Press release
Blood Collection Tube for Liquid Biopsy Market Records 19.6% CAGR Growth Outlook, Supported by Streck, QIAGEN, Greiner Bio-One
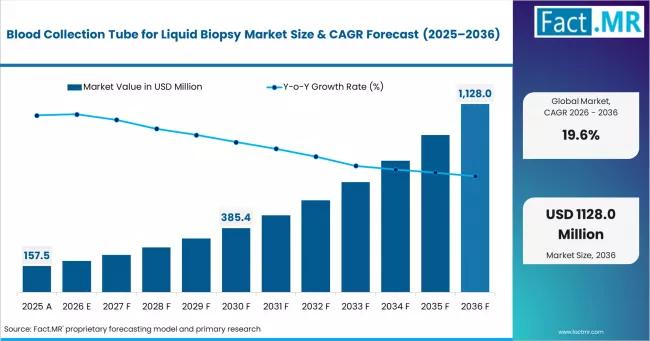
The global blood collection tube for liquid biopsy market is emerging as a critical enabling segment within the rapidly expanding precision diagnostics ecosystem, driven by rising adoption of non-invasive cancer testing, companion diagnostics, and molecular biomarker analysis.According to Fact.MR, the global market for blood collection tubes used in liquid biopsy applications is estimated at USD 188.4 million in 2026 and is forecast to expand at a remarkable 19.6% CAGR, reaching approximately USD 1,128.0 million by 2036. The market includes:
Get Detailed Market Forecasts, Competitive Benchmarking, and Pricing Trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=4495
Quick Stats Snapshot
Market Value (2026): USD 188.4 million
Projected Market Value (2036): USD 1.128 billion
Forecast CAGR (2026-2036): 19.6%
Core Applications: Oncology diagnostics, NIPT, molecular analysis, companion diagnostics
Leading Technologies: cfDNA stabilization, CTC preservation, multi-analyte collection systems
Primary End Users: Hospitals, molecular diagnostic labs, pharmaceutical companies, research institutes
According to Fact.MR, the global market for blood collection tubes used in liquid biopsy applications is estimated at USD 188.4 million in 2026 and is forecast to expand at a remarkable 19.6% CAGR, reaching approximately USD 1,128.0 million by 2036. The market includes:
Cell-free DNA (cfDNA) stabilization tubes
Circulating tumor cell (CTC) collection tubes
Multi-analyte stabilization systems
Molecular analysis blood collection platforms
Growth is being fueled by expanding clinical use of liquid biopsy in oncology, rising FDA-cleared assay approvals, and increasing pharmaceutical investment in biomarker-driven therapies.
Market Size and Strategic Outlook
Blood collection tubes for liquid biopsy are highly specialized specimen collection systems engineered to preserve:
Circulating tumor DNA (ctDNA)
Cell-free DNA (cfDNA)
Circulating tumor cells (CTCs)
RNA biomarkers
Extracellular vesicles
during transport, storage, and molecular analysis.
Unlike conventional blood tubes, these systems are designed to:
Prevent nucleic acid degradation
Minimize white blood cell contamination
Extend sample stability windows
Improve assay reproducibility
These capabilities are becoming increasingly important as liquid biopsy expands into:
Oncology screening
Minimal residual disease (MRD) monitoring
Treatment response assessment
Prenatal diagnostics
Multi-cancer early detection (MCED) platforms.
The market is evolving from a niche research-support segment into a strategic component of precision medicine infrastructure.
Key Growth Drivers
1. Rapid Expansion of Liquid Biopsy in Oncology
Liquid biopsy adoption is accelerating because it offers:
Minimally invasive testing
Real-time tumor monitoring
Repeatable sampling capability
Faster diagnostic workflows
Compared with conventional tissue biopsies, liquid biopsy reduces:
Patient discomfort
Surgical risk
Sampling limitations
Demand is rising rapidly in:
Lung cancer
Breast cancer
Colorectal cancer
Prostate cancer diagnostics.
As assay sensitivity improves, blood collection tube quality is becoming increasingly critical to ensuring accurate biomarker preservation.
2. Growth of Precision Medicine and Companion Diagnostics
Targeted cancer therapies increasingly rely on:
Biomarker-guided treatment selection
Mutation monitoring
Molecular profiling
Pharmaceutical companies are expanding investment in companion diagnostics linked to:
Immunotherapy
Personalized oncology drugs
Genomic sequencing platforms
This directly supports demand for advanced liquid biopsy specimen collection systems.
3. Rising FDA Approvals and Clinical Acceptance
The liquid biopsy ecosystem has seen growing regulatory momentum, including FDA-cleared assays for several cancer indications.
Clinical adoption is increasing across:
Academic cancer centers
Hospital laboratories
Commercial molecular diagnostics providers
Standardized blood collection systems are essential for:
Regulatory compliance
Clinical reproducibility
Laboratory workflow consistency
4. Expansion of Non-Invasive Prenatal Testing (NIPT)
Liquid biopsy collection technologies are also being increasingly used in:
Prenatal genetic testing
Fetal chromosomal screening
Maternal-fetal diagnostics
NIPT adoption is rising globally because of:
Improved diagnostic accuracy
Reduced procedural risk
Growing prenatal healthcare awareness.
Market Challenges
Pre-Analytical Variability Risks
Liquid biopsy testing is highly sensitive to:
Sample handling errors
Temperature exposure
Delayed processing
Biomarker degradation
This creates strong demand for advanced stabilization technologies but also increases product development complexity.
High Validation and Regulatory Requirements
Manufacturers must demonstrate:
Biomarker stability performance
Assay compatibility
Clinical reproducibility
Regulatory compliance
Validation timelines and regulatory costs remain substantial barriers for newer entrants.
Limited Standardization Across Platforms
Differences in:
Collection methods
Preservation chemistries
Assay protocols
can create interoperability challenges between diagnostic platforms and laboratory systems.
Emerging Opportunities
Multi-Cancer Early Detection (MCED)
MCED blood tests represent one of the largest future opportunities in precision oncology.
These platforms aim to detect:
Multiple cancer types
Early-stage disease
Minimal tumor burden
through a single blood draw.
As MCED adoption grows, demand for highly stable blood collection systems is expected to surge.
AI and Machine Learning Integration
Artificial intelligence is increasingly being integrated into:
Biomarker interpretation
ctDNA analysis
CTC identification
Predictive oncology workflows.
Improved sample quality from advanced collection tubes enhances AI model reliability and clinical performance.
Pharmaceutical Clinical Trials and Drug Development
Pharmaceutical companies are increasingly incorporating liquid biopsy into:
Oncology trials
Treatment monitoring
Drug response analysis
Minimal residual disease studies
This is creating strong demand for standardized molecular specimen collection products.
Emerging Market Expansion
Asia-Pacific healthcare systems are rapidly expanding:
Cancer screening infrastructure
Precision medicine programs
Molecular diagnostics laboratories
China, Japan, South Korea, and India are expected to become major growth markets.
Segmentation Insights
By Product Type
cfDNA stabilization tubes: Largest and fastest-growing segment
CTC collection tubes
cfRNA tubes
Multi-analyte stabilization tubes
cfDNA systems dominate because of their central role in oncology liquid biopsy testing.
By Application
Cancer diagnosis and monitoring: Largest segment
Non-invasive prenatal testing (NIPT)
Research applications
Infectious disease diagnostics
Oncology remains the primary revenue driver because of expanding liquid biopsy adoption in precision cancer care.
By End User
Hospitals and diagnostic laboratories
Research institutes
Pharmaceutical and biotechnology companies
Academic medical centers
Large centralized diagnostic laboratories continue to dominate high-volume liquid biopsy processing.
Regional Analysis
North America: Largest Market
The United States leads the market due to:
Strong oncology diagnostics infrastructure
High liquid biopsy adoption
Presence of major diagnostics companies
FDA regulatory advancement.
North America also benefits from:
Large-scale precision medicine investment
High cancer screening expenditure
Strong clinical trial activity
Europe: Regulation and Research Driven
European demand is supported by:
Advanced oncology research networks
Growing liquid biopsy reimbursement
Strong molecular diagnostics adoption
Germany, France, and the United Kingdom remain leading regional markets.
Asia-Pacific: Fastest Growth Region
Asia-Pacific is expected to record the fastest growth due to:
Rising cancer incidence
Expanding biotechnology sectors
Government precision medicine initiatives
Rapid healthcare modernization.
China and Japan are emerging as major innovation hubs for liquid biopsy technologies.
Competitive Landscape
The market remains innovation-intensive, with diagnostics and life sciences companies competing through:
Biomarker stabilization performance
Extended sample stability
Molecular assay compatibility
Automation integration
Key companies include:
Thermo Fisher Scientific
QIAGEN
Roche Diagnostics
Bio-Rad Laboratories
Streck
Greiner Bio-One
Competition increasingly centers on:
Biomarker preservation quality
Clinical validation
Multi-analyte compatibility
Global regulatory compliance
Strategic Implications for Industry Stakeholders
Invest in Advanced Stabilization Technologies: Sample integrity remains the core competitive differentiator
Align with Precision Oncology Ecosystems: Companion diagnostics partnerships will drive long-term growth
Expand Regulatory and Clinical Validation: FDA-aligned systems will gain stronger market positioning
Strengthen Pharma and Lab Partnerships: Integrated workflow compatibility is becoming essential
Future Outlook
The blood collection tube for liquid biopsy market is expected to become a foundational component of the next-generation diagnostics infrastructure over the next decade.
Industry evolution will likely focus on:
Multi-cancer early detection support
AI-enabled molecular diagnostics
Universal biomarker stabilization platforms
Home-based and decentralized testing workflows
Integrated precision medicine ecosystems
As oncology increasingly shifts toward non-invasive, data-driven disease management, blood collection technologies will become strategically important not only for diagnostics companies, but also for pharmaceutical developers, healthcare providers, and precision medicine platforms globally.
Browse Full Report: https://www.factmr.com/report/4495/blood-collection-tubes-for-liquid-biopsy-market
Executive Takeaways:
The market is projected to grow from USD 188.4 million in 2026 to USD 1.128 billion by 2036, at a robust 19.6% CAGR.
Growth is driven by precision oncology, liquid biopsy adoption, FDA-cleared assays, and companion diagnostics expansion.
cfDNA stabilization tubes remain the leading product category.
North America leads the market, while Asia-Pacific represents the fastest-growing regional opportunity.
Competitive advantage increasingly depends on sample stabilization quality, assay compatibility, regulatory validation, and integration into precision medicine workflows.
Unlock 360° Insights for Strategic Decision Making and Investment Planning : https://www.factmr.com/checkout/4495
Related Reports:
Blood Thawing System Market: https://www.factmr.com/report/blood-thawing-system-market
Blood Glucose Testing Devices Market: https://www.factmr.com/report/2681/blood-glucose-testing-devices-market
Blood Testing Devices Market: https://www.factmr.com/report/2680/blood-testing-devices-market
Blood-based Biomarker for Sports Medicine Market: https://www.factmr.com/report/blood-based-biomarker-for-sports-medicine-market
Contact:
US Sales Office
11140 Rockville Pike
Suite 400
Rockville, MD 20852
United States
Tel: +1 (628) 251-1583, +353-1-4434-232
Email: sales@factmr.com
About Fact.MR
We are a trusted research partner of 80% of fortune 1000 companies across the globe. We are consistently growing in the field of market research with more than 1000 reports published every year. The dedicated team of 400-plus analysts and consultants is committed to achieving the utmost level of our client's satisfaction.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Blood Collection Tube for Liquid Biopsy Market Records 19.6% CAGR Growth Outlook, Supported by Streck, QIAGEN, Greiner Bio-One here
News-ID: 4505636 • Views: …
More Releases from Fact.MR
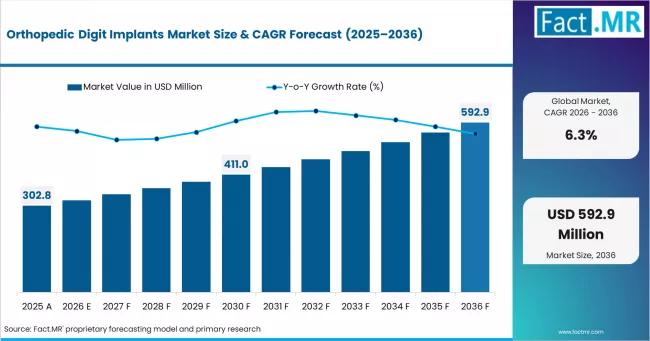
Orthopedic Digit Implants Market Climbs to USD 321.9 Million in 2026, Led by Str …
The global orthopedic digit implants market is gaining strategic importance within the broader orthopedic device industry as healthcare systems increasingly prioritize mobility restoration, minimally invasive extremity procedures, and functional joint preservation.
According to Fact.MR, the orthopedic digit implants market was valued at USD 321.9 million in 2026 and is projected to reach USD 592.9 million by 2036, reflecting sustained long-term expansion across developed and emerging healthcare systems.
The market is being…
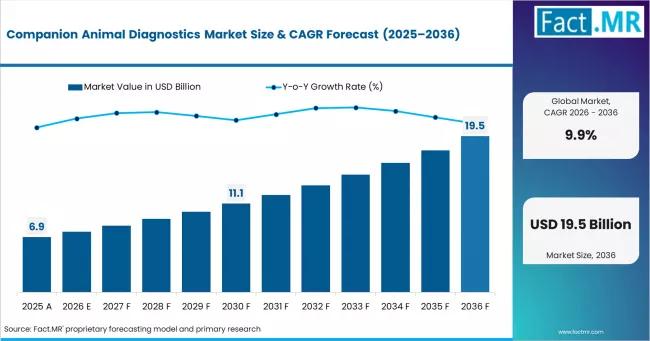
Companion Animal Diagnostics Market Valued at USD 7.6 Billion in 2026, Strengthe …
The global companion animal diagnostics market is rapidly evolving from a niche veterinary support segment into a critical pillar of the broader animal healthcare ecosystem, driven by rising pet ownership, preventive healthcare adoption, and growing demand for precision veterinary medicine.
According to Fact.MR, the companion animal diagnostics market was valued at approximately USD 7.6 billion in 2026 and is expected to grow steadily through 2036. Industry forecasts indicate strong long-term expansion…
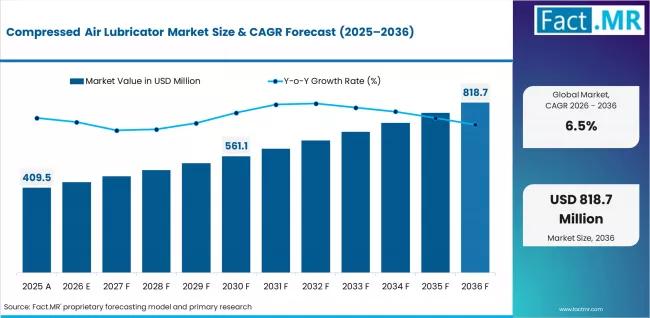
Compressed Air Lubricator Industry Projected to Grow at 6.5% CAGR, Driven by SMC …
The global compressed air lubricator market is entering a new phase of industrial relevance as manufacturers intensify investments in automation, pneumatic efficiency, and predictive maintenance infrastructure. Once considered a standard mechanical component, compressed air lubricators are increasingly being repositioned as critical reliability systems within automated manufacturing environments.
According to Fact.MR, the compressed air lubricator market was valued at USD 409.5 million in 2025 and is projected to grow to USD 818.7…
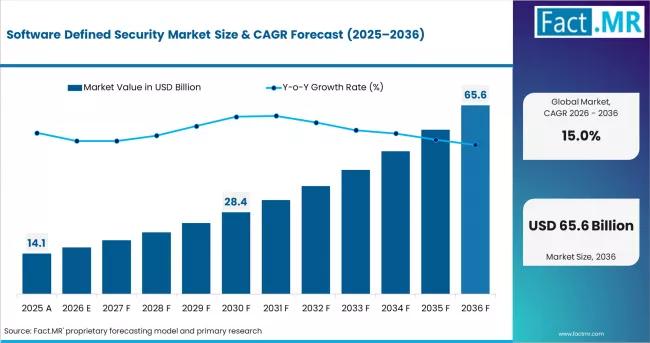
Software Defined Security Market to Reach USD 14.1 Billion in 2026, Led by Cisco …
The global software defined security (SDSec) market is moving from a specialized infrastructure security concept into a core enterprise cybersecurity architecture strategy, as organizations confront escalating cyber threats, hybrid cloud complexity, and the operational limitations of hardware-centric security models.
According to Fact.MR, the software defined security market was valued at USD 12.4 billion in 2025 and is projected to grow to USD 14.1 billion in 2026 and USD 57.3 billion by…
More Releases for DNA
High-Quality Plasmid DNA Fuels Growth in Global DNA Plasmid Manufacturing Market
🌍 Market Overview
The DNA Plasmid Manufacturing Market is experiencing robust growth as advancements in cell & gene therapy, DNA vaccines, and genetic engineering continue to expand globally. Plasmid DNA plays a critical role as a raw material in the development of advanced therapies, fueling demand across biopharmaceutical research and production.
Key factors driving the market include:
Increasing adoption of gene and cell therapies
Rising prevalence of chronic and rare genetic disorders
Expansion of DNA-based…
DNA Synthesis Market Increasing Demand for Synthetic Genes and DNA Sequences
As demonstrated by Precision Business Insights (PBI), the latest report, the global DNA synthesis market was valued at USD 3,702.0 million in 2023 and is expected to reach USD 10,289.5 million by 2029, growing at a CAGR of 18.6% during the forecast period 2024-2030. The key drivers for the growth of the global DNA synthesis market include increasing demand for synthetic genes and DNA sequences, growing applications in the agriculture…
Wealth DNA Code Review Legit Price? (Wealth Manifestation DNA Code Audio Frequen …
Wealth DNA Code Wealth DNA Code is a digital program with seven minutes of soundtracks that manifest and listen to daily to activate the "Wealth DNA," which is part of your DNA to help you attract wealth by making money a part of your mentality and making your dreams to come true.
https://bit.ly/Visit-The-Official-Website-Here-To-Order-Wealth-DNA-Code
Making money, creating assets as well as increasing wealth are the primary objectives that every human being has to…
DNA Paternity Testing Market Size [2022-2029] -DNA Diagnostics Center, EasyDNA, …
A recent market research report added to repository of MR Accuracy Reports is an in-depth analysis of global DNA Paternity Testing. On the basis of historic growth analysis and current scenario of DNA Paternity Testing place, the report intends to offer actionable insights on global market growth projections. Authenticated data presented in report is based on findings of extensive primary and secondary research. Insights drawn from data serve as excellent…
DNA Paternity Testing Market Trends 2020 | Growth by Top Companies: DNA Diagnost …
The report begins with the overview of the DNA Paternity Testing Market and offers throughout development. It presents a comprehensive analysis of all the regional and major player segments that gives closer insights upon present market conditions and future market opportunities along with drivers, trending segments, consumer behaviour, pricing factors and market performance and estimation. The forecast market information, SWOT analysis, DNA Paternity Testing market scenario, and feasibility study are…
DNA Paternity Testing Market Rapidly Growing in Healthcare, Competitor Analysis …
The exclusive research report on the Global DNA Paternity Testing Market 2020 examines the market in detail along with focusing on significant market dynamics for the key players operating in the market. Global DNA Paternity Testing Industry research report offers granulated yet in-depth analysis of revenue share, market segments, revenue estimates and various regions across the globe.
Overview of Global DNA Paternity Testing Market:
This report studies the Global DNA Paternity Testing…