Press release
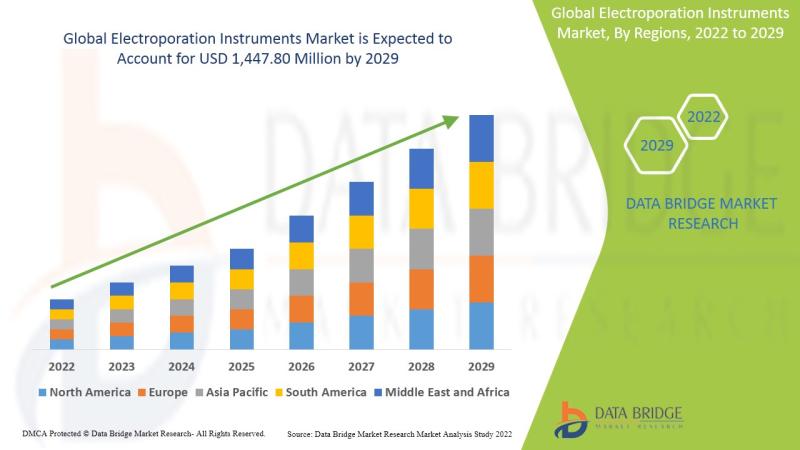
Electroporation Instruments Market Outlook 2026-2032: Market to Reach USD 1.84 Billion by 2032 Amid Rising Adoption of Gene and Cell Therapy Technologies
As per Data Bridge Market Research analysis, the Electroporation Instruments Market was estimated at USD 1.06 billion in 2025. The market is expected to grow from USD 1.14 billion in 2026 to USD 1.84 billion in 2032, at a CAGR of 8.23% during the forecast period with driven by the rising demand for gene therapy, cell-based therapeutics, advanced vaccine delivery technologies, and increasing investments in biotechnology research and clinical development.Get the full PDF sample copy of the report: (Includes full table of contents, list of tables and figures, and graphs): https://www.databridgemarketresearch.com/request-a-sample/?dbmr=global-electroporation-instruments-market
The market is witnessing strong expansion due to increasing adoption of electroporation systems in CRISPR gene editing, oncology research, DNA vaccine development, and cell therapy manufacturing. Rising R&D spending by pharmaceutical and biotechnology companies, combined with technological advancements in automated and high-throughput electroporation platforms, is accelerating commercialization across research and clinical applications. Regulatory support for advanced therapeutics and growing demand for non-viral transfection technologies are further strengthening market growth globally.
Market Size & Forecast
2025 Market Size: USD 1.06 Billion
2026 Projected Market Size: USD 1.14 Billion
2032 Projected Market Size: USD 1.84 Billion
CAGR (2026-2032): 8.23%
Largest Region: North America
Fastest Growing Region: Asia Pacific
Key Market Report Takeaways
North America accounted for the largest market share of approximately 38% due to strong biotechnology infrastructure, high R&D spending, and the presence of major market participants.
Asia Pacific is projected to register the fastest CAGR during the forecast period driven by expanding biopharmaceutical manufacturing and government-backed genomic research initiatives.
Clinical electroporation systems represented the largest product segment owing to increasing adoption in gene therapy and oncology applications.
Gene delivery and transfection applications dominated the market due to rising demand for CRISPR-based editing and cell engineering workflows.
Biotechnology and pharmaceutical companies remained the leading end-use segment because of increasing investment in cell therapy pipelines and biologics development.
Automated and high-throughput electroporation platforms are gaining rapid adoption across research laboratories and commercial manufacturing facilities.
Non-viral delivery technologies are increasingly preferred due to lower immunogenicity and improved scalability compared with viral vectors.
Market Trends & Highlights
North America continues to dominate the global market due to advanced healthcare infrastructure, strong clinical trial activity, and extensive investments in gene and cell therapy research.
Asia Pacific is emerging as the fastest-growing regional market supported by rising biotechnology funding, increasing genomic research programs, and expansion of pharmaceutical manufacturing capabilities in China, India, and Japan.
Gene editing and cell therapy applications remain the dominant market segment due to increasing commercialization of CRISPR-based therapeutics and engineered immune cell therapies.
Rising demand for non-viral transfection technologies, increasing prevalence of cancer and genetic disorders, and growth in precision medicine initiatives are major market growth drivers.
High-throughput and automated electroporation systems integrated with AI-enabled workflow optimization and scalable manufacturing technologies are reshaping laboratory and clinical applications.
Strategic collaborations between biotechnology firms, academic institutes, and technology providers are accelerating innovation, commercialization, and regulatory approvals across the market.
Details about the report and current availability can be viewed: https://www.databridgemarketresearch.com/reports/global-electroporation-instruments-market
Market Dynamics
Market Drivers
Rising Adoption of Gene and Cell Therapy Technologies
The increasing commercialization of gene and cell therapies is significantly driving demand for electroporation instruments. Electroporation enables efficient non-viral gene delivery into primary cells, T-cells, stem cells, and immune cells, supporting advanced therapeutic development. Growing clinical trials related to CAR-T therapy, CRISPR-based editing, and regenerative medicine are accelerating instrument adoption globally. North America and Europe remain key hubs for cell therapy manufacturing and translational research.
Expansion of CRISPR and Genome Editing Research
Rapid advancements in CRISPR-Cas9 and genome engineering technologies are increasing demand for high-efficiency transfection systems. Electroporation instruments offer improved editing precision, scalability, and reduced toxicity compared with chemical transfection approaches. Research institutions and biotechnology companies are increasingly integrating electroporation into genome editing workflows. Asia Pacific is witnessing particularly strong demand due to rising government investments in genomics and biotechnology innovation.
Growing Preference for Non-Viral Delivery Systems
Non-viral delivery methods are gaining traction because of safety concerns associated with viral vectors, including immunogenicity and insertional mutagenesis risks. Electroporation provides a scalable and clinically viable alternative for nucleic acid and protein delivery. Pharmaceutical companies are increasingly adopting electroporation systems to reduce manufacturing complexity and regulatory hurdles. This trend is particularly evident in advanced biologics and personalized medicine development.
Increasing Biopharmaceutical R&D Investments
Global investment in biotechnology and pharmaceutical R&D continues to rise, supporting broader deployment of advanced laboratory instruments. Electroporation technologies are increasingly utilized in vaccine development, oncology research, protein engineering, and biologics manufacturing. Public and private funding for precision medicine and genomic medicine programs is creating sustained demand across research institutions and commercial laboratories. The U.S., Germany, China, and Japan remain major investment centers.
Technological Advancements in Automated Electroporation Platforms
Manufacturers are introducing automated, scalable, and high-throughput electroporation systems to improve reproducibility and operational efficiency. Integration of digital controls, programmable parameters, and multi-sample processing capabilities is enhancing adoption in clinical and industrial environments. Advanced systems reduce processing time and improve transfection consistency across cell types. These innovations are supporting broader use in GMP-compliant manufacturing settings.
Market Restraints
High Capital and Operational Costs
Electroporation instruments involve significant upfront investment, particularly for clinical-grade and high-throughput systems. Maintenance costs, consumables, proprietary reagents, and software integration further increase total ownership expenses. Smaller laboratories and academic institutions in developing economies often face budget limitations that restrict adoption. Cost sensitivity remains a major challenge across Latin America and parts of Asia and Africa.
Complex Regulatory and Compliance Requirements
Clinical applications involving gene therapy and cell engineering are subject to strict regulatory oversight. Compliance with GMP standards, clinical validation requirements, and biosafety regulations increases operational complexity for manufacturers and end users. Regulatory variations across regions create additional commercialization barriers. Companies operating in Europe and North America face extensive documentation and validation obligations.
Technical Limitations in Sensitive Cell Applications
Certain primary cells and stem cells remain highly sensitive to electroporation-induced stress, reducing viability and transfection efficiency. Achieving optimal delivery conditions often requires extensive protocol optimization and technical expertise. Variability across cell types and molecular payloads can limit reproducibility in clinical and research workflows. These technical constraints may reduce adoption among less specialized laboratories.
Dependence on Proprietary Consumables and Protocols
Several leading instrument providers rely on proprietary buffers, cartridges, and software ecosystems, increasing operational dependency for customers. This limits flexibility and increases recurring expenses for laboratories. Integration challenges with existing laboratory workflows can also reduce adoption rates. Pricing pressure from customers is intensifying as alternative delivery technologies enter the market.
Competition from Alternative Delivery Technologies
Alternative gene delivery approaches including viral vectors, lipid nanoparticles, and microfluidic transfection systems are creating competitive pressure. Lipid nanoparticle technologies gained significant visibility following mRNA vaccine commercialization, increasing investment in competing platforms. Emerging technologies offering lower cell stress and higher throughput may challenge conventional electroporation systems over the long term.
Market Opportunities
Expansion of Personalized Medicine and Precision Therapies
Growing adoption of personalized medicine is creating strong opportunities for electroporation technologies in customized cell and gene therapies. Precision oncology and patient-specific immune cell engineering require efficient and scalable transfection platforms. Increasing healthcare investments in individualized treatment approaches are expected to drive long-term market expansion. North America and Europe are leading implementation, while Asia Pacific is emerging rapidly.
Growth Potential in Emerging Economies
Emerging markets including China, India, Brazil, and Southeast Asia present substantial growth opportunities due to expanding biotechnology infrastructure and healthcare modernization. Governments are increasing funding for genomic research and domestic biopharmaceutical manufacturing. Rising academic collaborations and foreign direct investments are strengthening regional market accessibility. Local manufacturing initiatives are also improving affordability and technology penetration.
Integration with Automated and High-Throughput Platforms
Demand for automated and scalable laboratory workflows is opening opportunities for advanced electroporation systems. Integration with robotic handling, AI-assisted optimization, and high-throughput screening platforms can significantly improve efficiency and reproducibility. Biotechnology companies increasingly require scalable transfection systems for commercial production. This trend supports premium product development and recurring software-based revenue models.
Strategic Partnerships and Licensing Agreements
Collaborations between biotechnology firms, CROs, and electroporation technology providers are accelerating commercialization opportunities. Licensing agreements supporting cell therapy manufacturing and gene editing workflows are becoming increasingly common. Strategic partnerships help companies expand geographic reach and strengthen product portfolios. These collaborations are expected to intensify as demand for advanced therapeutics increases globally.
Advancements in DNA Vaccine and Immunotherapy Research
Electroporation is gaining traction in DNA vaccine delivery and immunotherapy development due to improved cellular uptake and immune response generation. Rising investment in infectious disease preparedness and cancer immunotherapy is supporting market expansion. Clinical studies involving electroporation-assisted vaccine delivery are increasing across North America, Europe, and Asia Pacific.
Market Challenges
Maintaining Cell Viability During High-Efficiency Transfection
Balancing transfection efficiency with cell survival remains a critical technical challenge. High-voltage electrical pulses can damage sensitive primary cells and reduce therapeutic effectiveness. Achieving standardized protocols across multiple cell types requires extensive optimization and technical expertise. This challenge impacts both research reproducibility and large-scale clinical manufacturing.
Limited Standardization Across Platforms
Variability in electroporation parameters, consumables, and protocols across manufacturers creates interoperability challenges. Laboratories often face difficulties transferring protocols between systems, reducing operational flexibility. Lack of universal standardization can delay clinical validation and increase workflow complexity. This issue is particularly significant in multi-site research collaborations.
Supply Chain and Component Availability Risks
Electroporation systems rely on specialized electronic components, consumables, and precision manufacturing processes. Global semiconductor shortages and logistics disruptions have impacted instrument production and delivery timelines. Dependence on limited suppliers for proprietary consumables increases operational risk for laboratories and manufacturers alike.
Intense Competitive Pressure and Pricing Constraints
The market is becoming increasingly competitive with the entry of new biotechnology startups and alternative transfection technologies. Established companies face pricing pressure from low-cost regional manufacturers and emerging delivery platforms. Maintaining profitability while investing in R&D and regulatory compliance remains challenging, particularly for mid-sized players.
Complex Clinical Translation and Commercialization
Transitioning electroporation technologies from research environments to commercial clinical manufacturing involves significant technical and regulatory hurdles. Scalability, reproducibility, and GMP compliance requirements increase development timelines and operational costs. Clinical adoption remains dependent on successful validation and regulatory approvals in multiple regions.
Get Detailed Table of Contents (TOC) - Request Now for Complete Market Insights: https://www.databridgemarketresearch.com/toc/?dbmr=global-electroporation-instruments-market
Market Segmentation & Analysis
By Product Type
Clinical Electroporation Systems
Clinical electroporation systems accounted for the largest market share due to increasing use in gene therapy, immunotherapy, and oncology applications. These systems are designed for GMP-compliant workflows and large-scale cell processing. Growing commercialization of cell-based therapeutics continues to support segment growth. The segment is projected to maintain strong CAGR through 2032 due to rising clinical adoption.
Research Electroporation Systems
Research-grade electroporation systems are widely used across academic institutes and biotechnology laboratories for transfection and gene editing studies. The segment benefits from expanding genomic research and increasing CRISPR experimentation. Lower system costs compared with clinical platforms support broader accessibility. Asia Pacific is witnessing rapid demand growth in this category.
Consumables and Accessories
Consumables including cuvettes, electrodes, buffers, and cartridges represent a recurring revenue stream for manufacturers. Increasing instrument installations globally are supporting sustained demand for proprietary consumables. Companies are focusing on high-margin consumable ecosystems to improve profitability and customer retention.
By Application
Gene Delivery and Transfection
Gene delivery and transfection remained the dominant application segment due to rising demand for CRISPR editing, DNA/RNA delivery, and recombinant protein production. Electroporation offers higher efficiency and scalability for difficult-to-transfect cells. Pharmaceutical and biotechnology companies increasingly rely on this technology for therapeutic development.
Cancer Treatment
Electroporation-assisted cancer therapies, including electrochemotherapy and tumor ablation, are gaining clinical traction. Increasing prevalence of cancer and growing focus on targeted therapies are supporting segment expansion. Europe remains a leading region for clinical electrochemotherapy adoption.
Vaccine Development
DNA vaccine development is emerging as a high-growth application segment. Electroporation enhances antigen uptake and immune response generation, improving vaccine efficacy. Increased investment in infectious disease preparedness and next-generation vaccine technologies is driving adoption globally.
By End User
Biotechnology and Pharmaceutical Companies
This segment held the largest revenue share due to strong investment in cell therapy, biologics, and genomic medicine research. Companies require scalable and GMP-compliant electroporation systems for commercial manufacturing. Strategic licensing partnerships are further supporting segment dominance.
Academic and Research Institutes
Academic institutions are major adopters of research-grade electroporation instruments for molecular biology and genetic engineering studies. Government funding for life sciences research is accelerating demand, particularly in North America and Asia Pacific.
Contract Research and Manufacturing Organizations
CROs and CDMOs are emerging as fast-growing end users due to increasing outsourcing of cell therapy and biologics manufacturing. Demand for flexible and scalable transfection technologies is expanding rapidly within this segment.
Regional Analysis
By geography, the market is categorized into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
North America
North America represents the largest regional market, accounting for approximately 38% of global revenue. The region benefits from advanced biotechnology infrastructure, strong government funding, and extensive clinical research activity. The United States dominates regional demand due to the presence of leading biotechnology companies and strong adoption of gene therapy technologies. High R&D expenditure and favorable reimbursement frameworks continue to support market expansion.
Europe
Europe is a mature and steadily growing market driven by strong regulatory frameworks and significant investment in life sciences research. Germany, the U.K., and France remain major contributors due to advanced healthcare infrastructure and expanding biotechnology sectors. The region has strong adoption of electrochemotherapy and gene editing research applications. EU funding programs are further supporting innovation and commercialization activities.
Asia Pacific
Asia Pacific is projected to register the fastest growth rate during the forecast period. Rapid industrialization, increasing biotechnology investments, and government support for genomic research are driving market expansion. China, Japan, and India are major growth contributors due to expanding pharmaceutical manufacturing and increasing clinical research activities. Rising healthcare expenditure and local manufacturing capabilities are strengthening regional competitiveness.
Latin America
Latin America is an emerging market characterized by gradual technology adoption and improving healthcare infrastructure. Brazil and Mexico represent the largest regional contributors due to expanding biotechnology research and increasing healthcare modernization initiatives. Economic limitations and budget constraints continue to affect large-scale adoption. However, growing foreign investments are expected to support future growth.
Middle East & Africa
The Middle East & Africa market is experiencing steady but comparatively slower growth. Government investments in healthcare modernization and biotechnology development are supporting market expansion, particularly in Gulf countries. Private sector participation and research collaborations are increasing gradually. Infrastructure gaps and limited availability of advanced laboratory technologies continue to restrict market penetration across several countries.
Key Insights:
Largest Region: North America
Fastest Growing Region: Asia Pacific
Competitive Landscape
Market Structure Overview
The electroporation instruments market is moderately consolidated with the presence of established global leaders, regional manufacturers, and emerging biotechnology startups. Competition is driven by technological innovation, transfection efficiency, automation capabilities, and scalability for clinical applications. Companies are increasingly focusing on high-throughput systems, non-viral delivery technologies, and integrated software solutions. Competitive landscape analysis provides insight into market positioning, technological capabilities, and strategic expansion initiatives of key participants.
Key Industry Players
Leading market participants focus on portfolio expansion, advanced electroporation technologies, and strategic partnerships with biotechnology firms. Global players maintain strong competitive positions through extensive distribution networks and continuous innovation.
List of Key Industry Players
Thermo Fisher Scientific
Bio-Rad Laboratories
Lonza Group
MaxCyte
Harvard Bioscience
BTX Molecular Delivery Systems
Eppendorf
BEX Co. Ltd.
Inovio Pharmaceuticals
Competitive Strategies
Companies are increasingly investing in product innovation, automated platforms, and high-throughput transfection systems to strengthen market differentiation. Strategic collaborations with cell therapy developers and biotechnology firms are expanding commercial opportunities. Mergers, acquisitions, and licensing agreements are supporting portfolio diversification and geographic expansion. Leading players are also strengthening distribution networks and GMP-compliant manufacturing capabilities to improve customer value proposition and competitive positioning.
Emerging Players & Market Dynamics
Emerging biotechnology companies and niche innovators are intensifying market competition through specialized and cost-effective transfection technologies. Startups focusing on microfluidics, AI-assisted optimization, and non-viral delivery innovations are challenging traditional market leaders. Increasing venture capital investments and funding activities are accelerating technology commercialization. Digital transformation and automation remain major competitive themes shaping future market dynamics.
Latest Developments
Latest Developments
February 2026 - MaxCyte: The company launched the ExPERT DTx 96-well electroporation platform designed for high-throughput research and drug discovery applications. The launch strengthens automation capabilities and improves scalability for genomic and cell engineering workflows.
August 2025 - Fujifilm and HORIBA: The companies announced co-development of a continuous electroporation-based gene delivery system aimed at improving productivity in gene therapy manufacturing. The technology is expected to significantly enhance scalability and manufacturing efficiency for advanced therapeutics.
May 2024 - MaxCyte and Legend Biotech: MaxCyte entered a strategic platform licensing agreement with Legend Biotech to support cell therapy discovery and commercialization. The partnership expanded adoption of non-viral electroporation technologies in engineered cell therapy pipelines.
April 2024 - MaxCyte and Be Biopharma: The companies signed a strategic platform license agreement supporting development of engineered B-cell medicines. The collaboration highlights growing use of electroporation technologies in next-generation immune cell engineering.
January 2024 - MaxCyte and Lion TCR: The partnership aimed to accelerate development and global expansion of TCR-T cell therapies using electroporation-based cell engineering platforms. The agreement strengthened MaxCyte's presence across Asia and international markets.
January 2024 - MaxCyte and Wugen: The companies announced a licensing agreement to support scale-up of allogeneic cell therapy manufacturing for cancer treatment applications. The collaboration reinforced demand for scalable electroporation technologies in commercial cell therapy production.
Check out more related studies published by Data Bridge Market Research:
https://www.databridgemarketresearch.com/reports/north-america-medical-aesthetics-market
https://www.databridgemarketresearch.com/reports/north-america-pharmacy-automation-market
https://www.databridgemarketresearch.com/reports/asia-pacific-pharmacy-automation-market
https://www.databridgemarketresearch.com/reports/latin-america-allergy-diagnostics-market
https://www.databridgemarketresearch.com/reports/asia-pacific-medical-imaging-market
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email:- corporatesales@databridgemarketresearch.com
Data Bridge Market Research follow a wide array of models that allow proactive collaboration with clients, categorize new sources of incremental revenues, deliver revenue planning, and first-mover advantage about innovations and disruptions through early market research.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Electroporation Instruments Market Outlook 2026-2032: Market to Reach USD 1.84 Billion by 2032 Amid Rising Adoption of Gene and Cell Therapy Technologies here
News-ID: 4508732 • Views: …
More Releases from Data Bridge Market Research
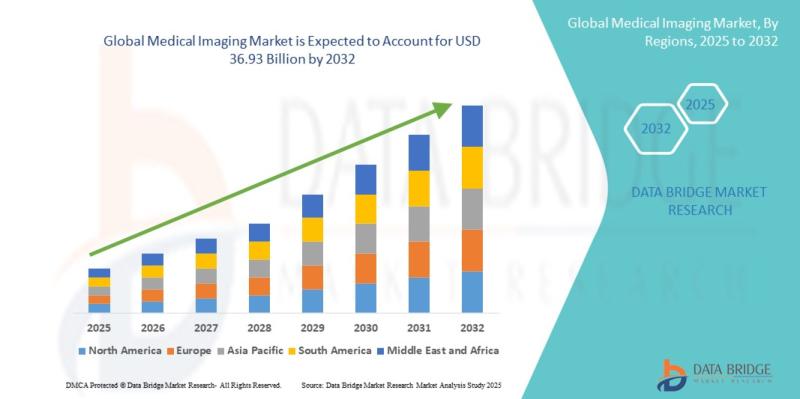
Medical Imaging Market Growth Report 2026-2032: Industry Expected to Reach USD 3 …
"As per Data Bridge Market Research analysis, the Medical Imaging was estimated at USD 25.07 billion in 2025. The market is expected to grow from USD 26.49 billion in 2026 to USD 36.93 billion in 2032, at a CAGR of 5.69% during the forecast period with driven by the rising demand for advanced diagnostic technologies, increasing prevalence of chronic diseases, expanding geriatric population, and rapid integration of artificial intelligence (AI)…
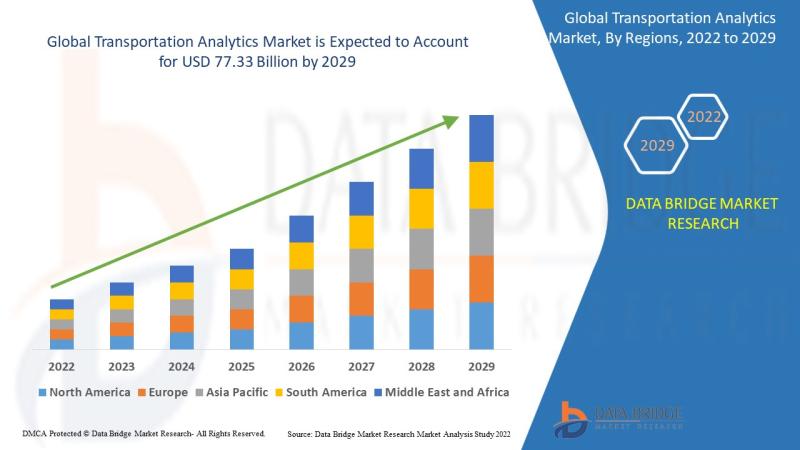
Is the Transportation Analytics Market Expanding Rapidly with Smart Mobility and …
As per Data Bridge Market Research analysis, the Transportation Analytics Market was estimated at USD 34.77 billion in 2025. The market is expected to grow from USD 42.45 billion in 2026 to USD 140.73 billion in 2032, at a CAGR of 22.10% during the forecast period with driven by the rising demand for real-time fleet optimization, increasing adoption of AI and IoT-enabled transportation systems, growing investments in smart city infrastructure,…
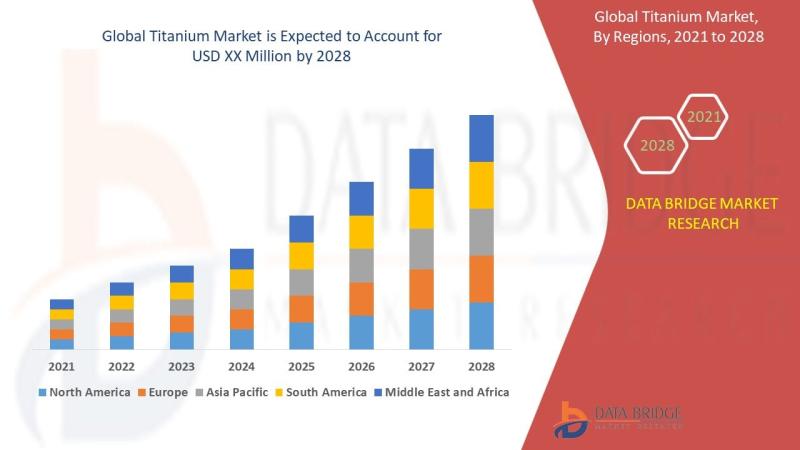
How Is the Titanium Market Evolving with Demand for Corrosion-Resistant and High …
As per Data Bridge Market Research analysis, the Titanium Market was estimated at USD 2.76 billion in 2025. The market is expected to grow from USD 2.93 billion in 2026 to USD 4.26 billion in 2032, at a CAGR of 6.40% during the forecast period with driven by the rising demand for lightweight and high-strength materials across aerospace, defense, medical, automotive, and industrial applications. Increasing adoption of titanium alloys in…
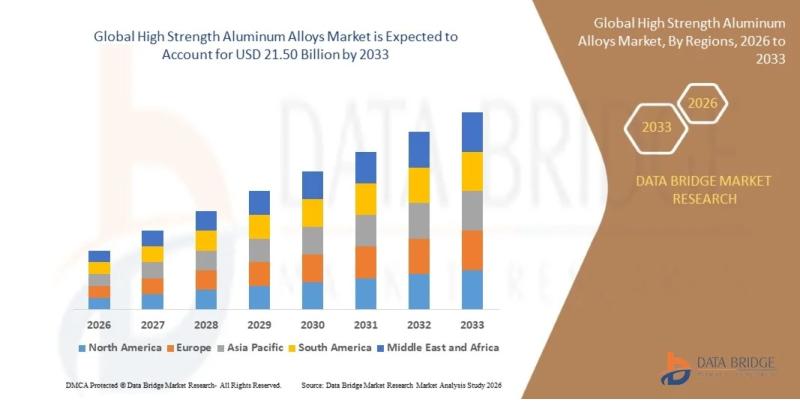
High Strength Aluminum Alloys Market to Grow at 7.95% CAGR Through 2033
Market Summary
"As per Data Bridge Market Research analysis, the High Strength Aluminum Alloys Market was estimated at USD 11.66 billion in 2025. The market is expected to grow from USD 12.59 billion in 2026 to USD 21.50 billion in 2033, at a CAGR of 7.95% during the forecast period with driven by the rising demand for lightweight materials in aerospace, automotive, defense, and industrial manufacturing applications."
Get the full PDF sample…
More Releases for Electroporation
Electroporation Instruments Market Insights Shared in Detailed Report
Electroporation Instruments Market: Introduction
According to the report, the global electroporation instruments market was valued at ~US$ 126 Mn in 2019 and is projected to reach ~US$ 244 Mn by 2030, expanding at a CAGR of ~6% from 2020 to 2030. The electroporation technique involves exposure of cell membranes to high-intensity electric pulses that cause a temporary destabilization in certain areas of the cell. During this transient destabilization event, the cell membrane becomes…
Global Medical Aesthetic Device with Electroporation | Weero
WEERO is a global aesthetic device manufacturer in Korea. It provides valuable medical aesthetic devices to the customers around the world so that they can improve their skin of the face and body to be comforted by the result.
Weero Apollo Duet +EL
Korean medical aesthetic device Weero Apollo Duet +EL has 4 in 1 Technologies of Electroporation + CRYO + Thermo + Pulse Selection
The electroporation uses high-voltage electric pulses to increase…
Innovation Backed By Elegance To Accelerate The Electroporation Instruments Mark …
An implants for the deformity spinal system are for single use only. The deformity spinal implant should never be re-sterilized after coming into with body tissues and fluids and never be reused after being removed from the body.
The deformity spinal system is used in open and minimally invasive surgery (MIS) and used for the treatment of degenerative disc disease, Spondylolisthesis, Spinal Stenosis, fracture or dislocation, Pseudoarthrosis and other deformities and…
Apollo Duet +EL Medical Aesthetic Device with Electroporation | Weero
WEERO is a global aesthetic device manufacturer in Korea and provides the most advanced and newest aesthetic medical devices and beauty devices to the world.
With research and development of the most cutting-edge technologies, WEERO is continuing to move upward staying, ahead of the market to always meet the needs of the customers.
WEERO will provide valuable medical aesthetic devices for customers to improve the skin and body and be comforted.
WEERO…
World-class Radio Frequency and Electroporation Technology | Weero
WEERO is a global manufacturer to provide most advanced and the newest aesthetic medical device and beauty device to the world.
The company name of WEERO means warm and upward. WEERO is continuing to research and develop the most cutting edge technologies to stay ahead of the market, and always moves upward and to be warm to comfort the customers.
WEERO will provide valuable medical aesthetic devices to the customers so that…
4 in 1 Medical Aesthetic Device with Electroporation | Weero
The company name of WEERO means warm and upward.
WEERO is a global aesthetic device manufacturer in Korea. It provides valuable medical aesthetic devices to the customers around the world so that they can improve their skin of face and body to be comforted by the result.
Korean Medical Aesthetic Device WEERO Apollo Duet +EL
WRREO Apollo Duet +EL has4 in 1 Technologies of Electroporation + CRYO…