Press release
Global Contract Pharmaceutical Fermentation Services Market Expands Across North America, Europe & Asia Pacific Lonza, TF
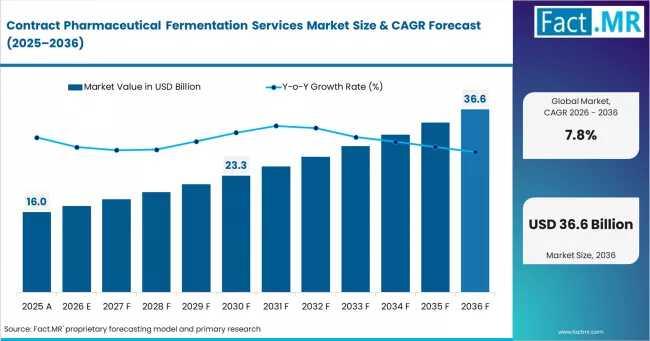
According to Fact.MR estimates, the global contract pharmaceutical fermentation services Market was valued at USD 16.0 billion in 2025. The market is projected to reach USD 17.2 billion in 2026 and is expected to grow to USD 36.6 billion by 2036, expanding at a CAGR of 7.8%. Commercial GMP is anticipated to account for 52.0% of the product segment in 2026, while Recombinant Proteins is expected to remain the leading application with around 44.0% share.Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=1517
The global contract pharmaceutical fermentation services market is entering a decisive growth phase as biologic drug development accelerates faster than available large-scale manufacturing infrastructure. Pharmaceutical companies are increasingly outsourcing fermentation-based production to specialized contract development and manufacturing organizations (CDMOs) to avoid the rising capital burden and technical complexity associated with biologics manufacturing. What was once a tactical outsourcing decision is now becoming a long-term strategic manufacturing model for biopharma companies seeking speed, flexibility, regulatory scalability, and global market access.
The market's transformation is being driven by the rapid expansion of recombinant protein therapeutics, vaccines, plasmid DNA production, mRNA technologies, and gene therapy vectors. At the same time, pressure on biologic pricing, particularly in biosimilars, is pushing sponsors toward more efficient continuous fermentation systems and highly optimized manufacturing partnerships. Industry participants increasingly view fermentation capacity as a strategic asset rather than a routine operational requirement.
Quick Stats: Contract Pharmaceutical Fermentation Services Market
• Global market value in 2025: USD 16 billion
• Estimated market value in 2026: USD 17.25 billion
• Forecast market value by 2036: USD 36.55 billion
• Forecast CAGR (2026-2036): 7.8%
• Absolute dollar opportunity: USD 19.3 billion
• Commercial GMP segment share in 2026: 52%
• Recombinant proteins segment share in 2026: 44%
• Fastest-growing countries: USA (8.6% CAGR), Mexico (8.2% CAGR)
• Core technologies: Microbial, mammalian, and yeast-based fermentation
• Key applications: Recombinant proteins, vaccines, plasmid DNA, gene therapy vectors, and RNA-based therapeutics
Market Size and Forecast Signal Long-Term Outsourcing Expansion
The contract pharmaceutical fermentation services market is projected to more than double over the next decade, reaching USD 36.55 billion by 2036 from USD 17.25 billion in 2026. This sustained expansion reflects structural changes in the pharmaceutical manufacturing ecosystem rather than temporary outsourcing cycles.
Biopharmaceutical companies are increasingly prioritizing asset-light operating models. Building fermentation facilities internally requires major investments in bioreactors, cleanrooms, process automation systems, validation infrastructure, and regulatory compliance capabilities. In addition to capital intensity, many developers lack the specialized technical expertise needed to manage advanced microbial or mammalian expression systems at commercial scale.
As biologics pipelines diversify, outsourcing is becoming critical not only for manufacturing execution but also for process optimization, analytical validation, technology transfer, and regulatory readiness. CDMOs capable of supporting clinical development through commercial launch are therefore becoming central strategic partners across the pharmaceutical value chain.
The market's projected CAGR of 7.8% reflects growing demand across both established biologic therapeutics and next-generation modalities such as cell and gene therapies, RNA therapeutics, and plasmid-based platforms.
Commercial GMP Manufacturing Emerges as the Revenue Core Commercial GMP manufacturing is expected to account for approximately 52% of the scale segment in 2026, underscoring the increasing maturity of outsourced biologics production.
Historically, many pharmaceutical companies outsourced only early-stage or clinical manufacturing. That dynamic has shifted significantly. Today, large pharmaceutical firms and emerging biotech companies alike are entering long-duration commercial manufacturing agreements with CDMOs to secure supply continuity and reduce operational risk.
Commercial-scale fermentation facilities require high-capacity stainless-steel bioreactors, contamination control systems, validated analytics, and strict compliance with international GMP standards. Because facility construction timelines can exceed four to six years, current demand is beginning to outpace available global supply.
This imbalance is strengthening pricing leverage for CDMOs with immediately available GMP capacity. Manufacturers capable of supporting both microbial and mammalian systems are especially well positioned because sponsors increasingly seek platform flexibility that can support multiple product modalities within a single infrastructure footprint.
Recombinant Proteins Continue to Dominate Demand Recombinant proteins are projected to account for approximately 44% of the modality segment in 2026, maintaining their position as the largest demand category within fermentation services.
Therapeutic proteins remain foundational to modern biologic medicine across oncology, immunology, endocrinology, and rare disease treatment. Continued demand for monoclonal antibodies, enzymes, hormones, cytokines, and biosimilar products is sustaining large-scale fermentation requirements globally.
The recombinant protein segment also benefits from well-established regulatory pathways and institutional procurement systems, particularly in mature healthcare markets. As healthcare systems continue expanding access to biologic therapies, manufacturing demand for recombinant products is expected to remain resilient throughout the forecast period.
At the same time, the rise of biosimilars is intensifying cost pressures. Pharmaceutical companies are therefore prioritizing manufacturing efficiency, yield optimization, and lower cost-per-batch economics. This trend is accelerating adoption of advanced fermentation technologies, including continuous manufacturing platforms.
Continuous Manufacturing Is Reshaping CDMO Competition
One of the most important technology trends shaping the market is the shift from traditional batch fermentation toward continuous manufacturing systems.
Continuous fermentation platforms improve process efficiency by maintaining uninterrupted production flows, reducing downtime, minimizing contamination risks, and improving consistency across production runs. These systems also reduce facility footprint requirements and improve long-term manufacturing economics.
The technology is gaining traction particularly in high-volume recombinant protein and biosimilar manufacturing where pricing competitiveness is essential. Sponsors increasingly evaluate CDMOs not only on available capacity but also on process sophistication and technology adaptability.
This shift is redefining competitive positioning within the CDMO sector. Companies investing in flexible continuous manufacturing systems are increasingly securing higher-value contracts with longer durations and broader strategic partnerships.
The industry is moving away from simple capacity outsourcing toward capability-driven collaboration models where manufacturing innovation directly affects commercial viability.
Gene Therapy and RNA Modalities Create New Fermentation Demand
The emergence of gene therapies, mRNA therapeutics, and plasmid DNA applications is expanding the functional role of fermentation services beyond conventional biologics manufacturing.
Plasmid DNA production, for example, relies heavily on microbial fermentation processes and is becoming increasingly important for gene editing platforms, viral vector manufacturing, and nucleic acid therapeutics.
Similarly, mRNA manufacturing workflows often require fermentation-derived raw materials and intermediates. As clinical pipelines for RNA-based therapeutics expand, CDMOs with integrated fermentation and analytical capabilities are expected to capture disproportionate growth opportunities.
These next-generation modalities require highly specialized process controls, contamination management systems, and analytical testing infrastructure. The technical barriers associated with these products are creating strong competitive differentiation opportunities for experienced biologics CDMOs.
Regional Analysis: North America Leads While Asia Pacific Accelerates
North America continues to maintain a dominant market position due to its mature pharmaceutical ecosystem, established biologics manufacturing infrastructure, and strong regulatory frameworks.
The United States is projected to grow at a CAGR of 8.6% through 2036, outperforming many mature pharmaceutical markets. Demand growth is supported by ongoing biologics innovation, strong institutional investment, and expanding commercial manufacturing requirements.
The U.S. market also benefits from concentration of biotech innovation hubs, favorable venture funding environments, and increasing FDA familiarity with advanced biologic modalities.
Europe remains a stable and strategically important market supported by robust clinical standards, harmonized regulatory systems, and advanced healthcare procurement networks. Germany, France, and the United Kingdom continue to represent core biologics manufacturing centers with strong demand for high-quality GMP fermentation services.
• Germany forecast CAGR: 7.8%
• France forecast CAGR: 7.7%
• UK forecast CAGR: 7.5%
Asia Pacific is emerging as the fastest-evolving regional landscape due to infrastructure expansion, manufacturing cost advantages, and rising domestic biologics demand. Although the dataset highlights Mexico at 8.2% CAGR, broader emerging-market expansion is also strengthening global supply diversification strategies.
Many multinational pharmaceutical firms are increasingly seeking geographically diversified manufacturing footprints to reduce supply-chain concentration risk and improve resilience following disruptions experienced during recent global health emergencies.
Market Constraints Continue to Challenge Expansion
Despite strong growth fundamentals, the market faces several operational and strategic constraints.
Capacity Limitations
Large-scale GMP fermentation infrastructure remains expensive and time-intensive to build. The industry currently faces a shortage of immediately available commercial manufacturing capacity, particularly for mammalian fermentation systems.
Regulatory Complexity
Global regulatory differences continue to create operational burdens for multinational CDMOs. Companies serving multiple jurisdictions must maintain compliance across varying quality, validation, and documentation frameworks.
Cost Sensitivity
Price pressure in healthcare procurement, particularly in biosimilars and emerging-market biologics, is limiting pricing flexibility for some manufacturers despite strong demand.
Talent Shortages
The industry also faces a shortage of highly skilled biologics manufacturing professionals with expertise in fermentation engineering, process development, GMP operations, and analytical validation.
Competitive Landscape Reflects Strategic Consolidation
The competitive environment remains moderately fragmented, although scale advantages are becoming increasingly important.
Leading companies are strengthening their positions through facility expansion, technology investments, geographic diversification, and integrated service models.
Key industry participants include:
• Lonza Group AG
• Thermo Fisher Scientific Inc.
• Fujifilm Diosynth Biotechnologies
• Samsung Biologics Co., Ltd.
• Catalent, Inc.
• Boehringer Ingelheim GmbH
• WuXi Biologics
• AGC Biologics
• Novasep Holding SAS
• Rentschler Biopharma SE
Recent developments highlight growing competitive intensity:
• In January 2026, Lonza expanded its portfolio to target rising demand segments.
• Thermo Fisher Scientific broadened geographic reach through distribution partnerships in 2025.
• Fujifilm Diosynth introduced technology upgrades focused on performance optimization and regulatory compliance.
Competition increasingly centers on manufacturing flexibility, technology sophistication, regulatory reliability, and speed-to-commercialization rather than simply production volume.
Strategic Implications for Pharmaceutical Executives
For pharmaceutical companies, fermentation outsourcing is evolving into a board-level strategic consideration rather than a procurement decision.
Several key implications are emerging:
Long-Term Capacity Agreements Are Becoming Essential
As commercial GMP capacity tightens, pharmaceutical firms are increasingly securing multi-year manufacturing partnerships earlier in product development cycles.
Manufacturing Technology Influences Product Economics
Continuous manufacturing and advanced fermentation systems are directly affecting biologic pricing competitiveness, particularly in biosimilars.
Flexible Infrastructure Has Strategic Value
CDMOs capable of supporting microbial, mammalian, plasmid, and RNA workflows within a unified platform are gaining strategic relevance.
Supply-Chain Diversification Is a Priority
Global pharmaceutical companies are reducing geographic concentration risks by diversifying manufacturing networks across multiple regions.
Future Outlook: From Outsourcing Vendor to Strategic Manufacturing Partner
The next decade is expected to redefine the relationship between pharmaceutical companies and fermentation service providers.
CDMOs are moving beyond transactional manufacturing roles toward integrated innovation partnerships that encompass process design, scalability optimization, analytical services, regulatory support, and commercial supply management.
As biologics continue expanding across therapeutic categories, fermentation manufacturing capacity will become increasingly central to pharmaceutical commercialization strategies. Companies capable of combining flexible manufacturing technologies, regulatory expertise, and multi-modality capabilities are expected to capture the strongest long-term growth opportunities.
The industry's future growth trajectory will likely depend on how rapidly new commercial-scale capacity can be deployed, how effectively continuous manufacturing technologies mature, and how successfully CDMOs adapt to emerging biologic modalities.
Executive-Level Takeaways
• The contract pharmaceutical fermentation services market is projected to reach USD 36.55 billion by 2036, driven by sustained biologics expansion.
• Commercial GMP manufacturing remains the dominant revenue engine, accounting for 52% of scale share in 2026.
• Recombinant proteins continue leading modality demand with 44% share, while gene therapy and RNA applications create new growth avenues.
• Continuous fermentation manufacturing is emerging as a critical competitive differentiator among CDMOs.
• Capacity shortages are strengthening pricing power for providers with available large-scale GMP infrastructure.
• North America remains the leading regional market, while emerging manufacturing regions continue gaining strategic importance.
• Pharmaceutical companies are increasingly prioritizing long-term, technology-driven manufacturing partnerships rather than short-term outsourcing arrangements.
Unlock 360° insights for strategic decision making and investment planning: https://www.factmr.com/checkout/1517
Browse Full Report : https://www.factmr.com/report/1517/contract-pharmaceutical-fermentation-services-market
To View Related Report:
Antibody Contract Manufacturing Market https://www.factmr.com/report/antibody-contract-manufacturing-market
Neurology Contract Research Organization Market https://www.factmr.com/report/neurology-contract-research-organization-market
Biologics Contract Manufacturing Market https://www.factmr.com/report/biologics-contract-manufacturing-market
Antibodies Contract Manufacturing Market https://www.factmr.com/report/antibodies-contract-manufacturing-market
- Contact Us -
11140 Rockville Pike, Suite 400, Rockville,
MD 20852, United States
Tel: +1 (628) 251-1583 | sales@factmr.com
About Fact.MR
Fact.MR is a global market research and consulting firm, trusted by Fortune 500 companies and emerging businesses for reliable insights and strategic intelligence. With a presence across the U.S., UK, India, and Dubai, we deliver data-driven research and tailored consulting solutions across 30+ industries and 1,000+ markets. Backed by deep expertise and advanced analytics, Fact.MR helps organizations uncover opportunities, reduce risks, and make informed decisions for sustainable growth.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Contract Pharmaceutical Fermentation Services Market Expands Across North America, Europe & Asia Pacific Lonza, TF here
News-ID: 4511859 • Views: …
More Releases from Fact.MR
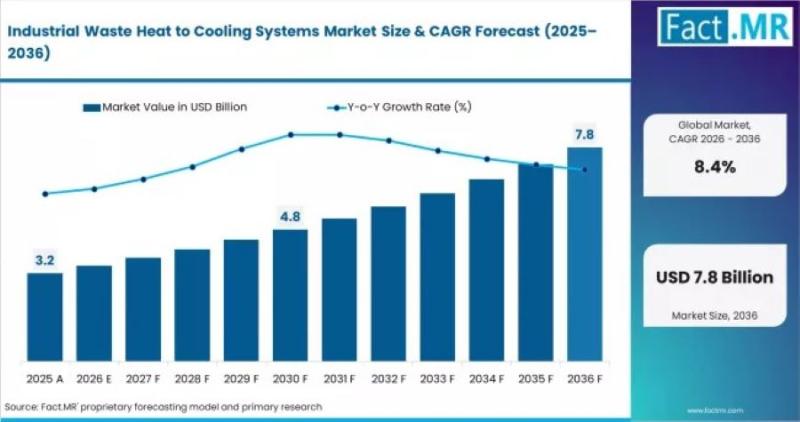
Industrial Waste Heat to Cooling Systems Gain Momentum as Manufacturers Push Ene …
Industrial manufacturers are increasingly turning to waste heat recovery technologies to reduce energy costs, improve operational efficiency, and support decarbonization goals. Among the fastest-growing solutions are industrial waste heat to cooling systems, which convert excess thermal energy from industrial processes into refrigeration or chilled water for process cooling and HVAC applications.
Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=14830
Quick Stats
• 2026 market size: USD 3.2 billion
• 2036 forecast value: USD…

Warehouse Digital Twin Orchestration Software Reshapes Modern Fulfillment Operat …
Warehouse operators are rapidly moving beyond traditional warehouse management systems toward digital orchestration platforms capable of simulating and optimizing logistics operations in real time. As e-commerce fulfillment volumes rise and automation becomes more complex, digital twin orchestration software is emerging as a critical technology layer for supply chain efficiency, predictive planning, and warehouse synchronization.
Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=14808
Quick Stats
• 2026 market size: USD 1.0 billion
• 2036…
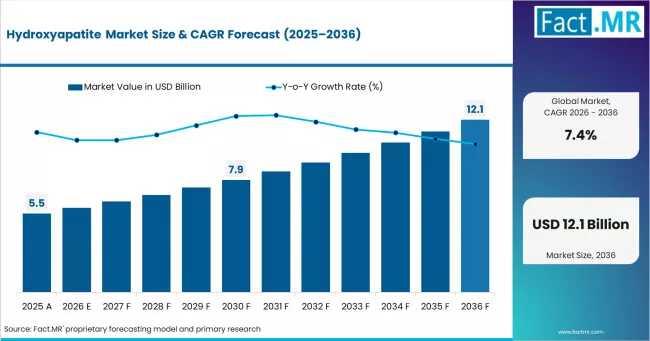
Global Hydroxyapatite Market Outlook 2025-2035: Asia Pacific Leads Growth; APS M …
According to Fact.MR analysis, the global hydroxyapatite market is projected to rise from approximately USD 5.9 billion in 2026 to USD 12.1 billion by 2036, expanding at a CAGR of 7.4% during the forecast period. Growth is being supported by rising orthopedic surgery volumes, rapid dental implant adoption, advances in nanotechnology processing, and growing demand for bioactive coatings that improve osseointegration and implant longevity.
The global hydroxyapatite market is entering a…
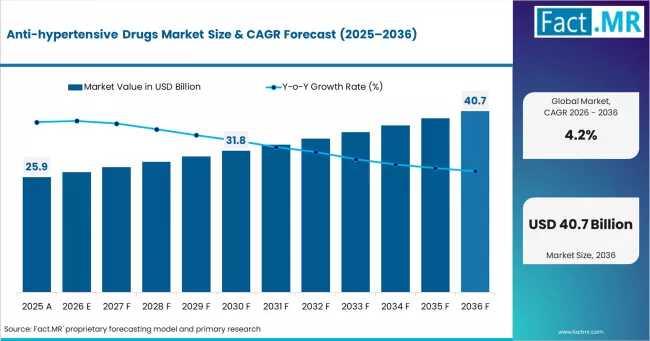
Global Anti-Hypertensive Drugs Market Outlook 2035: Novartis, Sanofi & Bayer Dri …
According to Fact MR's latest analysis, Anti-hypertensive Drugs Market Eyes Long-Term Expansion as Combination Therapies and Emerging Market Screening Programs Reshape Global Demand The global Cardiovascular Pharmacology landscape is entering a new phase of structural growth as healthcare systems intensify efforts to diagnose and manage hypertension earlier. According to Fact.MR analysis, the global anti-hypertensive drugs market is projected to rise from approximately USD 27.0 billion in 2026 to nearly…
More Releases for CDMO
FDP CDMO Research: China FDP CDMO market size is projected to reach USD 1.33 bil …
QY Research Inc. (Global Market Report Research Publisher) announces the release of 2025 latest report "Fraud Detection and Prevention (FDP) System- Global Market Share and Ranking, Overall Sales and Demand Forecast 2025-2031". Based on current situation and impact historical analysis (2020-2024) and forecast calculations (2025-2031), this report provides a comprehensive analysis of the global Wire Drawing Dies market, including market size, share, demand, industry development status, and forecasts for the…
Global Cmo And Cdmo Biotechnology Market Size by Application, Type, and Geograph …
According to Market Research Intellect, the global Cmo And Cdmo Biotechnology market under the Internet, Communication and Technology category is expected to register notable growth from 2025 to 2032. Key drivers such as advancing technologies, changing consumer behavior, and evolving market dynamics are poised to shape the trajectory of this market throughout the forecast period.
Biologics and sophisticated medicines are driving the biotechnology industry for Contract Manufacturing Organizations (CMO) and Contract…
Evolving Market Trends In The Inhalation CDMO Industry: Strategic Collaborations …
The Inhalation CDMO Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].
What Is the Expected Inhalation CDMO Market Size During the Forecast Period?
In recent times, the inhalation CDMO market has experienced significant growth. The market value is expected to increase from $2.08 billion in 2024…
What's Driving the Inhalation CDMO Market 2025-2034: Rising Respiratory Disorder …
How Is the Chondroplasty Market Projected to Grow, and What Is Its Market Size?
The chondroplasty market has seen strong growth in recent years. It will increase from $13.77 billion in 2024 to $14.68 billion in 2025 at a CAGR of 6.5%. This growth is attributed to the rise in sports-related injuries, patient preference for non-total joint replacement procedures, advances in postoperative care, healthcare provider training, and an increasing incidence of…
Lentiviral Vector (LVV) CDMO Services Market Delivering Cures: The Role of LVV C …
Lentiviral Vector (LVV) CDMO Services Market to Record an Exponential CAGR by 2031 - Exclusive Report by InsightAce Analytic Pvt. Ltd.
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Lentiviral Vector (LVV) CDMO Services Market - (By Type (IIT Grade, IND Grade, Clinical Trial Grade, Commercial Production Grade), By Application (Biopharmaceutical Company, Academic Scientific Research Institution)), Trends, Industry Competition Analysis, Revenue and Forecast To…
Electronic Chemicals CDMO Market Fueling the Electronics Boom: The Rise of the E …
Electronic Chemicals CDMO Market to Record an Exponential CAGR by 2031 - Exclusive Report by InsightAce Analytic Pvt. Ltd.
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Electronic Chemicals CDMO Market - (By Type (Metals and Pastes, Electronic Specialty Gases, Polymer Compounds, Others), By Application (Battery, Semiconductor, Integrated Circuit, Consumer Electronics, Others)), Trends, Industry Competition Analysis, Revenue and Forecast To 2031."
According to the latest…