Press release
Global Preclinical Antibody Development Market to Reach USD 9.7 Billion by 2036 at 9.3% CAGR Driven by Biologics Innovation
The journey from a computational antibody design to a clinical-grade therapeutic has long been dubbed the "Valley of Death," where promising candidates often wither due to unforeseen toxicity or poor developability. However, a fundamental shift is occurring. According to the latest analysis by Fact.MR, the global preclinical antibody development market is poised for a decade of high-stakes transformation. As drug pipelines shift toward increasingly complex modalities such as bispecifics, antibody-drug conjugates (ADCs), and single-domain antibodies the demand for rigorous, integrated preclinical characterization has moved from a "check-the-box" regulatory requirement to a critical strategic moat for biopharmaceutical success.Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=14111
Quick Stats: Market at a Glance
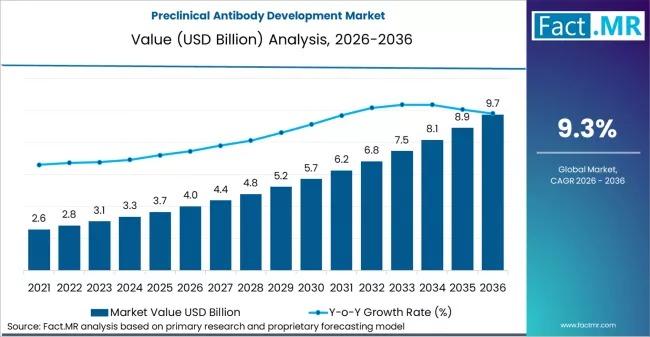
Estimated Market Value (2026E): US$ 4.0 Billion
Projected Market Value (2036F): US$ 9.7 Billion
Projected CAGR (2026-2036):3%
High-Growth Region: China (Projected 11.8% CAGR)
Key Growth Catalyst: FDA Modernization Act 2.0 and the shift toward non-animal testing.
Market Size and Growth Forecast
The preclinical antibody development sector is entering a period of accelerated expansion. Valued at an estimated US$ 4.0 billion in 2026, the market is projected to reach US$ 9.7 billion by 2036. This nearly 2.5x growth reflects a broader industry trend: the "fail fast, fail early" philosophy. Investors and large pharma are no longer satisfied with simple binding data; they require comprehensive "developability" profiles including aggregation risk, stability, and off-target effects before committing the hundreds of millions required for Phase I trials.
Strategic Growth Drivers: Beyond Monoclonals
The market is being propelled by three distinct forces:
Complexity of New Modalities: Conventional monoclonal antibodies are becoming the "low-hanging fruit." The next generation of therapeutics involves multi-specific antibodies that target two or more epitopes simultaneously. These require far more sophisticated preclinical validation of binding kinetics and functional synergy.
Regulatory Modernization: The FDA Modernization Act 2.0 has fundamentally changed the landscape by allowing for human-relevant, non-animal testing strategies. This has sparked a massive surge in demand for specialized preclinical services involving organ-on-a-chip, microphysiological systems (MPS), and patient-derived organoids.
The AI Validation Bottleneck: Artificial Intelligence has flooded the discovery phase with thousands of in silico This has created a physical bottleneck in the real world: these digital designs must be expressed, purified, and functionally validated in a wet lab. Preclinical service providers are the "physical layer" that translates AI potential into clinical reality.
Navigating Challenges: Cost and Standardization
Despite the optimistic outlook, the market faces structural hurdles. The rising complexity of preclinical programs has made them prohibitively expensive for early-stage startups, often forcing them to limit the scope of characterization. Furthermore, there is a glaring lack of data standardization across the industry. Discrepancies in assay platforms and modeling systems can lead to conflicting results when a program is transferred from a small CRO to a large-scale CDMO, creating regulatory friction and delaying timelines.
Segmentation Insights: Oncology Remains the Engine
By Service Type: Antibody discovery (via phage display and hybridoma) remains the largest segment, but developability assessment and humanization/engineering are the fastest-growing niches.
By Therapeutic Area:Oncology continues to dominate, accounting for the lion's share of preclinical spend. However, there is significant uptick in Autoimmune and Neurology as researchers explore the potential of antibodies to cross the blood-brain barrier.
By End User: While large pharma provides steady volume, Biotech Startups are the primary drivers of innovation, often relying on "virtual" models where 100% of preclinical work is outsourced to integrated service providers.
Regional Analysis: The Rise of Asia-Pacific
While North America remains the dominant hub due to its robust biomedical infrastructure, the Asia-Pacific region specifically China (11.8% CAGR) and India (11.2% CAGR) is seeing the highest growth rates. China has evolved from a low-cost service provider into a global leader in innovative antibody platforms, fueled by proactive government funding and a massive patient pool for translational research.
Competitive Landscape: The Shift to Integrated Platforms
The competitive environment is shifting away from fragmented, "boutique" service providers toward integrated, end-to-end platforms. Major players such as Charles River Laboratories, WuXi Biologics, and Eurofins are aggressively acquiring niche firms to offer a "discovery-to-IND" (Investigational New Drug) workflow.
Key Industry Players: Lonza, Thermo Fisher Scientific, Labcorp Drug Development, Samsung Biologics, and GenScript.
Trend: Strategic partnerships between AI-driven discovery firms and preclinical CROs are becoming the new standard for accelerating drug development timelines.
Future Outlook & Executive Takeaways
The next decade will see the preclinical phase become the most data-intensive part of the drug development lifecycle. For C-level executives and investors, the implications are clear:
Prioritize Developability: Success is no longer just about target affinity; it is about how a molecule behaves in a manufacturing bioreactor and a human body.
Outsource Strategically: Look for partners that offer integrated workflows rather than siloed services to minimize "tech-transfer" risks.
Invest in New Models: The shift away from animal testing is not just a trend it is a regulatory reality. Investing in human-relevant preclinical models (MPS, Organoids) is now a prerequisite for long-term clinical success.
In an era where the average cost of drug development continues to climb, the preclinical antibody development market stands as the critical filter that will determine which of today's scientific breakthroughs become tomorrow's blockbuster therapies.
Unlock 360° insights for strategic decision making and investment planning: https://www.factmr.com/checkout/14111
To View Our Related Report:
Preclinical Brain Imaging Market: https://www.factmr.com/report/2562/preclinical-brain-imaging-market
Preclinical CRO Market: https://www.factmr.com/report/preclinical-cro-market
Optical Preclinical Imaging Market: https://www.factmr.com/report/optical-preclinical-imaging-market
Antibody Conjugation Kits Market: https://www.factmr.com/report/antibody-conjugation-kits-market
- Contact Us -
11140 Rockville Pike, Suite 400, Rockville,
MD 20852, United States
Tel: +1 (628) 251-1583 | sales@factmr.com
Fact.MR is a global market research and consulting firm, trusted by Fortune 500 companies and emerging businesses for reliable insights and strategic intelligence. With a presence across the U.S., UK, India, and Dubai, we deliver data-driven research and tailored consulting solutions across 30+ industries and 1,000+ markets. Backed by deep expertise and advanced analytics, Fact.MR helps organizations uncover opportunities, reduce risks, and make informed decisions for sustainable growth.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Preclinical Antibody Development Market to Reach USD 9.7 Billion by 2036 at 9.3% CAGR Driven by Biologics Innovation here
News-ID: 4496326 • Views: …
More Releases from Fact.MR
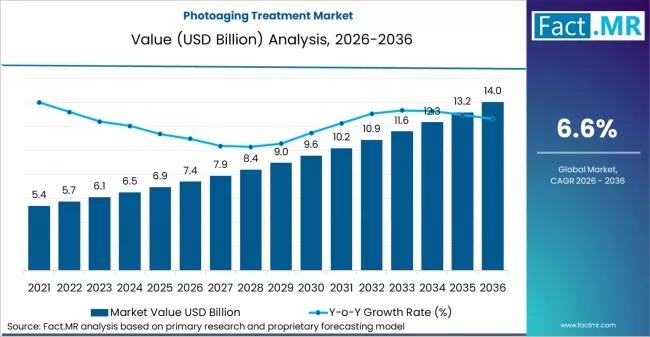
Global Photoaging Treatment Market to Reach USD 14 Billion by 2036 at 6.6% CAGR …
The global aesthetic landscape is undergoing a fundamental shift from reactive "anti-aging" to proactive "photo-repair." As cumulative UV exposure becomes a primary concern for an aging global population, the photoaging treatment market is evolving into a high-tech ecosystem of biologics, energy-based devices, and AI-guided diagnostics. For C-suite executives and investors, the sector represents more than just vanity; it is a high-growth intersection of medical dermatology and retail wellness. According to…
Global Skin Microbiome Market to Reach USD 28.639 Billion by 2036 at 7.9% CAGR, …
The global beauty and personal care industry is undergoing a fundamental shift from "sterilization" to "symbiosis." No longer viewed as a surface to be scrubbed clean, the skin is increasingly understood as a living ecosystem.
According to the latest analysis by Fact.MR, the global Skin Microbiome Market is poised for a transformative decade, projected to grow from a USD 12.409 billion valuation in 2025 to a staggering USD 28.639 billion by…
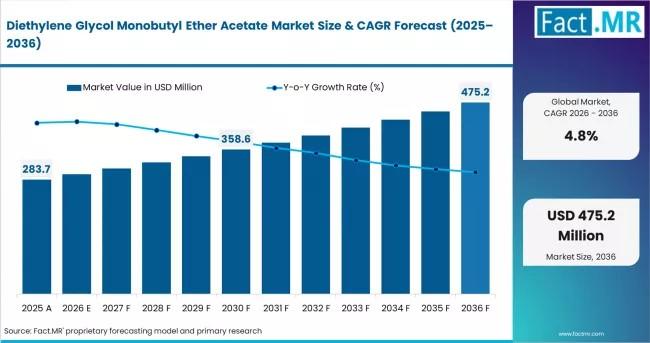
High-Performance Solvency: The Strategic Role of Diethylene Glycol Monobutyl Eth …
In the complex ecosystem of specialty solvents, certain compounds are valued not for their volatility, but for their precision. Diethylene Glycol Monobutyl Ether Acetate (DCAC) is a high-boiling, slow-evaporating ester that has become an indispensable "flow and leveling" agent for the global coatings and printing industries. As industrial manufacturing moves toward high-resolution digital printing and ultra-durable automotive finishes, the demand for solvents that can manage drying times without sacrificing film…
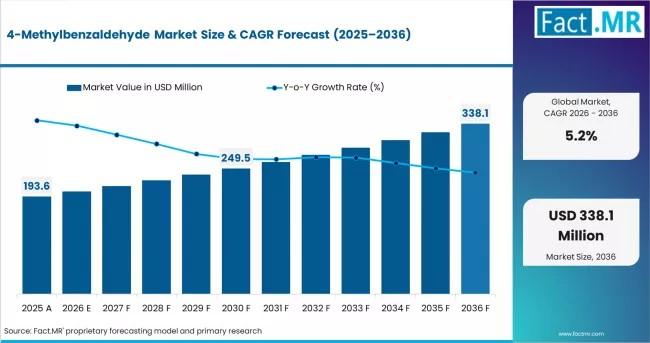
4-Methylbenzaldehyde: The Versatile Intermediate Powering Specialty Chemical Inn …
In the intricate supply chain of specialty chemicals, certain molecules act as "invisible anchors"-essential building blocks that bridge the gap between raw petrochemicals and high-value consumer end-products. 4-Methylbenzaldehyde (also known as p-Tolualdehyde) is one such pivot point. With its distinct cherry-like aroma and reactive aldehyde functionality, it has evolved from a simple fragrance additive into a critical intermediate for the global pharmaceutical, agrochemical, and polymer additive sectors. As industries demand…
More Releases for Preclinical
Rising Chronic Disease Burden Fuels Preclinical Imaging Market Growth Emerges as …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
What Is the Expected CAGR for the Preclinical Imaging Market Through 2025?
In the past few years, the preclinical imaging market has witnessed a significant increase in its size. The market is projected to surge from $3.46 billion in 2024 to $3.69 billion in 2025, recording a compound annual…
Driving Preclinical CRO Market Growth in 2025: The Role of Surging Demand For Pr …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
Preclinical CRO Market Size Growth Forecast: What to Expect by 2025?
In recent times, there has been a significant increase in the size of the preclinical CRO market. The market will develop from being worth $5.7 billion in 2024 to achieving a value of $6.26 billion the following year,…
DLL3 Inhibitors in Preclinical and Clinical Studies
Delta-Like Ligand 3 (DLL3) inhibitors have shown significant promise in preclinical and clinical studies, particularly for the treatment of aggressive cancers such as small cell lung cancer (SCLC) and neuroendocrine tumors. The selective expression of DLL3 in these malignancies makes it an attractive target for therapeutic intervention. This article explores the development and progress of DLL3 inhibitors in preclinical and clinical studies, highlighting their potential in modern oncology.
Download Report:
https://www.kuickresearch.com/report-dll3-targeted-therapies-dll3-cancer-therapies-dll3-protien-dll3-cancer-drugs-delta-like-ligand-3-dll3-gene-dll3-expression-dll3-amgen-dll3-inhibitor
DLL3…
Preclinical Imaging Market Opportunity | Size | Research Report
𝐔𝐒𝐀, 𝐍𝐞𝐰 𝐉𝐞𝐫𝐬𝐞𝐲- Preclinical Imaging Market was valued at USD 748.18 Million in 2023 and is projected to reach USD 1,109.10 Million by 2030, growing at a CAGR of 5.0% from 2024 to 2030.
The preclinical imaging market is being significantly driven by advancements in imaging modalities, such as MRI, PET, SPECT, CT, and optical imaging. These technologies provide researchers with non-invasive ways to visualize and study biological processes at the…
Preclinical Imaging Market - Transforming Research with Precision Imaging: Precl …
Newark, New Castle, USA: The "Preclinical Imaging Market" provides a value chain analysis of revenue for the anticipated period from 2023 to 2031. The report will include a full and comprehensive analysis of the business operations of all market leaders in this industry, as well as their in-depth market research, historical market development, and information about their market competitors
Preclinical Imaging Market: https://www.growthplusreports.com/report/preclinical-imaging-market/7673
This latest report researches the industry structure, sales, revenue,…
Preclinical Imaging Market - Unlocking Clarity, Preserving Scientific Excellence …
Newark, New Castle, USA - new report, titled Preclinical Imaging Market The report has been put together using primary and secondary research methodologies, which offer an accurate and precise understanding of the Preclinical Imaging market. Analysts have used a top-down and bottom-up approach to evaluate the segments and provide a fair assessment of their impact on the global Preclinical Imaging market. The report offers an overview of the market, which…