Press release
Pharma Packaging Enters the Zero-Defect Era: Checkweighers with Rejection Systems Become Mission-Critical Infrastructure
In pharmaceutical manufacturing, the margin for error is effectively zero. A single missing tablet in a blister pack can trigger recalls, regulatory action, and reputational damage. Against this backdrop, the Checkweighers with Rejection Systems for Pharma Blister Lines Market is witnessing a critical shift, as these systems evolve from auxiliary inspection tools into core infrastructure for compliance, quality assurance, and operational continuity.Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=14713
Quick Stats Snapshot
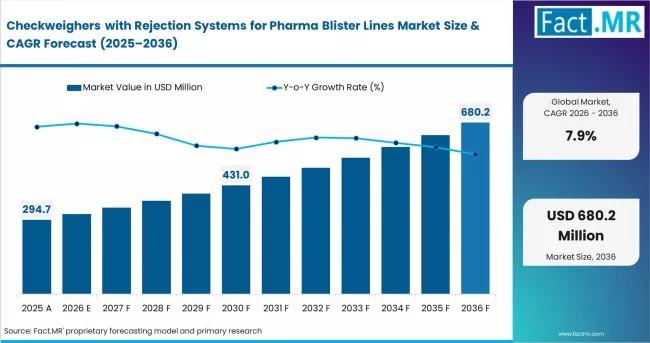
Market Size (2026): USD 294.7 million
Forecast (2036): USD 628.9 million
CAGR (2026-2036): 7.9%
Leading Application: Tablet blister packs (~41% share)
Dominant Speed Range: Up to 200 packs/min (~36%)
Market Size and Forecast: Steady Expansion Anchored in Compliance
The global market for checkweighers with rejection systems in pharma blister lines is projected to more than double over the next decade, growing from USD 294.7 million in 2026 to USD 628.9 million by 2036.
This growth reflects a structurally stable but regulation-driven market, where adoption is less cyclical and more tied to compliance mandates, validation requirements, and packaging line upgrades.
Growth Drivers: Compliance, Automation, and Zero-Error Expectations
Regulatory Enforcement Tightens
Pharmaceutical packaging is governed by strict frameworks such as cGMP and FDA 21 CFR standards. These require validated, automated inspection systems to ensure dosage accuracy and packaging integrity.
Serialization and Traceability Expansion
With global serialization mandates expanding, manufacturers must ensure that every unit is not only traceable but also physically accurate in content and weight-driving demand for inline verification systems.
Shift from Sampling to 100% Inspection
Manual sampling is increasingly inadequate in high-speed environments. Checkweighers enable real-time, continuous inspection, eliminating reliance on batch-based quality checks.
High-Speed Manufacturing Demands
Modern blister lines operate at high throughput, requiring synchronized systems that can detect and reject defective packs without disrupting flow.
Challenges: Capital Intensity and Integration Complexity
Despite strong adoption drivers, several constraints remain:
High upfront investment: Advanced checkweighing systems with integrated rejection mechanisms require significant capital expenditure.
Validation complexity: Systems must meet stringent pharmaceutical validation protocols, increasing implementation timelines.
Operational sensitivity: Factors like vibration, conveyor instability, and calibration drift can affect measurement accuracy.
Integration with legacy systems: Retrofitting older packaging lines can be technically challenging and costly.
Opportunities: Smart Inspection and Digital Integration
The next phase of growth lies in intelligent, connected inspection systems:
Integration with electronic batch records (EBR): Enhances audit readiness and traceability
AI-driven anomaly detection: Improves detection of subtle defects beyond weight deviations
IoT-enabled monitoring: Enables predictive maintenance and performance optimization
Multi-parameter inspection systems: Combining weight, vision, and serialization verification
These innovations are transforming checkweighers into data-generating assets, not just compliance tools.
Segmentation Insights: Tablets and Mid-Speed Lines Lead
By Application
Tablet blister packs dominate (41%), reflecting the scale of solid oral dosage manufacturing.
Capsule and combination packs follow, with growing demand in specialty pharmaceuticals.
By Speed Capacity
Up to 200 packs/min leads (36%), indicating widespread use in standardized production environments.
Higher-speed systems are gaining traction in large-scale facilities.
By Rejection Type
Air blast and pusher systems are widely adopted for fast, precise removal of defective units.
Regional Analysis: Asia-Pacific Drives Growth, Europe Sets Standards
Asia-Pacific: Fastest-growing region, led by China (8.8% CAGR) and India (8.5%), fueled by expanding pharmaceutical manufacturing and automation adoption.
Europe: A compliance-driven market with strong adoption due to stringent regulatory frameworks and mature pharma infrastructure.
North America: Growth is steady but mature, driven largely by system upgrades and replacements.
Asia-Pacific is increasingly becoming both a production hub and a demand center for advanced inspection technologies.
Competitive Landscape: Precision Engineering Meets Compliance Expertise
The market is moderately concentrated, with key players including:
Mettler Toledo International Inc.
WIPOTEC OCS GmbH
Ishida Co., Ltd.
Anritsu Corporation
Thermo Fisher Scientific Inc.
Minebea Intec GmbH
Bizerba SE & Co. KG
Competition is defined by:
Measurement accuracy and stability
Rejection precision and speed
Validation support and compliance readiness
Integration capability with packaging lines
Pharma manufacturers often adopt multi-vendor strategies to mitigate risk and ensure system redundancy.
Strategic Implications for Industry Stakeholders
For Pharmaceutical Manufacturers
Invest in inline inspection systems to reduce recall risk and ensure compliance
Align packaging lines with serialization and digital quality systems
For Equipment Suppliers
Focus on high-precision load cell technology and system integration capabilities
Develop solutions compatible with smart manufacturing ecosystems
For Investors
This is a defensive growth market, anchored in regulation rather than discretionary demand
Opportunities lie in automation, AI integration, and digital inspection platforms
Future Outlook: Toward Fully Autonomous Quality Control
The market is moving toward a future where packaging lines are self-monitoring, self-correcting, and fully compliant by design.
Key trends shaping the next decade:
Convergence of weighing, vision, and serialization technologies
Increased adoption of AI-driven quality control systems
Standardization of digital validation and audit processes
Expansion of continuous manufacturing models
Checkweighers with rejection systems will evolve into central nodes in pharmaceutical quality ecosystems, not just inspection endpoints.
Executive Takeaways
The market will grow from USD 294.7M (2026) to USD 628.9M (2036) at 7.9% CAGR
Regulatory compliance and zero-error packaging are the primary growth drivers
Tablet blister packaging dominates demand due to volume and sensitivity
Asia-Pacific is the fastest-growing region; Europe leads in compliance standards
Future growth will be driven by smart, connected, and AI-enabled inspection systems
In a pharmaceutical landscape defined by precision, traceability, and accountability, checkweighers with rejection systems are no longer just equipment-they are critical control points ensuring product integrity and patient safety at scale.
Browse Full Report - https://www.factmr.com/report/checkweighers-with-rejection-systems-for-pharma-blister-lines-market
Unlock 360° insights for strategic decision making and investment planning: https://www.factmr.com/checkout/14713
To View Related Report:
Toilet Roll Converting Lines Market https://www.factmr.com/report/toilet-roll-converting-lines-market
Semi-Auto Flex Filling Lines Market https://www.factmr.com/report/semi-auto-flex-filling-lines-market
Drone Pharmacy Delivery Market https://www.factmr.com/report/drone-pharmacy-delivery-market
Forklift Market https://www.factmr.com/report/forklift-market
- Contact Us -
11140 Rockville Pike, Suite 400, Rockville,
MD 20852, United States
Tel: +1 (628) 251-1583 | sales@factmr.com
About Fact.MR
Fact.MR is a global market research and consulting firm, trusted by Fortune 500 companies and emerging businesses for reliable insights and strategic intelligence. With a presence across the U.S., UK, India, and Dubai, we deliver data-driven research and tailored consulting solutions across 30+ industries and 1,000+ markets. Backed by deep expertise and advanced analytics, Fact.MR helps organizations uncover opportunities, reduce risks, and make informed decisions for sustainable growth.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Pharma Packaging Enters the Zero-Defect Era: Checkweighers with Rejection Systems Become Mission-Critical Infrastructure here
News-ID: 4499095 • Views: …
More Releases from Fact.MR
Biodegradable Cut Flower Packaging Market: From Regulatory Burden to Strategic D …
The global floriculture supply chain is being reshaped by a material shift that extends far beyond packaging. Biodegradable cut flower packaging-once viewed as a sustainability add-on-is now emerging as a structural requirement driven by regulation, retailer mandates, and evolving consumer expectations. As plastic-intensive floral logistics face mounting scrutiny, biodegradable alternatives are redefining procurement strategies, cost structures, and competitive positioning across the industry.
Get detailed market forecasts, competitive benchmarking, and pricing trends:…

Clean Label Food Ingredient Industry Surges at 6.2% CAGR Through 2032, Powered b …
The global clean label food ingredient market is transitioning from a consumer-driven trend into a structural transformation of the food industry, redefining how products are formulated, marketed, and regulated. Clean label is no longer a niche positioning-it has become a baseline expectation, compelling manufacturers to replace synthetic additives with recognizable, natural, and minimally processed ingredients.
This shift is reshaping supply chains, R&D priorities, and competitive dynamics across food, beverage, and nutrition…

Azodicarbonamide Market Records 8.0% CAGR Growth Outlook with Kum Yang, Honeywel …
The global azodicarbonamide (ADA) market is evolving within a complex dual-demand structure, where industrial applications continue to expand while food-related usage faces increasing regulatory and consumer scrutiny. Widely used as both a chemical blowing agent in plastics and a dough conditioner in baking, azodicarbonamide sits at the intersection of materials science and food processing.
While Fact.MR projects the market to expand at a 5.6% CAGR by 2027, the underlying growth dynamics…

Fat Soluble Filling Market to Expand at 6.8% CAGR Through 2036, Led by Cargill, …
The global fat-soluble filling market is emerging as a specialized but strategically relevant segment within the bakery and confectionery ingredient ecosystem, driven by evolving consumer preferences for indulgent, high-quality baked products and the expansion of organized bakery retail.
These fillings-comprising fat-compatible ingredients such as chocolate-based compounds, flavored fats, and specialty flour blends-play a critical role in texture, flavor delivery, and shelf stability in baked goods, making them essential for both artisanal…
More Releases for Pharma
Schizophrenia Market: Expanding Revenue Landscape to 2034 - DelveInsight | Teva …
The Key Schizophrenia Companies in the market inlclude - Teva Pharmaceutical Industries, Royalty Pharma, MedinCell, Sumitomo Pharma, Otsuka Pharmaceuticals, Reviva Pharmaceuticals, Minerva Neurosciences, Mitsubishi Tanabe Pharma, Lundbeck, Intra-cellular Therapies, Sumitomo Pharma, Otsuka Pharma, Reviva Pharmaceuticals, and others.
DelveInsight's report "Schizophrenia Market Insights, Epidemiology, and Market Forecast-2034" provides a comprehensive analysis of the Schizophrenia landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the…
Dacarbazine Market Size, Share and Forecast By Key Players-Lingnan Pharma, Ruiyi …
𝐔𝐒𝐀, 𝐍𝐞𝐰 𝐉𝐞𝐫𝐬𝐞𝐲- According to the MRI Team's Market Research Intellect, the global Dacarbazine market is anticipated to grow at a compound annual growth rate (CAGR) of 14.63% between 2024 and 2031. The market is expected to grow to USD 25.32 Billion by 2024. The valuation is expected to reach USD 65.84 Billion by 2031.
The Dacarbazine market is projected to witness significant growth in the coming years, driven by increasing…
Dacarbazine Market Size, Share and Forecast By Key Players-Lingnan Pharma, Ruiyi …
𝐔𝐒𝐀, 𝐍𝐞𝐰 𝐉𝐞𝐫𝐬𝐞𝐲- According to the MRI Team's Market Research Intellect, the global Dacarbazine market is anticipated to grow at a compound annual growth rate (CAGR) of 14.63% between 2024 and 2031. The market is expected to grow to USD 25.32 Billion by 2024. The valuation is expected to reach USD 65.84 Billion by 2031.
The dacarbazine market is experiencing steady growth, driven by the rising incidence of cancer globally. As…
Epilepsy Pipeline Assessment 2024: Therapies, Clinical Trials, and Market Insigh …
(Las Vegas, Nevada, United States) As per DelveInsight's assessment, globally, Epilepsy pipeline constitutes 75+ key companies continuously working towards developing 90+ Epilepsy treatment therapies, analysis of Clinical Trials, Therapies, Mechanism of Action, Route of Administration, and Developments analyzes DelveInsight.
The Epilepsy Pipeline report embraces in-depth commercial and clinical assessment of the pipeline products from the pre-clinical developmental phase to the marketed phase. The report also covers a detailed description of the…
Global Indomethacin Market Seeking Excellent Growth by 2028 | Ratiopharm, Merck, …
"IBI has published a report entitled Global Indomethacin Market Research Report which provides comprehensive data on emerging trends, market drivers, growth opportunities, and barriers that could change the dynamics of the industry market. It provides an in-depth analysis of market segments which includes the product, application, and competitor analysis.
Click Here To Order A Sample Copy Of Indomethacin Global Market Report : https://www.infinitybusinessinsights.com/request_sample.php?id=380382
The Indomethacin Global Market Research Report provides close monitoring…
Roxatidine Market 2020: Top Companies Analysis To Grow Healthcare Business by 20 …
Business Industry Reports Research has recently announced a report on Global Roxatidine Market based on the Category Industry. The Roxatidine Market report emphasizes various key aspects, which include growth drivers, restraints, opportunities and recent market trends for the forecast period 2020-2024.
Global Roxatidine Market overview:
Business Industry Reports Analyst covers the Major Players data, including: shipment, revenue, gross profit, interview record, business distribution etc., these data help the consumer know about the…