Press release
Cockayne Syndrome Market to Grow at a CAGR of 4.30% During 2026-2032
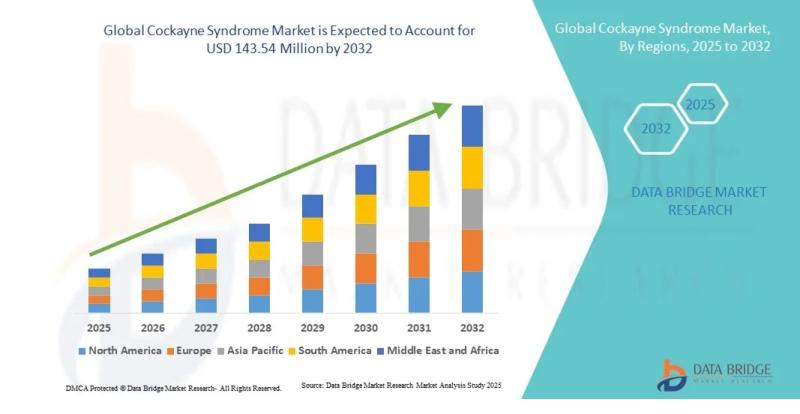
Market Summary"As per Data Bridge Market Research analysis, the Cockayne Syndrome Market was estimated at USD 106.91 million in 2025. The market is expected to grow from USD 111.51 million in 2026 to USD 143.54 million in 2032, at a CAGR of 4.30% during the forecast period with driven by the rising demand for advanced rare disease therapeutics, increasing investments in gene therapy research, expanding orphan drug development programs, and improving genetic diagnostic capabilities."
The global Cockayne syndrome market is witnessing steady growth due to increasing awareness regarding rare genetic disorders, advancements in genomic sequencing technologies, and rising investments in precision medicine. The market is primarily driven by the growing focus on orphan drug development, favorable regulatory frameworks for rare disease therapies, and increasing collaborations between biotechnology firms, academic institutions, and research organizations. Improvements in early diagnosis through next-generation sequencing (NGS) and molecular diagnostics are also supporting market expansion. Additionally, ongoing clinical research into gene therapy and DNA repair-targeted therapeutics is creating long-term growth opportunities across North America, Europe, and Asia-Pacific.
Get the full PDF sample copy of the report: (Includes full table of contents, list of tables and figures, and graphs) https://www.databridgemarketresearch.com/request-a-sample/?dbmr=global-cockayne-syndrome-market
Market Size & Forecast
2025 Market Size: USD 106.91 Million
2026 Projected Market Size: USD 111.51 Million
2032 Projected Market Size: USD 143.54 Million
CAGR (2026-2032): 4.30%
Largest Region: North America
Fastest Growing Region: Asia-Pacific
Key Market Report Takeaways
North America accounted for the largest market share of approximately 41% due to strong rare disease research infrastructure, high healthcare expenditure, and favorable orphan drug regulations.
Asia-Pacific is projected to register the fastest CAGR during the forecast period owing to increasing genetic testing adoption, improving healthcare access, and growing biotechnology investments in China, Japan, and India.
The diagnostic testing segment holds the highest market share due to rising early disease screening and wider availability of genomic sequencing technologies.
The gene therapy and precision medicine application segment is emerging as the dominant research focus driven by advancements in DNA repair technologies and rare disease therapeutics.
The specialty hospitals and research institutes segment represents the leading end-use category due to concentration of advanced diagnostic and treatment capabilities.
Increasing orphan drug incentives and government-backed rare disease initiatives are accelerating pipeline development globally.
Strategic collaborations among biotech firms, academic institutions, and patient advocacy groups are enhancing clinical research activities.
Details about the report and current availability can be viewed : https://www.databridgemarketresearch.com/reports/global-cockayne-syndrome-market
Market Trends & Highlights
North America remains the leading regional market due to advanced rare disease treatment infrastructure, high R&D expenditure, strong reimbursement systems, and active participation of biotechnology companies in orphan drug development.
Asia-Pacific is the fastest-growing regional market supported by increasing investments in genomic medicine, improving healthcare accessibility, and rising awareness regarding rare genetic disorders.
Diagnostic and genetic testing applications dominate the market owing to the increasing use of next-generation sequencing and molecular diagnostics for early identification of Cockayne syndrome.
Rising prevalence awareness of ultra-rare disorders, expanding orphan drug policies, and increasing public-private research funding continue to drive market growth globally.
Emerging technologies including CRISPR-based gene editing, viral vector gene therapy, AI-enabled genomic analysis, and precision medicine platforms are reshaping therapeutic development.
Regulatory support from agencies such as the U.S. FDA and EMA for orphan drugs and rare disease clinical trials is accelerating innovation and investment activities.
Market Dynamics
Market Drivers
Increasing Focus on Rare Disease Research
Governments and healthcare organizations are increasing investments in rare disease research programs globally. North America and Europe continue to allocate substantial funding toward orphan disease therapeutics and genetic disorder studies. This has accelerated research activity in DNA repair disorders including Cockayne syndrome. Increased patient advocacy and awareness campaigns are also improving disease recognition and diagnosis rates.
Advancements in Genetic Testing Technologies
Rapid adoption of next-generation sequencing (NGS), whole genome sequencing, and molecular diagnostic tools is significantly improving early and accurate diagnosis of Cockayne syndrome. These technologies enable precise mutation identification in ERCC6 and ERCC8 genes. Improved diagnostic capabilities are expanding the patient identification pool, especially in developed healthcare systems across the U.S., Germany, Japan, and the U.K.
Growth in Gene Therapy and Precision Medicine
The market is benefiting from advancements in gene therapy, viral vector technology, and personalized medicine approaches targeting DNA repair mechanisms. Research institutions and biotech firms are increasingly exploring AAV-based therapies and genomic correction technologies. Growing clinical-stage developments in neurodegenerative and ultra-rare disorders are expected to create long-term therapeutic opportunities.
Supportive Orphan Drug Regulations
Regulatory incentives including orphan drug designation, tax credits, accelerated approvals, and market exclusivity are encouraging pharmaceutical companies to invest in rare disease therapeutics. Regulatory support from the U.S. FDA, EMA, and Japan's PMDA has improved the commercial viability of ultra-rare disease drug development. This is supporting pipeline expansion and clinical research investments.
Rising Healthcare Expenditure and Specialized Care Centers
Increasing healthcare spending and expansion of specialized genetic disease treatment centers are improving access to advanced diagnostics and supportive therapies. Developed economies continue to strengthen rare disease infrastructure through academic collaborations and precision medicine programs. This is particularly evident in the U.S., Canada, Germany, and Japan.
Growing Public and Private Funding Activities
Patient advocacy groups, philanthropic organizations, and biotechnology investors are increasingly funding rare disease therapy programs. Significant funding toward experimental gene therapies and clinical manufacturing capabilities is supporting market expansion. Collaborative funding initiatives are helping accelerate translational research into clinical applications.
Get Detailed Insights Before You Buy - Request Complete Market Intelligence Now. https://www.databridgemarketresearch.com/inquire-before-buying/?dbmr=global-cockayne-syndrome-market
Market Restraints
Limited Patient Population
Cockayne syndrome is an ultra-rare genetic disorder with a very small diagnosed patient pool globally. The low prevalence significantly limits commercial revenue opportunities for pharmaceutical manufacturers. Small patient populations also create difficulties in conducting large-scale clinical trials and achieving economies of scale in therapy development.
High Cost of Gene Therapy Development
The development of gene therapies and advanced rare disease treatments involves substantial research, manufacturing, and regulatory expenses. Viral vector production, clinical testing, and long-term safety monitoring contribute to elevated operational costs. Smaller biotechnology firms often face financial constraints in sustaining long development timelines.
Complex Regulatory Approval Processes
Rare disease therapeutics face stringent regulatory requirements related to clinical efficacy, safety validation, and post-market surveillance. Due to limited patient populations, collecting statistically robust clinical data remains challenging. Regulatory complexity increases time-to-market and raises development risks for manufacturers operating globally.
Lack of Disease Awareness in Emerging Markets
Awareness regarding Cockayne syndrome remains limited in several low- and middle-income countries. Insufficient genetic screening infrastructure and shortage of specialized healthcare professionals contribute to underdiagnosis. Delayed diagnosis negatively affects treatment opportunities and reduces overall market penetration in developing regions.
Limited Availability of Approved Therapies
Currently, treatment options remain largely supportive rather than curative, limiting therapeutic adoption and commercial expansion. The absence of widely approved disease-modifying therapies reduces physician treatment confidence and slows market growth. Research-stage therapies are still years away from large-scale commercialization.
Pricing and Reimbursement Challenges
Advanced therapies for ultra-rare diseases often face reimbursement scrutiny due to extremely high treatment costs. Healthcare payers in Europe, Latin America, and emerging markets frequently impose pricing restrictions and reimbursement limitations. This impacts therapy accessibility and revenue generation potential for market participants.
Market Opportunities
Expansion of Gene Therapy Pipelines
The growing focus on AAV-based therapies and genomic medicine presents significant opportunities for future market expansion. Research organizations and biotechnology firms are increasingly targeting DNA repair disorders using precision gene editing technologies. Successful commercialization of disease-modifying therapies could significantly transform the competitive landscape.
Growth Potential in Asia-Pacific
Asia-Pacific represents a major untapped opportunity due to expanding healthcare infrastructure, rising investments in biotechnology, and improving genetic testing adoption. Countries such as China, India, Japan, and South Korea are increasing funding for rare disease initiatives. The growing middle-class population and healthcare modernization further support long-term market growth.
Strategic Collaborations and Research Partnerships
Partnerships between academic institutions, biotechnology firms, contract manufacturers, and patient advocacy groups are accelerating therapeutic development. Collaborative models help reduce development risks, improve manufacturing capabilities, and enhance clinical trial execution. Strategic alliances are expected to remain a major growth strategy within the market.
Advancements in AI-Driven Genomic Diagnostics
Artificial intelligence and bioinformatics platforms are enhancing mutation detection, disease mapping, and patient stratification processes. AI-enabled genomic analysis can improve early diagnosis accuracy and accelerate personalized treatment planning. These technological advancements are expected to improve clinical outcomes and support market expansion.
Increasing Government Support for Rare Diseases
Several governments are expanding national rare disease frameworks and reimbursement programs. Financial incentives for orphan drug development and research grants are encouraging innovation. Favorable regulatory environments in North America, Europe, and Japan continue to create growth opportunities for pharmaceutical and biotechnology companies.
Emergence of Personalized Treatment Models
Precision medicine approaches tailored to patient-specific genetic mutations are creating new therapeutic opportunities. Personalized therapies improve treatment effectiveness while reducing adverse effects. Advances in molecular medicine and targeted therapeutics are expected to strengthen future market potential.
Market Challenges
Difficulty in Conducting Clinical Trials
Clinical trial recruitment for ultra-rare diseases remains a major challenge due to the extremely limited patient population. Geographical dispersion of patients complicates trial coordination and increases operational costs. This significantly affects therapy development timelines and regulatory approval pathways.
Complex Manufacturing Requirements
Gene therapies and advanced biologics require highly specialized manufacturing environments and stringent quality controls. Viral vector production capacity remains limited globally, creating supply bottlenecks. Manufacturing scalability challenges continue to impact commercialization strategies for rare disease therapies.
Limited Long-Term Clinical Data
Many investigational therapies for Cockayne syndrome remain in early-stage development with limited long-term efficacy and safety data. Regulatory agencies often require extensive monitoring for gene therapies and precision medicine treatments. This uncertainty affects investor confidence and commercial adoption rates.
Healthcare Infrastructure Gaps in Emerging Economies
Several developing regions lack specialized diagnostic laboratories, genomic testing facilities, and trained genetic specialists. Infrastructure limitations delay disease detection and reduce patient access to advanced treatment options. Market growth remains uneven across global regions due to disparities in healthcare systems.
Economic Pressure on Healthcare Systems
High therapy prices and rising healthcare expenditure are increasing pressure on public reimbursement systems. Budget limitations in Europe and emerging markets are influencing pricing negotiations and market access strategies. Cost-effectiveness evaluations continue to challenge widespread adoption of advanced therapies.
Intense Competition in Rare Disease Therapeutics
The broader rare disease market is becoming increasingly competitive due to rising investment in orphan drug development. Larger pharmaceutical companies with stronger financial capabilities are intensifying competitive pressure on smaller biotechnology firms. This increases barriers for market entry and commercialization success.
Market Segmentation & Analysis
By Type
Diagnostic Testing
Diagnostic testing represents the largest segment due to increasing adoption of genomic sequencing and molecular diagnostics for rare genetic disorders. The segment benefits from rising awareness of inherited neurological diseases and expanding newborn screening initiatives. Hospitals and specialized laboratories increasingly use NGS-based testing to identify ERCC6 and ERCC8 mutations. The segment accounted for the highest revenue share in 2025 and is expected to maintain dominance during the forecast period.
Supportive Therapeutics
Supportive therapeutics include symptomatic treatments such as nutritional support, neurological care, physiotherapy, and ophthalmic management. This segment remains essential due to the absence of widely approved curative therapies. Demand is supported by growing patient management programs and multidisciplinary treatment approaches across specialty healthcare centers.
Gene Therapy
Gene therapy is projected to register the fastest CAGR through 2032 owing to increasing research into DNA repair mechanisms and AAV-based therapeutic approaches. Advancements in viral vector manufacturing and precision medicine technologies are supporting segment growth. Research collaborations and clinical-stage developments are expected to accelerate commercialization potential.
By Application
Neurological Disorder Management
Neurological disorder management dominates the market due to the severe neurodegenerative impact of Cockayne syndrome. This segment includes therapies and interventions targeting developmental delays, hearing loss, and motor dysfunction. Increased clinical focus on neuroprotection and disease progression monitoring continues to support segment expansion.
Genetic Research
Genetic research is a rapidly growing segment supported by increasing investments in DNA repair studies and rare disease genomics. Research institutions and biotechnology firms are actively exploring gene-editing technologies and molecular treatment pathways. This segment is expected to witness strong growth due to expanding translational research activities.
Pediatric Rare Disease Care
Pediatric care applications remain critical because Cockayne syndrome primarily affects children. Specialized pediatric hospitals and genetic disorder centers are increasing investment in integrated treatment models. Rising awareness among pediatric neurologists and genetic specialists supports segment growth globally.
By End User
Specialty Hospitals
Specialty hospitals hold the largest market share due to access to advanced diagnostic infrastructure and multidisciplinary treatment capabilities. These facilities offer specialized neurological, genetic, and pediatric care services. North America and Europe continue to dominate this segment owing to strong healthcare infrastructure.
Research Institutes
Research institutes are expected to witness the fastest growth due to increasing funding for rare disease studies and gene therapy development. Academic collaborations and biotechnology partnerships are accelerating innovation in precision medicine. Government-supported genomic research programs further support segment expansion.
Diagnostic Laboratories
Diagnostic laboratories play a critical role in molecular testing and genomic sequencing services. Rising demand for genetic confirmation testing and improved laboratory automation technologies are supporting segment growth. Expansion of private genomic testing services is further strengthening market demand.
By Technology
Next-Generation Sequencing (NGS)
NGS technology dominates the market due to its high accuracy and ability to detect rare genetic mutations efficiently. The technology is increasingly used in clinical diagnostics and research applications. Falling sequencing costs and broader adoption across hospitals and laboratories continue to support growth.
PCR-Based Diagnostics
PCR-based diagnostics remain widely utilized for mutation analysis and genetic confirmation testing. These systems provide cost-effective and rapid testing solutions, particularly in emerging economies. Continued use in routine diagnostic workflows supports segment stability.
Gene Editing Technologies
Gene editing technologies including CRISPR-based platforms are expected to witness the highest CAGR during the forecast period. Increasing investment in precision medicine and DNA repair therapies is accelerating research activities. The technology holds strong future commercialization potential for rare genetic disorders.
Regional Analysis
By geography, the market is categorized into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
North America
North America dominates the global Cockayne syndrome market with approximately 41% revenue share in 2025. The region benefits from advanced healthcare infrastructure, strong orphan drug regulations, and significant investments in genomic medicine. The U.S. remains the largest contributor due to the presence of leading biotechnology companies, specialized research institutes, and high adoption of genetic testing technologies. Strong FDA support for rare disease clinical trials and precision medicine continues to accelerate market growth.
Europe
Europe represents a mature and steadily growing market supported by strong R&D investments and regulatory frameworks for orphan drugs. Countries including Germany, the U.K., and France are leading contributors due to established healthcare systems and increasing genomic research activities. The region also benefits from active collaboration between academic institutions and biotechnology firms. Growing awareness regarding rare pediatric neurological disorders continues to support market expansion.
Asia Pacific
Asia-Pacific is projected to register the fastest CAGR during the forecast period. Rapid healthcare modernization, increasing government support for rare disease management, and growing biotechnology investments are driving regional growth. China, Japan, and India are key contributors due to expanding genetic testing adoption and rising healthcare expenditure. Increasing availability of specialized diagnostics and precision medicine technologies is further supporting market penetration.
Latin America
Latin America represents an emerging market with gradual adoption of rare disease diagnostics and treatment solutions. Brazil and Mexico remain the primary contributors due to improving healthcare infrastructure and increasing access to specialized care centers. Economic limitations and restricted reimbursement systems continue to impact broader market growth. However, expanding awareness initiatives and public healthcare investments are improving long-term prospects.
Middle East & Africa
The Middle East & Africa market is experiencing steady but slower growth compared to developed regions. Government investments in healthcare modernization and increasing private sector participation are supporting market development. However, limited genetic testing infrastructure and shortage of specialized rare disease treatment centers remain major constraints. Gulf countries are expected to witness relatively stronger growth due to improving healthcare capabilities.
Key Insights
Largest Region: North America
Fastest Growing Region: Asia-Pacific
Mature markets including North America and Europe benefit from advanced healthcare infrastructure, established reimbursement systems, and strong biotechnology ecosystems. Asia-Pacific represents a high-growth market driven by industrialization, healthcare investments, and increasing precision medicine adoption. Latin America and Middle East & Africa remain emerging regions with improving healthcare access but ongoing infrastructure limitations. Supply chain capabilities, regulatory frameworks, and local manufacturing ecosystems continue to influence regional competitiveness.
Competitive Landscape
Market Structure Overview
The global Cockayne syndrome market is moderately consolidated with the presence of specialized biotechnology firms, genomic diagnostic companies, research institutions, and emerging rare disease therapy developers. Competition is largely driven by innovation in gene therapy, molecular diagnostics, and precision medicine technologies. Market participants are increasingly focusing on strategic collaborations, orphan drug development, and clinical research initiatives. Competitive landscape analysis helps stakeholders evaluate market positioning, innovation capabilities, and long-term strategic direction.
Key Industry Players
Leading companies operating in the market focus on rare disease therapeutics, genetic diagnostics, and precision medicine solutions. Major players are investing heavily in R&D activities, advanced genomic technologies, and strategic partnerships to strengthen market presence. Companies with strong orphan drug portfolios and global research capabilities maintain competitive advantages. Innovation intensity and pipeline development remain critical differentiating factors within the market.
List of Key Industry Players
F. Hoffmann-La Roche Ltd.
Thermo Fisher Scientific Inc.
Illumina Inc.
Pfizer Inc.
Novartis AG
Andelyn Biosciences
Quest Diagnostics Incorporated
Eurofins Scientific
BioMarin Pharmaceutical Inc.
Ultragenyx Pharmaceutical Inc.
Competitive Strategies
Companies are increasingly adopting strategies including product innovation, gene therapy research expansion, strategic acquisitions, and partnerships with academic institutions. Investment in advanced sequencing technologies and personalized medicine platforms remains a key competitive focus. Several firms are strengthening global distribution capabilities and expanding orphan disease portfolios. Strategic collaborations with patient advocacy groups and contract manufacturing organizations are also accelerating clinical development activities.
Emerging Players & Market Dynamics
Emerging biotechnology startups are increasingly entering the market with specialized gene editing and rare disease therapy platforms. These companies focus on targeted, cost-effective, and precision-based therapeutic solutions that challenge traditional treatment approaches. Rising venture capital investments and funding activities are accelerating innovation in ultra-rare disease treatment development. Increasing adoption of digital genomics, AI-enabled diagnostics, and advanced analytics is reshaping competitive dynamics globally.
Latest Developments
July 2024 - Andelyn Biosciences: The company partnered with UMass Chan Medical School to manufacture clinical-grade AAV9-CSA vectors for Cockayne syndrome gene therapy development. The collaboration supports future clinical trials and strengthens advancement in precision gene therapy manufacturing capabilities.
June 2024 - UMass Chan Medical School: UMass Chan received USD 2.2 million in funding from the Riaan Research Initiative to accelerate development of gene therapy for Cockayne syndrome. The funding supports toxicology studies and GMP manufacturing preparation for future human trials.
May 2024 - Sentynl Therapeutics: Sentynl Therapeutics acquired global rights to Zokinvy from Eiger BioPharmaceuticals, strengthening its rare and ultra-rare disease portfolio. The acquisition reflects increasing pharmaceutical interest in orphan genetic disease therapeutics.
August 2024 - Regulatory Advancements in Rare Disease Therapeutics: Increased regulatory support for rare disease drug development across the U.S., Europe, and Japan accelerated clinical-stage investment activity in genomic medicine and orphan therapeutics. This trend is improving commercialization prospects for ultra-rare disease treatments.
2024 - Expansion of AI-Driven Genomic Diagnostics: Multiple genomic diagnostics companies expanded AI-enabled mutation analysis platforms to improve rare disease detection accuracy and speed. These developments are improving early diagnosis capabilities and supporting precision medicine adoption globally.
2024 - Growth in Rare Disease Research Collaborations: Academic institutions and biotechnology firms increased cross-border research partnerships focused on DNA repair disorders and neurodegenerative rare diseases. Collaborative models are improving access to specialized expertise, funding, and advanced therapeutic technologies.
2023-2024 - Increased Orphan Drug Funding Initiatives: Governments and patient advocacy organizations across North America and Europe expanded funding initiatives for orphan disease research. Increased grants and incentives are accelerating innovation in gene therapy and personalized medicine.
Check out more related studies published by Data Bridge Market Research:
https://www.databridgemarketresearch.com/reports/global-gout-therapeutics-market
https://www.databridgemarketresearch.com/reports/global-interstitial-cystitis-market
https://www.databridgemarketresearch.com/reports/global-mek-inhibitors-market
https://www.databridgemarketresearch.com/reports/global-phenylpropanolamine-ppa-market
https://www.databridgemarketresearch.com/reports/global-selective-estrogen-receptor-degraders-serd-therapeutics-market
https://www.databridgemarketresearch.com/reports/global-shingles-treatment-market
https://www.databridgemarketresearch.com/reports/global-venous-thromboembolism-market
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email:- corporatesales@databridgemarketresearch.com
Data Bridge Market Research is a market research and consulting company that educates its clients about the market and encourages growth and expansion. We offer customized reports, syndicated research, consulting services, cloud-connected intelligence, and a holistic suite of offerings including competitive intelligence, epidemiology analyses, trade analytics, country analysis, and pharma insights.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Cockayne Syndrome Market to Grow at a CAGR of 4.30% During 2026-2032 here
News-ID: 4514816 • Views: …
More Releases from Data Bridge Market Research
Commercial Aircraft Wings Market Size, Share & Forecast to 2032 - Industry Growt …
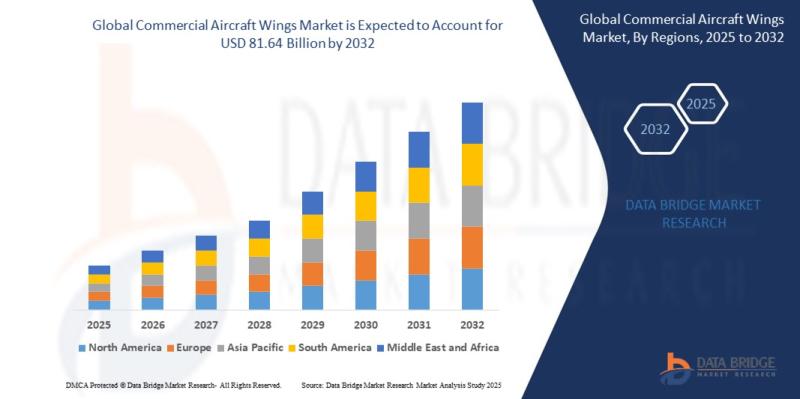
As per Data Bridge Market Research analysis, the Commercial Aircraft Wings Market was estimated at USD 49.72 billion in 2025. The market is expected to grow from USD 46.32 billion in 2024 to USD 81.64 billion by 2032, at a CAGR of 7.34% during the forecast period with driven by the rising demand for fuel-efficient aircraft, increasing air passenger traffic, and advancements in lightweight composite wing technologies.
Growth is supported by…
Embedded Analytics Market Size, Share & Trends Analysis Report - Industry Outloo …
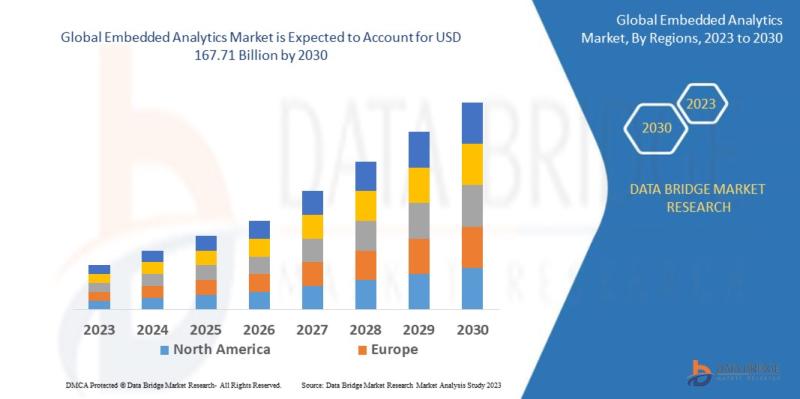
As per Data Bridge Market Research analysis, the Embedded Analytics Market was estimated at USD 84.40 billion in 2025. The market is expected to grow from USD 96.80 billion in 2026 to USD 167.71 billion in 2030, at a CAGR of 14.70% during the forecast period with driven by the rising demand for real-time business intelligence, cloud-based analytics integration, AI-enabled decision-making platforms, and increasing enterprise digital transformation initiatives.
Get the full…
Corporate Wellness Market Size, Share & Industry Analysis, By Service Type, Deli …
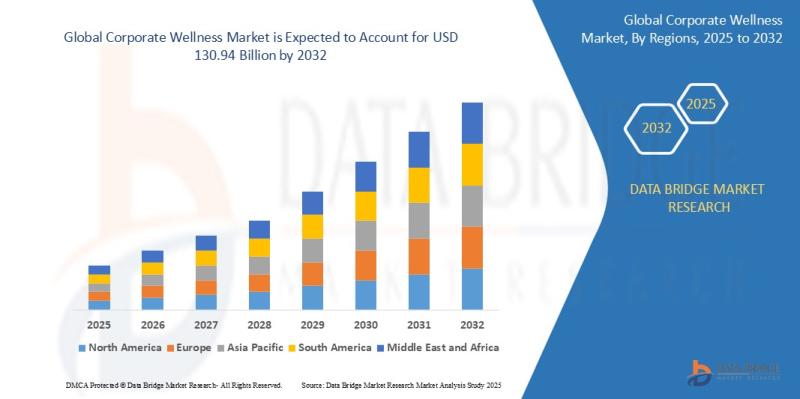
"As per Data Bridge Market Research analysis, the Corporate Wellness Market was estimated at USD 81.54 billion in 2025. The market is expected to grow from USD 87.25 billion in 2026 to USD 130.94 billion in 2032, at a CAGR of 7.00% during the forecast period with driven by the rising demand for employee mental health programs, digital wellness platforms, preventive healthcare initiatives, and increasing employer focus on productivity optimization."
Get…
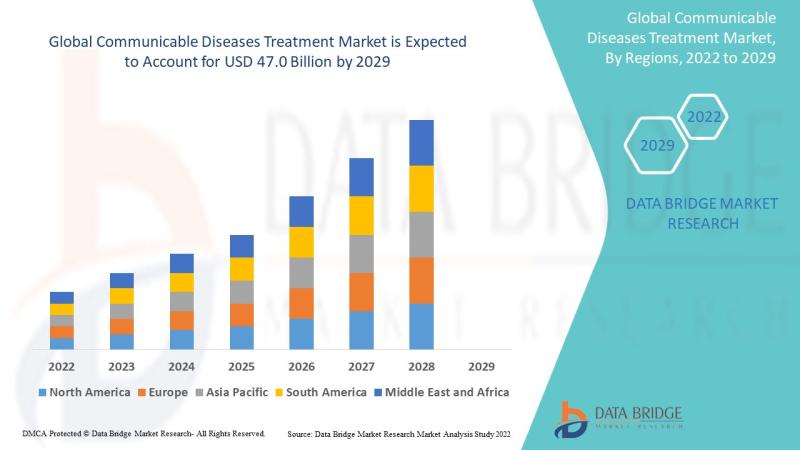
Communicable Diseases Treatment Market to Reach USD 56.99 billion by 2032, Growi …
As per Data Bridge Market Research analysis, the Communicable Diseases Treatment Market was estimated at USD 36.34 billion in 2025. The market is expected to grow from USD 34.08 billion in 2024 to USD 56.99 billion by 2032, at a CAGR of 6.64% during the forecast period with driven by the rising demand for advanced anti-infective therapies, increasing prevalence of infectious diseases, and expanding global healthcare infrastructure.
Growth is supported by…
More Releases for Cockayne
Cockayne Syndrome Market Will Grow at a CAGR of 4.30% by 2028
The widespread Cockayne Syndrome report has been built with the appropriate expertises that utilize established and unswerving tools and techniques such as SWOT analysis and Porter's Five Forces analysis to conduct the research study. To attain knowhow of market landscape, brand awareness, latest trends, possible future issues, industry trends and customer behaviour, this premium market research report is very crucial. The report also identifies and analyses the intensifying trends along…
Intra-Abdominal Pressure Measurement Devices Market Would Rocket Up to CAGR of 2 …
Global Intra-Abdominal Pressure Measurement Devices market research report helps to identify trends in consumer and supply chain dynamics and accordingly interpret marketing, promotional, and sales strategies for business growth and an utmost success. The report aids in developing a successful marketing strategy for the business. This industry analysis report has been prepared based on detailed market analysis with inputs from industry experts. It includes historic data, present market trends, environment,…
Biochips Market Will Grow at a rate of 14.22% by 2028
To uncover the general market conditions and tendencies, Biochips market research report acts as a perfect source. This report helps in planning by providing precise and state-of-the-art information about the consumer's demands, preferences, attitudes and their changing tastes about the specific product. This report is a window to the Healthcare industry which explains what market definition, classifications, applications, engagements and market trends are. The numerical data of this report is…
Chromatography Columns Market is Going to Reach USD 18,441.09 million by 2029
Chromatography Columns market research report directs the manufacturer about planning of advertising and sales promotion efforts and makes it more effective. This report has been prepared by considering various steps for collecting, recording and analysing market data. This market analysis report covers strategic profiling of key players in the market, comprehensively analyzing their core competencies, and drawing a competitive landscape for the market. It also delivers list of the leading…
Cockayne Syndrome Market Will Grow at a CAGR of 4.30% by 2028
The widespread Cockayne Syndrome report has been built with the appropriate expertises that utilize established and unswerving tools and techniques such as SWOT analysis and Porter's Five Forces analysis to conduct the research study. To attain knowhow of market landscape, brand awareness, latest trends, possible future issues, industry trends and customer behaviour, this premium market research report is very crucial. The report also identifies and analyses the intensifying trends along…
Cockayne Syndrome Market Size, Industry Outlook and Forecast to 2028
The report “Global Cockayne Syndrome Market Growth 2021-2028” from Databridge Market Research includes data and information about market structure and size. The purpose of this research is to give market knowledge and strategic insights to assist in decision making, making informed investment decisions, and identifying potential growth opportunities.
Download Free Sample Report: To Know The Impact Of COVID-19 On This Industry @ https://www.databridgemarketresearch.com/request-a-sample/?dbmr=global-cockayne-syndrome-market
The Global Cockayne Syndrome Market market report also indicates…