influence reimbursement approvals and patient treatment continuity in some regions.
Stringent Regulatory Approval Processes
Biologic and biosimilar approvals require extensive clinical validation, quality testing, and pharmacovigilance compliance. Regulatory complexities across the U.S., Europe, and Asia-Pacific increase development timelines and commercialization costs. Differences in biosimilar interchangeability regulations create additional market entry barriers for manufacturers. Compliance-related expenses remain particularly challenging for smaller biotechnology firms.
Patent Litigation and Intellectual Property Disputes
The etanercept market has experienced prolonged patent-related legal disputes, especially in North America. Patent protection strategies adopted by originator companies can delay biosimilar market entry and restrict pricing competition. Litigation costs increase financial burdens for biosimilar developers and create uncertainty around commercialization timelines. These factors can limit rapid expansion of competitive biologic alternatives.
Adverse Effects and Safety Concerns
Etanercept therapy may be associated with risks including infections, injection-site reactions, and immune-related complications. Long-term biologic therapy monitoring requirements increase healthcare management complexity. Concerns regarding immunosuppression risks may reduce treatment adoption among specific patient populations. Strict pharmacovigilance requirements also increase operational responsibilities for healthcare providers and manufacturers.
Pricing Pressure from Competitive Biologics
The market faces increasing competition from alternative TNF inhibitors and next-generation biologics targeting autoimmune diseases. New therapeutic classes such as IL inhibitors and JAK inhibitors are intensifying pricing and market share pressure. Pharmaceutical companies are being forced to offer discounts and value-based pricing strategies to maintain competitiveness. These pressures may impact profit margins and long-term revenue growth.
Market Opportunities
Expansion in Emerging Healthcare Markets
Emerging economies across Asia-Pacific, Latin America, and the Middle East offer significant untapped growth potential due to improving healthcare infrastructure and rising autoimmune disease awareness. Governments are increasing investments in biologic therapy accessibility and specialty care facilities. Expanding insurance coverage in countries such as China and India is expected to improve patient access to biologic therapies. Pharmaceutical companies are actively targeting these regions through partnerships and localized commercialization strategies.
Growth of Biosimilar Development Programs
The increasing demand for cost-effective biologics is creating strong opportunities for biosimilar manufacturers. Regulatory agencies are streamlining biosimilar approval pathways to accelerate market competition and improve healthcare affordability. Strategic investments in biosimilar R&D and manufacturing capacity expansion are expected to drive future market growth. Companies with strong biosimilar portfolios are likely to gain competitive advantages in price-sensitive markets.
Advancements in Personalized Medicine
Precision medicine and biomarker-based treatment approaches are creating opportunities for targeted autoimmune disease management. Advances in genomic diagnostics and patient stratification technologies are improving treatment response predictability. Pharmaceutical companies are increasingly integrating real-world evidence and personalized treatment models into biologic therapy strategies. These developments are expected to improve therapeutic outcomes and patient retention rates.
Strategic Collaborations and Licensing Agreements
Pharmaceutical companies are increasingly engaging in co-development agreements, regional licensing partnerships, and distribution alliances to strengthen market reach. Collaborations between biotechnology firms and regional healthcare providers are accelerating commercialization in emerging economies. Such partnerships help companies optimize supply chains, regulatory approvals, and market penetration. Strategic alliances are expected to remain a major competitive growth strategy over the forecast period.
Increasing Adoption of Home Healthcare Solutions
The shift toward home-based biologic administration is creating opportunities for advanced self-injection systems and digital patient monitoring solutions. Demand for convenient long-term autoimmune disease management is increasing globally. Pharmaceutical companies are investing in patient-support programs and smart injection technologies to improve adherence rates. The home healthcare trend is expected to drive demand for user-friendly etanercept formulations.
Market Challenges
Complex Biologics Manufacturing Processes
Etanercept production requires sophisticated biologics manufacturing infrastructure, strict sterility standards, and advanced quality control systems. Manufacturing complexity increases operational costs and limits rapid production scalability. Supply disruptions in raw materials or bioprocessing components can significantly affect market availability. Smaller manufacturers often face barriers in achieving efficient large-scale biologic production capabilities.
Global Supply Chain Vulnerabilities
Biologic drugs require highly controlled cold-chain logistics and temperature-sensitive transportation systems. Geopolitical tensions, transportation delays, and raw material shortages can disrupt global distribution networks. Supply chain instability particularly affects emerging markets with limited cold-chain infrastructure. Maintaining product integrity throughout international distribution remains a major operational challenge.
Regulatory Variability Across Regions
Different regulatory standards for biosimilars across North America, Europe, and Asia-Pacific create commercialization complexity for multinational companies. Variations in interchangeability policies and pharmacovigilance requirements increase administrative burdens. Companies must navigate region-specific clinical documentation and approval pathways, resulting in higher compliance costs. Regulatory inconsistencies can delay product launches and restrict cross-border market expansion.
Competitive Pressure from Alternative Therapies
The autoimmune disease treatment landscape is becoming increasingly competitive with the emergence of novel biologics and oral therapies. New drug classes with differentiated mechanisms of action are attracting physician and patient interest. Strong competition may reduce etanercept prescription volumes in specific therapeutic areas. Companies must continuously invest in product differentiation and lifecycle management to maintain market positioning.
Reimbursement and Pricing Constraints
Healthcare payers are increasingly implementing cost-control measures and formulary restrictions for high-cost biologics. Insurance providers in several markets are prioritizing lower-cost biosimilars and alternative therapies. Pricing negotiations and reimbursement delays can impact treatment accessibility and manufacturer profitability. These financial pressures are particularly prominent in publicly funded healthcare systems.
Market Segmentation & Analysis
By Product Type
Branded Etanercept
Branded etanercept products continue to dominate the market due to strong physician trust, established efficacy, and extensive clinical evidence across autoimmune indications. These products accounted for the largest revenue share in 2025, particularly in North America and developed European markets. Strong brand recognition and long-term patient adherence continue to support market leadership. However, growth is expected to moderate due to increasing biosimilar competition.
Etanercept Biosimilars
The biosimilar segment is projected to register the fastest CAGR during the forecast period due to rising cost-containment initiatives and favorable regulatory support. Europe remains the leading biosimilar adoption market, while Asia-Pacific is witnessing rapid commercialization growth. Biosimilars are gaining traction among healthcare providers seeking affordable biologic alternatives. Expanding reimbursement inclusion and competitive pricing are accelerating segment expansion globally.
By Application
Rheumatoid Arthritis
Rheumatoid arthritis represents the dominant application segment due to high global disease prevalence and long-term biologic treatment dependency. Etanercept is widely prescribed for moderate-to-severe rheumatoid arthritis management because of its proven anti-inflammatory effectiveness. North America and Europe contribute significantly to segment revenue due to established treatment protocols. The segment is expected to maintain stable growth throughout the forecast period.
Psoriatic Arthritis
The psoriatic arthritis segment is witnessing strong demand due to increasing diagnosis rates and improved awareness regarding biologic therapies. Etanercept remains a preferred TNF inhibitor for reducing joint inflammation and improving mobility. Growing dermatology-rheumatology integrated care models are supporting segment growth. Asia-Pacific is expected to witness increasing adoption due to expanding biologics accessibility.
Plaque Psoriasis
Plaque psoriasis treatment applications continue to expand with growing patient preference for targeted biologic therapies. Rising demand for long-term disease management solutions and improved treatment efficacy is supporting segment growth. Increased awareness campaigns and dermatology consultations are contributing to higher biologic therapy adoption. Europe remains a significant contributor due to advanced biologic reimbursement systems.
Ankylosing Spondylitis
Etanercept adoption for ankylosing spondylitis treatment is increasing due to its effectiveness in reducing spinal inflammation and improving physical mobility. The segment benefits from rising diagnosis rates and improved access to rheumatology care. Healthcare providers increasingly recommend biologic therapies for patients with inadequate response to conventional treatments. Moderate but stable growth is expected globally.
By End User
Hospitals
Hospitals hold the largest market share due to high biologic prescription rates, advanced infusion infrastructure, and specialist availability. Major healthcare institutions in North America and Europe dominate biologic therapy administration. Hospital-based treatment ensures better patient monitoring and pharmacovigilance compliance. The segment continues to benefit from increasing autoimmune disease hospitalization and specialist consultations.
Specialty Clinics
Specialty clinics are emerging as a fast-growing segment due to increasing outpatient autoimmune disease treatment trends. Rheumatology and dermatology clinics are witnessing rising biologic therapy adoption for chronic disease management. Improved patient accessibility and lower treatment costs compared to hospital settings are supporting segment expansion. Asia-Pacific is experiencing rapid specialty clinic growth.
Homecare Settings
Homecare settings are gaining traction with the increasing adoption of self-injectable biologic devices. Patients increasingly prefer home-based administration for convenience and reduced hospital visits. Pharmaceutical companies are investing in patient-support programs and digital monitoring tools to strengthen this segment. The homecare segment is projected to witness above-average growth during the forecast period.
Regional Analysis
By geography, the market is categorized into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
North America
North America dominates the global etanercept market, accounting for approximately 42% of total revenue in 2025. The region benefits from advanced healthcare infrastructure, strong biologics adoption, and high autoimmune disease diagnosis rates. The U.S. remains the primary contributor due to favorable reimbursement frameworks and the presence of major biologics manufacturers. Strong R&D investments and widespread specialist access continue to support regional market leadership. Robust regulatory systems and high biologic treatment penetration further strengthen demand across the region.
Europe
Europe represents a mature and established market driven by strong biosimilar adoption and advanced regulatory support. Countries such as Germany, the U.K., and France are major contributors due to high healthcare expenditure and widespread biologic therapy reimbursement. The region has emerged as a global leader in biosimilar commercialization, supporting cost-efficient autoimmune disease treatment. Continuous investments in biologics research and healthcare modernization are sustaining market expansion. Favorable regulatory pathways continue to encourage competition and pricing optimization.
Asia Pacific
Asia-Pacific is projected to register the fastest growth during the forecast period due to rapid healthcare expansion and increasing autoimmune disease prevalence. China, India, and Japan are leading contributors supported by rising biologics awareness and improving reimbursement systems. Government initiatives to strengthen domestic biologics manufacturing and biosimilar approvals are accelerating regional growth. Expanding middle-class populations and rising healthcare spending are improving treatment accessibility. The region is also witnessing increasing investments from multinational pharmaceutical companies.
Latin America
Latin America represents an emerging market with gradual biologics adoption and improving healthcare infrastructure. Brazil and Mexico account for the majority of regional demand due to expanding autoimmune disease treatment capabilities. Economic constraints and reimbursement limitations continue to restrict widespread biologic penetration. However, growing physician awareness and biosimilar availability are improving market accessibility. Investments in specialty healthcare services are expected to support long-term market development.
Middle East & Africa
The Middle East & Africa market is witnessing steady but comparatively slower growth due to uneven healthcare infrastructure development. Government healthcare investments and private sector expansion are improving access to advanced biologic therapies in select countries. Gulf Cooperation Council nations are leading regional adoption due to stronger healthcare spending and insurance coverage. However, limited specialist availability and infrastructure gaps continue to challenge broader market penetration. Increasing awareness regarding autoimmune disease treatment is expected to support gradual growth.
Key Insights
Largest Region: North America
Fastest Growing Region: Asia-Pacific
Mature Markets: North America, Europe
High-Growth Markets: Asia-Pacific
Emerging Markets: Latin America, Middle East & Africa
Regional growth differences are influenced by healthcare expenditure levels, biosimilar adoption rates, local manufacturing capabilities, regulatory environments, and reimbursement accessibility. Developed markets benefit from advanced healthcare systems and strong biologics infrastructure, while emerging regions are driven by expanding healthcare access and increasing pharmaceutical investments.
Competitive Landscape
Market Structure Overview
The global etanercept market is moderately consolidated, characterized by the presence of multinational biologics manufacturers, biosimilar developers, and regional pharmaceutical companies. Competition is strongly influenced by biologic innovation, biosimilar pricing strategies, regulatory approvals, and global distribution capabilities. Leading players maintain strong market positions through extensive product portfolios, advanced manufacturing infrastructure, and strategic commercialization partnerships. Competitive landscape analysis helps assess market positioning, technological strengths, geographic reach, and long-term strategic direction of key participants.
Key Industry Players
Leading market participants focus on biologics innovation, biosimilar expansion, and strategic partnerships to strengthen global market presence. Major companies emphasize manufacturing scalability, regulatory approvals, and autoimmune disease portfolio diversification. Biosimilar developers are increasingly competing through cost-efficient alternatives and regional expansion strategies. Companies are also investing in patient-support services and advanced self-administration technologies to improve treatment adherence and customer retention.
List of Key Industry Players
Amgen
Pfizer
Samsung Bioepis
Sandoz
Biocon Biologics
Lupin
Celltrion
Fresenius Kabi
Teva Pharmaceutical Industries
Alvotech
Competitive Strategies
Market participants are actively pursuing biosimilar launches, product differentiation, and geographic expansion strategies to strengthen competitiveness. Strategic partnerships and licensing agreements are enabling companies to accelerate commercialization across emerging markets. Mergers, acquisitions, and manufacturing capacity expansions remain key approaches for improving operational scale and portfolio diversification. Companies are increasingly investing in biologics analytics, digital healthcare integration, and patient-centric delivery technologies to improve market positioning and long-term profitability.
Emerging Players & Market Dynamics
Emerging biosimilar companies and regional biotechnology firms are increasing market competition through affordable biologic alternatives and specialized commercialization strategies. Startups are focusing on cost-efficient manufacturing technologies and targeted autoimmune therapy development. Rising investments in biosimilars research and favorable regulatory reforms are encouraging new market entrants globally. Increasing digital transformation and AI-driven biologics development are expected to further reshape competitive dynamics during the forecast period.
Latest Developments
July 2025 - Biocon Biologics launched Nepexto®, a biosimilar to Enbrel® (etanercept), in Australia through a partnership with Generic Health. The launch expanded biosimilar accessibility in the Asia-Pacific region and strengthened competitive positioning in immunology therapeutics.
May 2024 - Lupin launched the Rymti® etanercept biosimilar in Canada in collaboration with Sandoz Canada. The product expansion improved patient access to lower-cost biologic therapies and intensified competition in the North American biosimilars market.
February 2026 - European Medicines Agency (EMA) adopted a positive opinion for Fubelv, a biosimilar etanercept product developed by Biosimilar Collaborations Ireland Limited. The approval supports continued biosimilar penetration across Europe and strengthens cost-reduction strategies in autoimmune disease treatment.
April 2025 - Sandoz filed an antitrust lawsuit against Amgen in the U.S. regarding Enbrel biosimilar market access restrictions. The case highlighted increasing legal and competitive pressures surrounding biologic patent exclusivity and biosimilar commercialization.
November 2025 - A published clinical review on etanercept biosimilar switching highlighted growing real-world evidence supporting biosimilar efficacy and safety. The findings are expected to strengthen physician confidence and support broader biosimilar adoption globally.
March 2025 - A multicenter Japanese clinical study evaluating etanercept biosimilars demonstrated positive therapeutic outcomes in rheumatoid arthritis patients. The research reinforced the role of biosimilars in expanding affordable autoimmune disease treatment options across Asia-Pacific markets.
October 2025 - Regulatory guidance updates from U.S. authorities simplified biosimilar approval requirements by reducing comparative efficacy study burdens for certain biologics. The regulatory shift is expected to accelerate biosimilar development pipelines and increase future market competition.
Browse More Reports:
https://www.databridgemarketresearch.com/reports/canada-home-healthcare-market
https://www.databridgemarketresearch.com/reports/global-cephalosporins-market
https://www.databridgemarketresearch.com/reports/north-america-musculoskeletal-msk-disease-management-market
https://www.databridgemarketresearch.com/reports/global-ulcerative-colitis-market
https://www.databridgemarketresearch.com/reports/global-urinary-incontinence-market
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email:- corporatesales@databridgemarketresearch.com
About Data Bridge Market Research:
An absolute way to forecast what the future holds is to comprehend the trend today!
Data Bridge Market Research set forth itself as an unconventional and neoteric market research and consulting firm with an unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Etanercept Market Size, Share & Industry Trends Analysis Report, 2026-2033: Market Expected to Reach USD 26.81 Billion by 2033 at a CAGR of 3.7% here
News-ID: 4514851 • Views: …
More Releases from Data Bridge Market Research
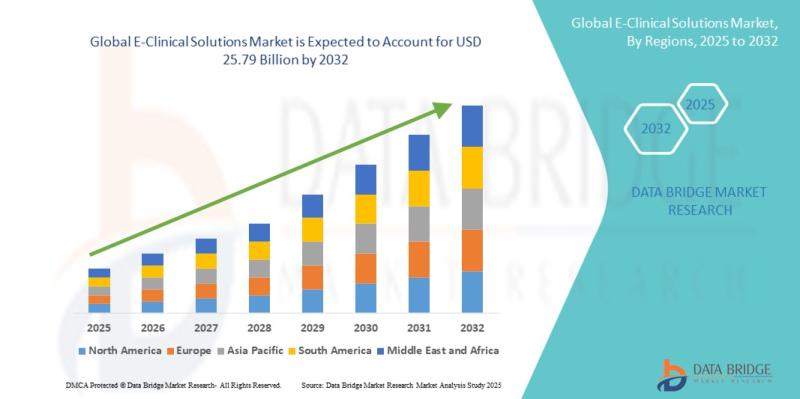
E-Clinical Solutions Market Size, Share & Industry Analysis, By Product, Deploym …
As per Data Bridge Market Research analysis, the E-Clinical Solutions Market was estimated at USD 10.62 billion in 2025. The market is expected to grow from USD 12.06 billion in 2026 to USD 25.79 billion in 2032, at a CAGR of 13.50% during the forecast period, driven by the rising demand for decentralized clinical trials, increasing adoption of cloud-based clinical data management platforms, growing pharmaceutical R&D investments, and expanding integration…
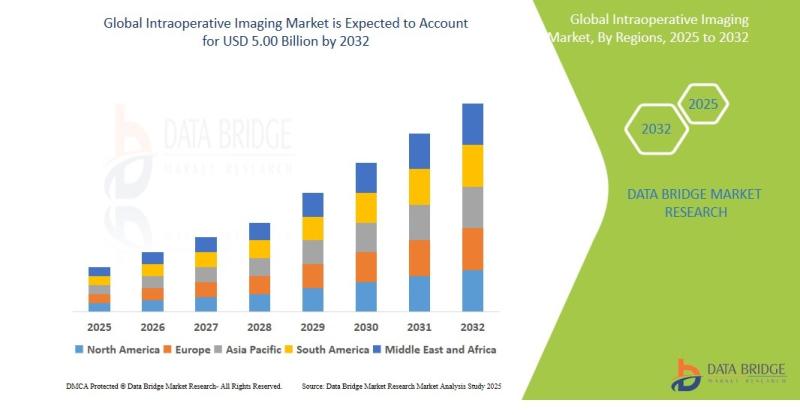
Intraoperative Imaging Market Size, Share & Industry Trends Analysis Report, 202 …
Intraoperative Imaging Market Summary
As per Data Bridge Market Research analysis, the Intraoperative Imaging Market was estimated at USD 3.37 billion in 2025. The market is expected to grow from USD 3.56 billion in 2026 to USD 5.00 billion in 2032, at a CAGR of 5.82% during the forecast period with driven by the rising demand for advanced surgical visualization technologies, increasing adoption of minimally invasive procedures, expanding investments in hybrid…
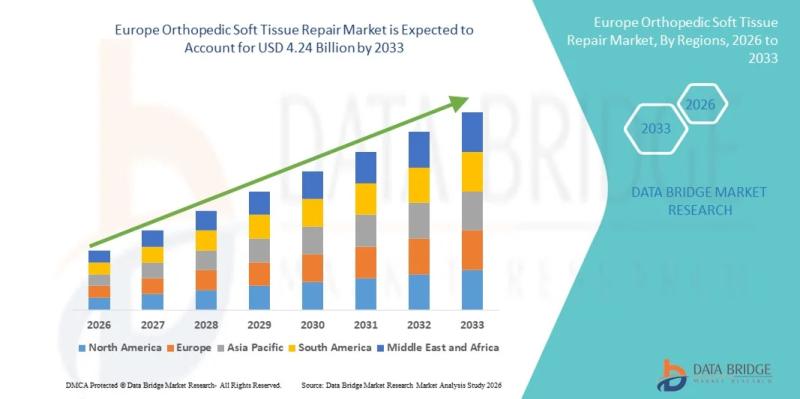
Europe Orthopedic Soft Tissue Repair Market Size, Share & Forecast 2026-2033
As per Data Bridge Market Research analysis, the Europe Orthopedic Soft Tissue Repair Market was estimated at USD 2.33 billion in 2025. The market is expected to grow from USD 2.33 billion in 2025 to USD 4.24 billion by 2033, at a CAGR of 7.8% during the forecast period with driven by the rising demand for minimally invasive orthopedic procedures, increasing sports-related injuries, and advancements in biomaterials and surgical technologies.
Growth…
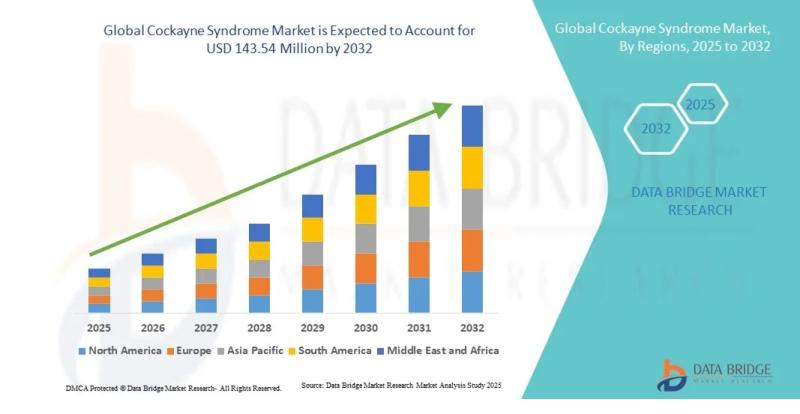
Cockayne Syndrome Market to Grow at a CAGR of 4.30% During 2026-2032
Market Summary
"As per Data Bridge Market Research analysis, the Cockayne Syndrome Market was estimated at USD 106.91 million in 2025. The market is expected to grow from USD 111.51 million in 2026 to USD 143.54 million in 2032, at a CAGR of 4.30% during the forecast period with driven by the rising demand for advanced rare disease therapeutics, increasing investments in gene therapy research, expanding orphan drug development programs, and…
More Releases for America
Stabilit America Highlights Applications of Fiberglass Roof Panels with Stabilit …
Roofing materials are very important in the realm of modern construction, as they should be long lasting, economical and attractive. Fiberglass roof panels are a few of the numerous choices among several alternatives that have received a reputation of being versatile, long life, and adaptable in various sectors. They are favored by the architects, contractors, and property developers due to their lightweight construction, resistance to weather factors, and the ease…
Deodorants Market Report by Region (North America, EMEA, Latin America, Asia)
2025 - Pristine Market Insights, a leading market research firm, announced the release of its latest and comprehensive market research report on Deodorants market. The report spans over 500 pages and delivers 10-year market forecast in US dollars (or custom currencies upon request). It provides in-depth analysis of market dynamics (drivers, opportunities, restraints), PESTLE insights, latest industry trends, and demand factors. The report includes segmented market value, share (%), compound…
Sequestrant Market Report by Region (North America, EMEA, Latin America, Asia)
2025 - Pristine Market Insights, a leading market research firm, announced the release of its latest and comprehensive market research report on Sequestrant market. The report spans over 500 pages and delivers 10-year market forecast in US dollars (or custom currencies upon request). It provides in-depth analysis of market dynamics (drivers, opportunities, restraints), PESTLE insights, latest industry trends, and demand factors. The report includes segmented market value, share (%), compound…
Buttermilk Market Study by Region (North America, Latin America, Europe, Asia, M …
2025 - Pristine Market Insights, a leading market research firm, announced the release of its latest and comprehensive market research report on Buttermilk market. The report spans over 500 pages and delivers 10-year market forecast in US dollars (or custom currencies upon request). It provides in-depth analysis of market dynamics (drivers, opportunities, restraints), PESTLE insights, latest industry trends, and demand factors. The report includes segmented market value, share (%),…
Textiles Market Analysis Report, Regional Outlook - Europe, North America, South …
Adroit Market Research has announced the addition of the “Global Textiles Market Size Status and Forecast 2025”, The report classifies the global Textiles in a precise manner to offer detailed insights about the aspects responsible for augmenting as well as restraining market growth.
This report studies the global Textiles Speaker market, analyzes and researches the Textiles Speaker development status and forecast in Europe, North America, Central America, South America, Asia Pacific…
Global Gaucher Disease Market 2018 Covering North America, South America, Europe
Gaucher Disease Market
Summary
The Global Gaucher Disease Market is defined by the presence of some of the leading competitors operating in the market, including the well-established players and new entrants, and the suppliers, vendors, and distributors. The key players are continuously focusing on expanding their geographic reach and broadening their customer base, in order to expand their product portfolio and come up with new advancements.
Gaucher Disease market size to maintain the average annual growth…