Press release
Melanoma Drugs Market 2016 : Evolving Technology, Trends And Industry Analysis – 2023
Melanoma is a form of skin cancer. Some of the major risk factors for melanoma are high levels of exposure to ultraviolet (UV) light leading to blistering sunburns, especially among population groups who are either in countries, which are in proximity to the equator or at very high altitudes.According to the American Academy of Dermatology, the prevalence of melanoma is gradually increasing and rapidly spreads to other parts of the body. However, if melanoma is detected at an early stage i.e. in situ or stage 0. In this stage, the cure rate with surgical removal is nearly 100i% without any delays in seeking treatment; the survival rates from the disease are nearly 100 percent. When melanoma has spread to other body parts or has penetrated deeper into the skin or spreads, the treatment often becomes more complex. Hence, the systemic drug therapy for melanoma largely depends on the properties and stage at which melanoma is detected.
Obtain Report Details @ http://www.transparencymarketresearch.com/melanoma-drugs-market.html
Since, 2011 various drugs have received FDA approval for treatment of melanoma. The melanoma drugs market has been segmented based on type of drug therapy into chemotherapy, immunotherapy and, targeted therapy. Immunotherapy works on the mechanism of stimulating persons own immune system response to distinguish cancer cells from normal cells more efficiently. A number of types of immunotherapy treatments can be used to treat melanoma. During the past few years, many new immunotherapy drugs for melanoma have entered the market. Immunotherapy drugs for melanoma include Keytruda(pembrolizumab) and Opdivo (nivolumab) which got FDA approval in 2014. Above mentioned drugs target PD-1, a protein on immune system cells called T cells. By blocking PD-1 protein, these drugs increase the immune response of the body against melanoma cells. Recently, the FDA approval of Yervoy (ipilimumab) in October 2015 has opened new market opportunities for market expansion of immunotherapy drugs in melanoma drugs market. Ipilimumab (Yervoy) is also another type of drug that upsurges immune response. However, it blocks CTLA-4, another protein on T cells.
These drugs are given as an intravenous (IV) infusion every 2 or 3 weeks. This results in shrinking of tumor. This treatment can help number of people with stage IV melanoma to live longer. However, it’s not yet clear if these drugs can completely cure melanoma.
These drugs act as checkpoint inhibitors to fight against melanoma cancer. In addition, there is a strong pipeline for melanoma treatment drugs under development by major key players and other new entrants in this market. Thus, taking into consideration the strategic new product launches after FDA approval the melanoma drugs market is expected to grow exponentially.
Similarly the Cytokines are proteins in the body that increase the immune system. Artificial cytokines, such as interferon-alfa and interleukin-2 (IL-2), are occasionally used in patients with melanoma.
When chemotherapy drugs fail to deliver positive results in melanoma patients, targeted therapy drugs are administered to them. The targeted therapy drugs include BRAF inhibitors such as Zelboraf (vemurafenib) and Tafinlar (dabrafenib). The BRAF drugs work on the mechanism of shrinking tumors among those metastatic melanoma patients who have undergone genetic mutation. They are found to increase the progression free survival rate of patients. The MEK inhibitors such as trametinib (Mekinist) and cobimetinib (Cotellic) block the MEK proteins. In November 2015, Cotellic (cobimetinib) of Genentech Inc. received approval from FDA for the treatment of BRAF V600E or V600K melanoma. Generally Cotellic is used in combination with vemurafenib, a BRAF inhibitor, to treat melanoma.
On October 27th 2015, Amgen Inc.’s Oncolytic virus therapy for a deadly form of skin cancer/ melanoma was approved by U.S. FDA. The drug is known as talimogene laherparepvec, or T-vec, was cleared as a treatment for melanoma lesions in the skin and lymph nodes that can’t be removed completely by surgery. According to FDA brand-name of the drug is Imlygic. It uses adapted version of the herpes simplex virus to attack cancerous cells. After being injected into the tumor, the virus replicates until it causes the cells to breach and it results in provoking an immune system response that communicates the body to recognize the cancer.
Fill The Form To Gain Deeper Insights On This Market @ http://www.transparencymarketresearch.com/sample/sample.php?flag=B&rep_id=8443
Targeted therapy drugs have lower severity of side effects compared to other melanoma cancer drugs. However, despite of the new market opportunities created as a result of recent FDA approvals, some of the major restraints in the market are attributed to the lack of innovative and successful therapies in the market to reduce the risk of mortality of advanced metastatic melanoma patients. Thus, targeted therapy and their combinations could lead the melanoma drugs market and play a potential role in driving in melanoma drug market.
Some of the major players in this market include Amgen, Inc., Bristol-Myers Squibb, Eisai Co. Ltd., F. Hoffmann-La Roche Ltd., Genentech Inc. Janssen Biotech, Inc., and Novartis International AG. Pfizer, Vical and ZIOPHARM.
About Us
Transparency Market Research (TMR) is a global market intelligence company providing business information reports and services. The company’s exclusive blend of quantitative forecasting and trend analysis provides forward-looking insight for thousands of decision makers. TMR’s experienced team of analysts, researchers, and consultants use proprietary data sources and various tools and techniques to gather and analyze information.
Contact us:
Transparency Market Research
90 State Street,
Suite 700,
Albany
NY - 12207
United States
Tel: +1-518-618-1030
USA - Canada Toll Free 866-552-3453
Email: sales@transparencymarketresearch.com
Website: http://www.transparencymarketresearch.com/
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Melanoma Drugs Market 2016 : Evolving Technology, Trends And Industry Analysis – 2023 here
News-ID: 602497 • Views: …
More Releases from Transparency Market Research

Medium Voltage Fuse Market Outlook 2031: Global Market to Reach US$ 1.8 Billion …
The global medium voltage fuse market is steadily transitioning from a traditional grid-protection niche to a strategic enabler of modern power systems. Rising investments in renewable energy integration, large-scale electrification programs, and infrastructure upgrades are reshaping demand patterns worldwide. Medium voltage fuses-typically rated between 1 kV and 35 kV-are no longer viewed as passive safety components; instead, they are increasingly recognized as critical assets for grid stability, asset protection, and…
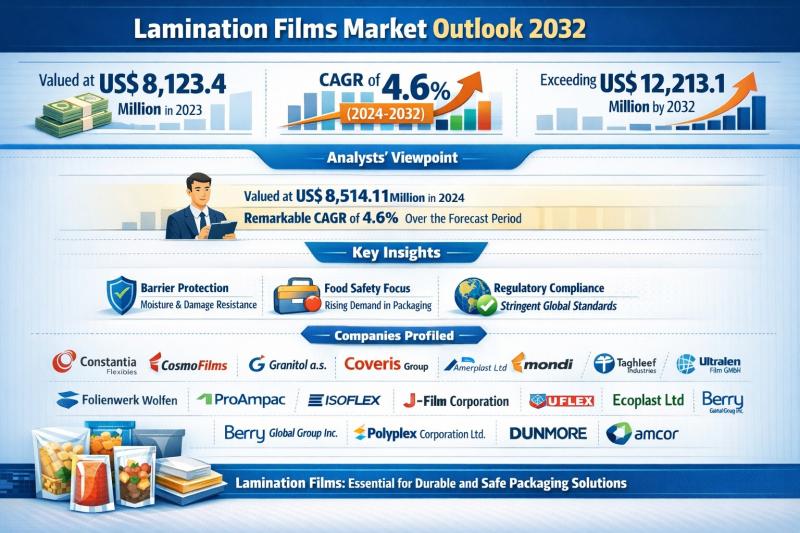
Lamination Films Market Outlook 2032: Global Industry Size to Surpass US$ 12.21 …
The global lamination films market was valued at US$ 8,123.4 million in 2023 and is forecast to reach US$ 8,514.1 million in 2024. Over the forecast period from 2024 to 2032, the market is projected to expand at a compound annual growth rate (CAGR) of 4.6%, ultimately exceeding US$ 12,213.1 million by 2032. This steady growth trajectory reflects the indispensable role of lamination films in modern packaging ecosystems across food,…

Global Curcumin Market Outlook 2031: Natural Antioxidant Demand, Regional Growth …
The global curcumin market is entering a structurally strong growth phase, underpinned by rising consumer preference for natural, plant-based ingredients and increasing clinical validation of curcumin's antioxidant and anti-inflammatory properties. As consumers shift away from synthetic additives and chemical-based therapeutics, curcumin is emerging as a high-value bioactive ingredient across , functional foods, cosmetics, and pharmaceuticals. Premiumization trends in organic products, combined with regulatory validation from food safety authorities, are expected…

Hemoglobin A1c Testing Devices Market to be Worth More Than USD 3.3 Bn by 2034 - …
The global Hemoglobin A1c (HbA1c) Testing Devices Market was valued at US$ 1.4 Bn in 2023 and is projected to expand at a CAGR of 7.7% from 2024 to 2034, reaching more than US$ 3.3 Bn by the end of 2034. The market growth is primarily attributed to the increasing global burden of diabetes, growing awareness about disease management, and technological advancements in diagnostic devices.
Get a concise overview of key…
More Releases for FDA
DreaMed receives 5th FDA Clearance
TEL AVIV, Israel: DreaMed Diabetes LTD. ("DreaMed" or the "Company"), developer of the endo.digital Clinical Decision Support System announced today that it has received its 5th U.S Food and Drug Administration (FDA) clearance that expands the scope of AI enhanced treatment recommendations to patients on fixed meal insulin regimens. endo.digital is the first decision support system that has been cleared to assist healthcare providers in the management of diabetes…
FDA Compliant Blood Storage and Preservation
Accsense Monitoring System Automates Data Archive and Alarming
CAS DataLoggers provided the temperature alarming and monitoring system to a hospital blood bank looking to replace their old paper chart recorders as they became unreliable and spare parts were harder to find. For proper blood storage and preservation, the lab’s medical units needed to maintain storage temperatures between 2°C to 6°C (36°F to 43°F), given the perishability of blood components. The facility…
FDA grants orphan drug status to Vicore
US Food and Drug Administration has awarded Vicore Pharmaceuticals with orphan Drug designation for the treatment of Idiopathic Pulmonary Fibrosis (IPF). FDA’s Orphan Drug Designation program provides certain incentives for companies developing therapeutics to treat rare diseases or conditions, defined as those affecting less than 200,000 individuals in the U.S. A drug candidate and its sponsor must meet several key criteria in order to qualify for, and obtain, orphan drug…
New FDA Design Control Training Courses
Salt Lake City, Utah - February 23 2017 - Procenius Consulting is a medical device consulting firm specializing solely in medical device design controls regulation (21 CFR 820.30).
Announcing New Design Control Training Courses
Procenius Consulting has just launched two new training courses covering basic and advanced topics of medical device design control regulation. These courses focus on compliance, practical implementation and industry best practices techniques for developing or improving a…
fda online training
GRC Training Solutions provides end-to-end FDA compliance solutions for those companies who want to maximize security, minimize operational costs, improve staff productivity and stay on top of all their compliance documentation.
GRC Training Solutions boasts a team of experts and specialists who have a proven track record in working with the biotechnology, medical device, diagnostic and pharmaceutical fields. Our team will work with you closely and develop solutions that meet…
FDA online training
Description:
Device firms, establishments or facilities that are involved in the production and distribution of medical devices intended for use in the U.S are required to register annually. Most establishments that are required to register with the FDA are also required to list the devices that are made there and the activities that are performed on those devices. Initially, FDA issued a 28-page Proposed Rule that would amend its regulations regarding…
