Press release
Insulin Pens Manufacturing Plant DPR 2026: Cost Structure, Production Process & ROI
Setting up an insulin pens manufacturing plant positions investors in one of the most critical and high-growth segments of the medical device and diabetes care value chain, backed by sustained global growth driven by the rising global prevalence of diabetes, increasing adoption of patient-friendly drug delivery devices, technological advancements in injectable systems, and growing demand for home-based diabetes management solutions. As the global diabetic population continues to expand, healthcare systems increasingly shift toward home-based treatment models, and pharmaceutical companies differentiate insulin products through pen-based delivery systems, the global insulin pens industry continues to present compelling opportunities for manufacturers and entrepreneurs seeking long-term profitability in a high-demand sector.Market Overview and Growth Potential:
The global insulin pens market demonstrates a strong growth trajectory, valued at USD 8.40 Billion in 2025. According to IMARC Group's comprehensive market analysis, the market is expected to reach USD 17.07 Billion by 2034, exhibiting a CAGR of 8.2% from 2026 to 2034. This sustained expansion is driven by the escalating prevalence of diabetes and the increasing need for effective self-administration devices, aging populations, sedentary lifestyles, and rising obesity levels that have contributed to higher insulin dependency globally.
Request for a Sample Report: https://www.imarcgroup.com/insulin-pens-manufacturing-plant-project-report/requestsample
Insulin pens are medical drug delivery devices designed to administer insulin subcutaneously in a controlled, accurate, and user-friendly manner. They consist of a pen-like body incorporating an insulin cartridge or prefilled reservoir, a dosing mechanism, and a disposable or replaceable needle. Insulin pens are categorized into reusable and disposable variants, both offering precise dose selection, portability, and ease of use compared to traditional vial-and-syringe methods. Insulin pens play a critical role in diabetes management by improving patient compliance, dosing consistency, and overall treatment outcomes.
The global insulin pens industry is primarily driven by the escalating prevalence of diabetes and the increasing need for effective self-administration devices. The International Diabetes Federation predicts that new cases of diabetes in people will continue to rise year after year, reaching 853 million by 2050. Additionally, healthcare systems are emphasizing home-based treatment models, accelerating adoption of easy-to-use insulin pens. Continuous innovation in device design, including dose accuracy improvements and user-safety mechanisms, further supports market expansion. Pharmaceutical companies increasingly prefer pen-based delivery systems to differentiate insulin products and enhance patient compliance.
Plant Capacity and Production Scale:
The proposed insulin pens manufacturing facility is designed with an annual production capacity ranging between 10 - 50 million units, enabling economies of scale while maintaining operational flexibility. This capacity range allows manufacturers to cater to diverse market segments-from pharmaceuticals and healthcare to medical device manufacturing, home healthcare, and diabetes care-ensuring steady demand and consistent revenue streams across multiple industry verticals. The facility is designed to serve both domestic supply chains and export requirements, positioning the plant at the intersection of industrial efficiency and healthcare facilitation.
Speak to Analyst for Customized Report: https://www.imarcgroup.com/request?type=report&id=9600&flag=C
Financial Viability and Profitability Analysis:
The insulin pens manufacturing business demonstrates healthy profitability potential under normal operating conditions. The financial projections reveal:
Gross Profit Margins: 35-45%
Net Profit Margins: 10-20%
These margins are supported by stable demand across pharmaceuticals, healthcare, medical device manufacturing, home healthcare, and diabetes care sectors, value-added specialty insulin pen positioning including reusable and disposable variants with precision dosing mechanisms, and the critical role of insulin pens in enabling safe and effective diabetes management worldwide. The project demonstrates strong return on investment (ROI) potential, making it an attractive proposition for both new entrants and established medical device or pharmaceutical manufacturers looking to diversify their portfolio.
Cost of Setting Up an Insulin Pens Manufacturing Plant:
Operating Cost Structure:
Understanding the operating expenditure (OpEx) is crucial for effective financial planning and cost management. The cost structure for an insulin pens manufacturing plant is primarily driven by:
Raw Materials: 40-50% of total OpEx
Utilities: 10-15% of OpEx
Other Expenses: Including labor, packaging, transportation, maintenance, depreciation, and taxes
Raw materials constitute the largest portion of operating costs, with medical plastics being the primary input material. Insulin cartridges form the secondary raw material requirement. Establishing long-term contracts with reliable medical plastics and insulin cartridge suppliers helps mitigate price volatility and ensures consistent raw material supply, which is critical given that medical-grade material price fluctuations represent the most significant cost factor in insulin pens manufacturing.
Capital Investment Requirements:
Setting up an insulin pens manufacturing plant requires substantial capital investment across several critical categories:
Land and Site Development:
Selection of an optimal location with strategic proximity to medical plastics and insulin cartridges suppliers. Proximity to target pharmaceutical, healthcare, and medical device markets will help minimize distribution costs. The site must have robust infrastructure, including reliable transportation, utilities, and waste management systems. Compliance with local zoning laws, medical device manufacturing regulations, and environmental requirements must also be ensured.
Machinery and Equipment:
The largest portion of capital expenditure (CapEx) covers specialized manufacturing equipment essential for production. The process used includes microforming, tempering, and automated assembly. Key machinery includes:
• Cleanrooms: for maintaining controlled, contamination-free environments required for the manufacture of medical-grade insulin pen components and assemblies in compliance with GMP standards
• Automated filling and assembly lines: for high-speed, precision assembly of insulin pen components including pen housings, cartridge housings, dosing mechanisms, and needle interfaces
• Sterilizing systems: for sterilization of insulin pen components and sub-assemblies to meet medical device safety and regulatory standards for patient use
• Leak-testing machinery: for detection and elimination of any leakage defects in insulin pen cartridge housings and reservoir systems prior to final assembly and packaging
• Quality control inspection stations: for comprehensive dimensional, functional, and visual inspection of insulin pen components and finished devices against specifications and regulatory requirements
• Automated packaging systems: for blister packing, protective casing, and transport container assembly of finished insulin pens in compliance with pharmaceutical packaging standards
• Cold storage units: for temperature-controlled storage of insulin cartridges and other temperature-sensitive raw materials and components throughout the manufacturing and storage process
Civil Works:
Building construction, factory layout optimization, and infrastructure development designed to enhance workflow efficiency, ensure workplace safety, and minimize material handling complexities throughout the production process. The layout should be optimized with separate areas for raw material storage, cleanroom manufacturing zone, assembly section, sterilization area, quality control station, finished goods warehouse, cold storage area, utility block, and administrative block.
Other Capital Cost:
Pre-operative expenses, machinery installation costs, regulatory compliance and medical device certification costs (FDA, CE, etc.), initial working capital requirements, and contingency provisions for unforeseen circumstances during plant establishment.
Buy Now: https://www.imarcgroup.com/checkout?id=9600&method=2175
Major Applications and Market Segments:
Insulin pens products find extensive applications across diverse market segments, demonstrating their versatility and critical importance across the global diabetes care and medical device value chain:
Drug Delivery Components: Insulin pens serve as the primary platform for pen housings, cartridges, and dosing mechanisms used in the subcutaneous delivery of insulin to diabetic patients. They are foundational to modern diabetes management systems, enabling precise dose selection, portability, and user-friendly self-administration across hospital, clinic, and home healthcare settings.
Needle Systems: Insulin pens incorporate microneedles, safety shields, and detachable needles that enable safe, minimally invasive subcutaneous injection of insulin. Needle system components across pharmaceutical, medical device, and home healthcare sectors rely on insulin pens as the universal standard for patient-controlled insulin delivery and dosing.
Electronics & Sensors: Advanced insulin pen variants integrate digital dose counters, connectivity modules, and tracking systems that enable real-time dose monitoring, data logging, and digital health integration. Electronics and sensor components support connected diabetes management platforms, patient adherence tracking, and healthcare provider monitoring across digital health and home healthcare verticals.
Packaging & Distribution: Insulin pens require specialized blister packs, protective cases, and transport containers to maintain product integrity, sterility, and regulatory compliance throughout the distribution chain. Pharmaceutical packaging solutions support the safe delivery of insulin pens from manufacturing facilities to hospitals, pharmacies, and end-users across global healthcare supply chains.
Why Invest in Insulin Pens Manufacturing?
Several compelling factors make insulin pens manufacturing an attractive investment opportunity:
Essential Healthcare Component:
Insulin pens are critical for diabetes management, providing precise, safe, and convenient drug delivery for millions of patients worldwide. Their role in improving adherence and patient outcomes makes them an indispensable healthcare product. This structural indispensability ensures consistent, non-cyclical demand that spans across all pharmaceutical, healthcare, and diabetes care sectors.
Moderate but Justifiable Entry Barriers:
Manufacturing insulin pens requires specialized tooling, precision engineering, stringent quality controls, and regulatory approvals (FDA, CE, etc.). While the investment is lower than for high-tech electronics, these requirements favor experienced producers who can consistently deliver high-quality, reliable products.
Megatrend Alignment:
Rising global prevalence of diabetes, increasing adoption of self-administered therapies, and a shift toward patient-centric care are driving sustained demand for insulin pens. Markets in emerging economies are witnessing strong growth, creating long-term expansion opportunities.
Policy & Healthcare Initiatives:
Government interventions like diabetes care support programs, insurance coverage of insulin delivery pens, and schemes supporting domestic pharma production (Make in India, PLI schemes) have an indirect positive effect on demand volume of insulin pens.
Localization and Supply Chain Dependability:
Hospitals, clinics, and pharmaceutical distribution firms also prefer local and reliable sources in order to facilitate the steady supply of their needs without any logistics hassles, thereby paving the way for local manufacturers to be more efficient and reliable in their sourcing and processing.
Manufacturing Process Excellence:
The insulin pens manufacturing process involves several precision-controlled stages to deliver standardized, regulatory-compliant, and market-ready medical devices. The process used includes microforming, tempering, and automated assembly:
• Component Manufacturing: Medical plastics are processed through microforming operations to produce precision-dimensioned pen housings, cartridge housings, dosing mechanism components, and needle interface parts to tight tolerances
• Tempering: Manufactured metal and plastic components undergo tempering processes to achieve target mechanical properties, dimensional stability, and structural integrity required for reliable insulin pen function and long-term durability
• Component Inspection: Individual components are inspected at quality control inspection stations for dimensional accuracy, surface finish, and compliance with medical device specifications before advancing to assembly
• Automated Assembly: Precision components including pen housings, dosing mechanisms, cartridge housings, needle interfaces, and precision spring components are assembled using automated assembly lines to form complete insulin pen sub-assemblies and finished devices
• Cartridge Integration: Insulin cartridges are integrated into the assembled pen bodies under controlled conditions in cleanroom environments to maintain sterility and product integrity throughout the filling and assembly process
• Sterilization: Assembled insulin pen components and sub-assemblies undergo sterilization using validated sterilizing systems to meet medical device safety requirements and regulatory standards for patient-contact devices
• Leak Testing: Finished insulin pen assemblies are subjected to leak-testing machinery to detect and reject any units with cartridge housing or reservoir system leakage defects prior to packaging
• Quality Control Inspection: Finished insulin pens undergo comprehensive functional testing including dose accuracy verification, dose counter operation, needle attachment integrity, and dimensional compliance at quality control inspection stations
• Automated Packaging: Approved finished insulin pens are packed into blister packs, protective cases, and transport containers using automated packaging systems in compliance with pharmaceutical packaging and labeling regulations
• Cold Storage: Packaged insulin pens containing insulin cartridges are transferred to cold storage units for temperature-controlled storage and dispatch to maintain product efficacy and regulatory compliance
Industry Leadership:
The global insulin pens industry is led by established pharmaceutical and medical device manufacturers with extensive production capabilities and diverse application portfolios. Key industry players include:
• Novo Nordisk
• Sanofi
• Eli Lilly and Company
• BD (Becton Dickinson)
• Ypsomed
These companies serve diverse end-use sectors including pharmaceuticals, healthcare, medical device manufacturing, home healthcare, and diabetes care, demonstrating the broad market applicability of insulin pen products across global diabetes management and drug delivery verticals.
Recent Industry Developments:
November 2024: Medtronic plc announced U.S. FDA clearance for its new InPen app featuring missed meal dose detection, paving the way for the launch of its Smart MDI system with the Simplera continuous glucose monitor (CGM).
Browse Full Report: https://www.imarcgroup.com/insulin-pens-manufacturing-plant-project-report
About Us:
IMARC Group is a global management consulting firm that helps the world's most ambitious changemakers create a lasting impact. The company excels in understanding its clients' business priorities and delivering tailored solutions that drive meaningful outcomes. IMARC Group provides a comprehensive suite of market entry and expansion services, including market assessment, feasibility studies, company incorporation assistance, factory setup support, regulatory approvals and licensing navigation, branding, marketing and sales strategies, competitive landscape and benchmarking analyses, pricing and cost research, and procurement research.
Contact Us:
IMARC Group
134 N 4th St. Brooklyn, NY 11249, USA
Email: sales@imarcgroup.com
Tel No: (D) +91 120 433 0800
United States: (+1-201-971-6302)
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Insulin Pens Manufacturing Plant DPR 2026: Cost Structure, Production Process & ROI here
News-ID: 4506277 • Views: …
More Releases from IMARC Group

India Bottled Water Market to Reach USD 29.70 Billion by 2034, Growing at 12.00% …
How is India Bottled Water Market Performing?
India's bottled water industry is entering a high-growth expansion phase, driven by rising concerns over drinking water safety, accelerating urbanization, and increasing consumer preference for convenient, hygienic hydration solutions. As one of the fastest-growing packaged beverage categories in the country, bottled water has evolved from a travel necessity into an everyday essential across households, workplaces, hospitality, healthcare, and institutional environments.
Behind this strong market momentum…

Ethylene Carbonate Production Plant DPR 2026: Raw Materials, Machinery Cost and …
Setting up an Ethylene Carbonate (EC) production plant positions investors in one of the most strategically critical and fast-growing segments of the specialty chemicals and clean energy materials value chain, backed by sustained global growth driven by the rising demand for lithium-ion batteries, growth in electric vehicles, expansion of electronics manufacturing, and increasing use in lubricants and chemical intermediates. As battery cell manufacturers, electric vehicle producers, consumer electronics companies, specialty…
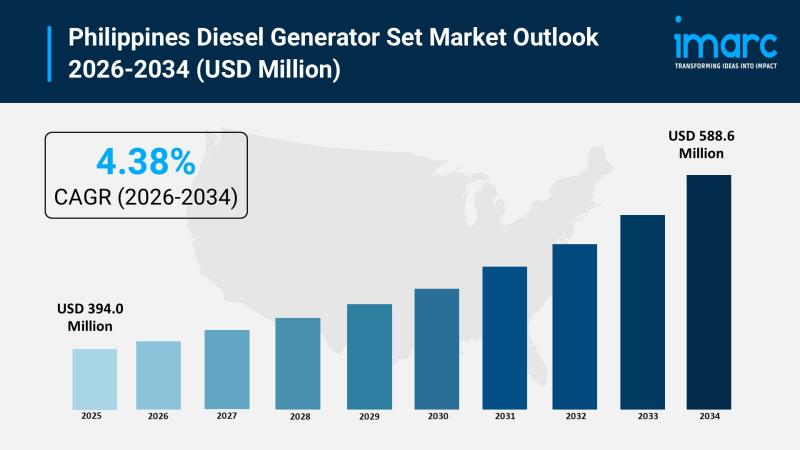
Philippines Diesel Generator Set Market Anticipated to Reach USD 588.6 Million D …
Philippines Diesel Generator Set Market Overview:
The Philippines diesel generator set market size reached USD 394.0 Million in 2025. Looking forward, the market is expected to reach USD 588.6 Million by 2034, exhibiting a growth rate (CAGR) of 4.38% during 2026-2034. The market encompasses generator sets across four capacity ranges (0-100 kVA, 100-350 kVA, 350-1000 kVA, above 1000 kVA) serving standby backup power, prime power, and peak shaving power applications through…
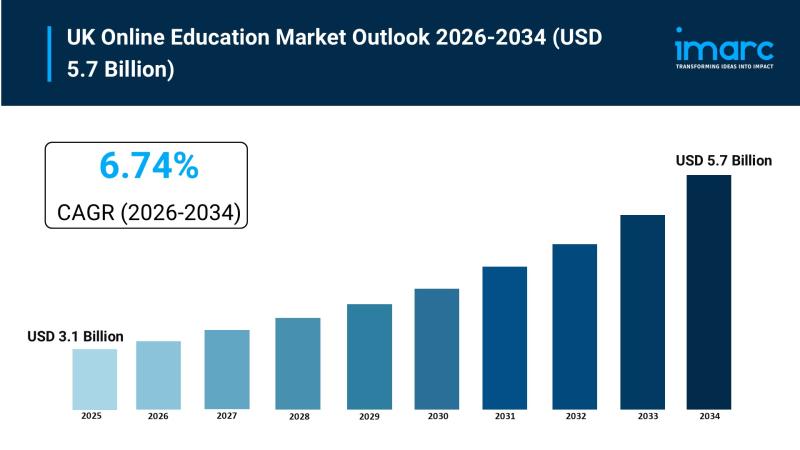
UK Online Education Market Size to Hit USD 5.7 Billion by 2034 | With a 6.74% CA …
UK Online Education Market Overview
Market Size in 2025: USD 3.1 Billion
Market Size in 2034: USD 5.7 Billion
Market Growth Rate 2026-2034: 6.74%
According to IMARC Group's latest research publication, "UK Online Education Market Report by Course Type, Learning Type, End User, and Region, 2026-2034," the UK online education market size reached USD 3.1 Billion in 2025. Looking forward, IMARC Group expects the market to reach USD 5.7 Billion by 2034, exhibiting a…
More Releases for Insulin
Insulin Delivery System Advancement Drives the Global Insulin Path Pumps Market
Inkwood Research expects the Global Insulin Patch Pumps Market to surge with a 10.63% CAGR by 2032, and is set to generate $2760.98 million during the forecast period 2023-2032.
Browse 50 market data Tables and 47 Figures spread over 173 Pages, along with an in-depth analysis of the Global Insulin Patch Pumps Market by Type, Delivery Mode, Disease Indication, Distribution Channel, & by Geography.
Refer to the Report Summary Here: https://inkwoodresearch.com/reports/insulin-patch-pumps-market/#report-summary?utm_source=PaidPRNew&utm_medium=OpenPR&utm_campaign=InkwoodPR
This…
Advancing Insulin Delivery Systems boost the Global Insulin Path Pumps Market
Inkwood Research expects the Global Insulin Patch Pumps Market to surge with a 10.63% CAGR by 2032, and is set to generate $2760.98 million during the forecast period 2023-2032.
Browse 50 market data Tables and 47 Figures spread over 173 Pages, along with an in-depth analysis of the Global Insulin Patch Pumps Market by Type, Delivery Mode, Disease Indication, Distribution Channel, & by Geography.
Refer to the Report Summary Here: https://inkwoodresearch.com/reports/insulin-patch-pumps-market/#report-summary
This insightful…
Advancing Insulin Delivery Systems boost the Global Insulin Path Pumps Market
Inkwood Research expects the Global Insulin Patch Pumps Market to surge with a 10.63% CAGR by 2032, and is set to generate $2760.98 million during the forecast period 2023-2032.
Browse 50 market data Tables and 47 Figures spread over 173 Pages, along with an in-depth analysis of the Global Insulin Patch Pumps Market by Type, Delivery Mode, Disease Indication, Distribution Channel, & by Geography.
Refer to the Report Summary Here: https://inkwoodresearch.com/reports/insulin-patch-pumps-market/#report-summary
This insightful…
Insulin Pumps Market Share, Trends and Growth Analysis By Type (Traditional Insu …
Insulin Pumps Market is expected to register a CAGR of 15.5% and acquire the market value of USD 11.5 Billion by 2028 during forecast period 2023-2032. Insulin pumps are discreet, electronic medical devices that can be tucked beneath clothing or fastened to a belt. In order to regulate the rise in blood glucose levels, the pump is used to deliver controlled quantities of insulin within the body at regular intervals…
Insulin Patch Pumps Market Report 2018: Segmentation by Insulin Type (Bolus Insu …
Global Insulin Patch Pumps market research report provides company profile for Roche Holding AG, Cellnovo Group SA, Spring Health Solution Ltd., Debiotech, CeQur SA, Valeritas, Becton Dickenson & Company, Insulet Corporation, Johnson & Johnson, Medtrum Technologies Inc. and Others.
This market study includes data about consumer perspective, comprehensive analysis, statistics, market share, company performances (Stocks), historical analysis 2012 to 2017, market forecast 2018 to 2025 in terms of volume, revenue,…
Insulin pump Market Explore Future Growth 2018-2026 by Global Type-Traditional i …
Insulin pump is a portable device attached to the body that continuously delivers preset amounts of short or rapid acting insulin in the body to control diabetes. Insulin therapy is required in type 1 diabetes and sometime in type 2 diabetes. It serves as an effective alternative to insulin injections. Furthermore, insulin pump delivers the insulin according to the need of body as basal rate, where small amount of insulin…
