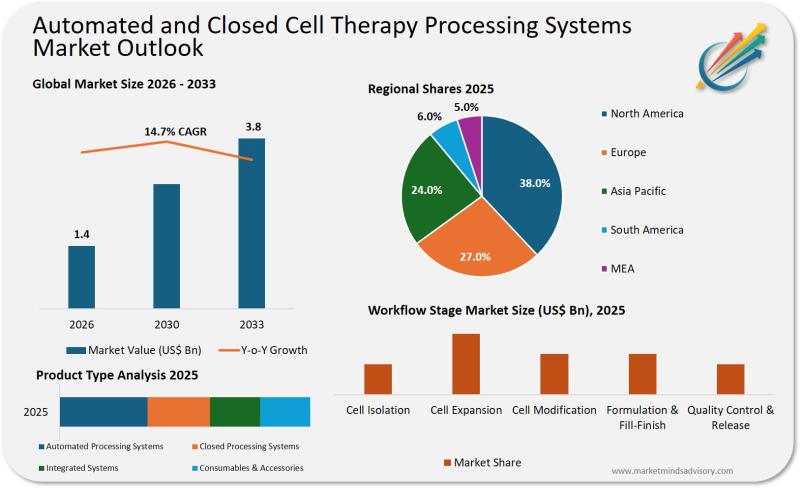
Press release
Cell Therapy Manufacturing Is Hitting a Scalability Wall And Most Companies Are Unprepared
The promise of personalized medicine is colliding with industrial reality. As CAR-T therapies and regenerative treatments move from clinical trials to commercial scale, manufacturers are discovering that manual processing methods cannot support the volume, consistency, or economics required for mass adoption.Why Manual Processing Is No Longer Viable
Cell therapy manufacturing today resembles artisan production in an industry demanding automotive-scale precision. Each batch requires specialized cleanroom environments, highly trained personnel, and weeks of processing time. The result is a bottleneck that limits patient access, inflates costs to unsustainable levels, and introduces variability that regulators increasingly view as unacceptable.
The economics are stark. Manual CAR-T production can cost upwards of $400,000 per patient, with labor representing nearly 40% of total manufacturing expenses. Contamination rates in open systems range between 2-5%, and each failure means not just financial loss but delayed treatment for critically ill patients. As approval pipelines expand beyond oncology into autoimmune disorders, neurological conditions, and genetic diseases, the gap between therapeutic potential and manufacturing capacity is widening dangerously.
Companies that continue relying on legacy processing methods face a compounding risk: they are building commercial strategies on infrastructure that cannot scale. The question is no longer whether automation will dominate cell therapy manufacturing, but how quickly laggards will be priced out of the market.
Request Report Sample: https://marketmindsadvisory.com/request-sample/?report_id=8388
Structural Shifts Driving the Market
Regulatory Pressure Is Forcing Standardization
Global health authorities are tightening manufacturing standards as cell therapies transition from experimental treatments to mainstream medicine. The FDA's recent guidance on Chemistry, Manufacturing, and Controls (CMC) for cellular therapies explicitly favors closed, automated systems that minimize human intervention. European regulators are following suit, creating a compliance environment where manual processing increasingly represents regulatory risk rather than flexibility.
This shift is not merely procedural. Regulators are demanding reproducibility data that open systems struggle to provide. Automated platforms generate digital audit trails, real-time monitoring, and batch-to-batch consistency that manual processes cannot match. For companies seeking approval in multiple jurisdictions, closed systems are becoming the only viable path to global commercialization.
Economic Realities Are Reshaping Investment Priorities
The cell therapy sector attracted over $18 billion in investment capital last year, but investors are becoming more discriminating about manufacturing strategies. Due diligence now routinely includes assessments of production scalability, cost-per-dose trajectories, and automation roadmaps. Companies without credible plans to reduce manufacturing costs by 60-70% over the next five years are facing valuation discounts or outright funding rejections.
Payers are equally focused on economics. Insurance providers and national health systems are signaling that reimbursement models will not support current pricing indefinitely. The UK's National Institute for Health and Care Excellence (NICE) has already rejected several cell therapies on cost-effectiveness grounds. Automated processing is not just an operational improvement but a market access requirement.
Technology Convergence Is Enabling True Closed-Loop Manufacturing
Recent advances in bioreactor design, AI-driven process control, and single-use technologies are finally making fully automated cell therapy production technically feasible. Systems can now handle the entire workflow from apheresis to formulation without breaking sterile barriers. Machine learning algorithms optimize culture conditions in real-time, adapting to patient-specific cell characteristics that previously required expert intervention.
The integration of these technologies is creating a new competitive dynamic. Early adopters are achieving 10-15 day reductions in vein-to-vein time, 80%+ decreases in contamination risk, and manufacturing costs approaching $100,000 per dose. These improvements are not incremental; they represent a fundamental shift in what is economically and operationally possible.
Where the Real Opportunity Lies
The highest-value opportunities are concentrated in three areas. Allogeneic therapy platforms represent the most compelling use case for automation because they require industrial-scale production from healthy donor cells. Companies developing off-the-shelf products need systems capable of producing thousands of doses per batch with pharmaceutical-grade consistency. The economics only work with full automation.
Autologous therapies for solid tumors present a different but equally urgent opportunity. Unlike hematological cancers where CAR-T has proven successful, solid tumor applications require more complex cell engineering and shorter processing windows. Automated systems that can handle multi-step genetic modifications while maintaining cell viability are becoming essential as next-generation therapies enter clinical development.
Contract development and manufacturing organizations (CDMOs) face existential pressure to automate. As therapy developers increasingly outsource production, CDMOs must demonstrate they can handle multiple product types, maintain segregated workflows, and scale rapidly. Manual operations cannot meet these requirements. CDMOs investing in modular, automated platforms are capturing disproportionate market share while those relying on traditional infrastructure are losing clients to more capable competitors.
Browse the Complete Report: https://marketmindsadvisory.com/automated-and-closed-cell-therapy-processing-systems-market/
The Competitive Landscape Is Fragmenting
The market is splitting into two distinct tiers. Integrated platform providers offering end-to-end automation are commanding premium positioning and long-term partnerships with major therapy developers. These systems, while requiring significant capital investment, provide the scalability and regulatory confidence that commercial-stage companies demand.
Point solution providers focusing on specific process steps face commoditization pressure. As integrated platforms mature, the value of standalone automation for individual unit operations is declining. Companies in this segment must either move up the value chain by developing broader capabilities or accept margin compression as their products become interchangeable components in larger systems.
The risk extends beyond technology providers. Therapy developers that have built internal manufacturing around manual or semi-automated processes face a strategic dilemma. Retrofitting existing facilities is often more expensive than building new automated capacity, but abandoning sunk investments is politically and financially difficult. This hesitation is creating a two-speed market where automated manufacturers are pulling ahead in cost structure and production capacity while traditional players struggle to compete.
The Cost of Delayed Action
Companies that postpone automation investments face compounding disadvantages:
* Regulatory vulnerability: As standards tighten, manual processes will require increasingly expensive validation and monitoring to maintain compliance, eventually becoming unapprovable for new indications.
* Talent scarcity: The specialized workforce required for manual cell therapy manufacturing is not scaling with demand. Labor costs are rising 15-20% annually in key markets, and turnover rates exceed 30% in some facilities.
* Competitive displacement: Early movers in automation are establishing cost structures that will be impossible for manual manufacturers to match, effectively pricing them out of competitive bids and partnership opportunities.
* Stranded assets: Facilities designed around manual processing are becoming obsolete faster than their depreciation schedules, creating balance sheet write-downs and limiting strategic flexibility.
* The window for orderly transition is narrowing. Companies that wait for automation technology to fully mature will find themselves too far behind to catch up.
What This Means for Decision-Makers
For Cell Therapy Developers and Manufacturers
Your manufacturing strategy is now a commercial strategy. Product candidates with compelling clinical data will fail if production economics do not support sustainable pricing. Evaluate your current cost structure against automated benchmarks and develop realistic transition roadmaps. Consider whether internal manufacturing still makes strategic sense or if partnerships with automated CDMOs offer faster paths to market. Most importantly, ensure your regulatory filings are not locking you into manual processes that will become liabilities.
For Equipment Manufacturers and Technology Providers
The market is moving beyond selling individual instruments to providing integrated manufacturing solutions. Customers increasingly expect turnkey systems with process development support, regulatory documentation, and ongoing optimization services. Companies that cannot offer comprehensive platforms should identify strategic partners or acquisition targets that fill capability gaps. The alternative is relegation to commodity supplier status with corresponding margin pressure.
For Investors and Capital Allocators
Manufacturing scalability is the primary gating factor for cell therapy commercialization. Due diligence must include detailed assessment of production strategies, automation roadmaps, and cost-per-dose trajectories. Companies with credible paths to sub-$150,000 manufacturing costs deserve valuation premiums. Those relying on manual processing without clear automation plans represent significant risk regardless of clinical promise. Consider direct investments in automation infrastructure as the picks-and-shovels play in the cell therapy gold rush.
For Healthcare Systems and Payers
Current cell therapy pricing is unsustainable, but automation offers a path to affordability. Engage proactively with manufacturers on volume-based agreements that share the economic benefits of automated production. Support regulatory frameworks that incentivize manufacturing innovation rather than entrenching legacy approaches. The difference between cell therapies remaining niche treatments and becoming standard care depends largely on manufacturing economics that automation can deliver.
Request Report Customization: https://marketmindsadvisory.com/request-customization/?report_id=8388
The manufacturing model that brought cell therapy from lab to clinic cannot take it to scale
The industry is at an inflection point where manufacturing capability will determine which therapies reach patients and which remain promising science. Automated and closed processing systems are not future technology; they are the present requirement for any company serious about commercial success. The strategic question is not whether to automate but how quickly you can complete the transition before market dynamics make your current approach uncompetitive. The companies moving decisively now are not just improving operations; they are securing their position in a market that will not accommodate inefficiency much longer.
Contact Us
Market Minds Advisory
86 Great Portland Street, Mayfair, London,
W1W 7FG, England, United Kingdom
T: +44 020 3807 7725
Email:sales@marketmindsadvisory.com
Website:https://marketmindsadvisory.com/
LinkedIn: https://www.linkedin.com/company/market-minds-advisory/
Facebook: https://www.facebook.com/resvaultmmadvisory/
Twitter: https://x.com/MarketMindsA
Instagram: https://www.instagram.com/marketmindsadvisory
Why choose Market Minds Advisory
Market Minds Advisory delivers decision-grade intelligence trusted by executives across machinery & equipment, packaging, chemical, automotive, information & communication technology, food & beverage, consumer goods, healthcare and other industries. We provide market expansion strategies, go-to-market strategies, market share acceleration, brand positioning analysis, and account enablement and growth. Our forecasting methodology integrates primary interviews, proprietary demand models and continuous market validation to ensure accuracy in volatile and emerging industries. With over 10 years of industry experience and insights derived from primary interviews with several industry stakeholders, our research provides actionable insights and white space analysis for the emerging segments providing the opportunity gaps in the market accounting recent market developments and geopolitical risks. We believe in unlocking growth by helping businesses to see the future of their markets.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Cell Therapy Manufacturing Is Hitting a Scalability Wall And Most Companies Are Unprepared here
News-ID: 4494183 • Views: …
More Releases from Market Minds Advisory

Automotive Bioplastics Face a Profitability Paradox as Regulatory Pressure Mount …
The automotive industry's sustainability pivot is creating winners and losers faster than most manufacturers anticipated, and bioplastics sit at the center of this disruption.
The Compliance Trap That's Reshaping Material Economics
Automakers are discovering that meeting 2025-2030 emissions and recyclability mandates isn't just about electrification. Material composition now directly impacts regulatory compliance scores, fleet-level carbon accounting, and increasingly, consumer purchasing decisions in key markets. The challenge: bioplastics currently cost 20-40% more than…
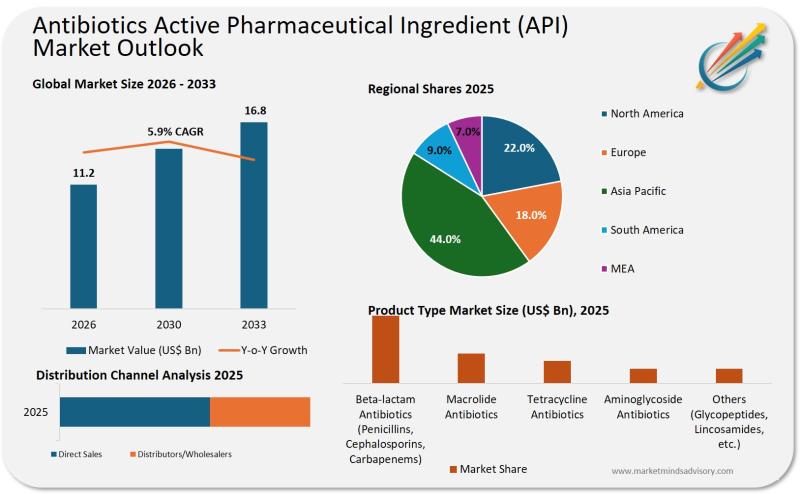
Antibiotics API Supply Chains Are Fracturing And Most Pharma Companies Aren't Re …
The global antibiotics API market is entering a period of structural instability, driven by geopolitical fragmentation, regulatory divergence, and the collapse of economic incentives for new antibiotic development. Companies that fail to reconfigure their sourcing strategies now risk supply disruptions, margin erosion, and regulatory non-compliance within the next 18 to 24 months.
Why This Market Shift Matters Now
For decades, the antibiotics API market operated on a simple logic: consolidate production in…
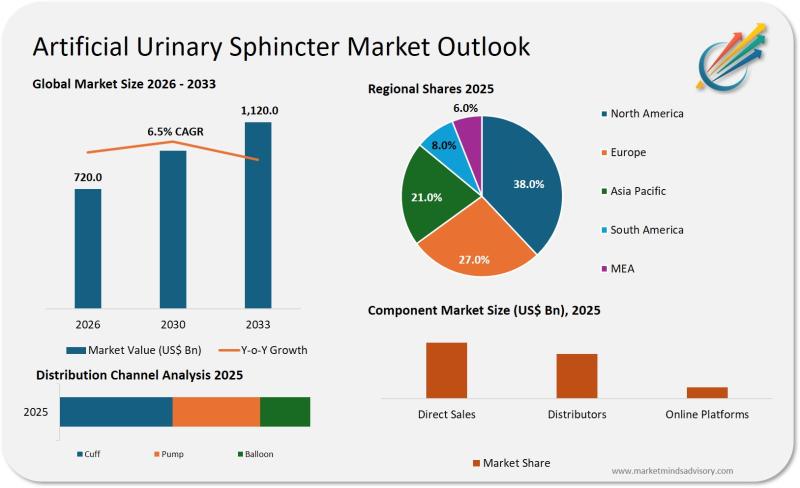
Artificial Urinary Sphincter Market Faces Critical Inflection as Patient Demand …
The gap between clinical need and device evolution is widening, creating strategic risk for medtech players and healthcare systems unprepared for demographic and regulatory shifts.
The Quiet Crisis in Urological Care
Healthcare systems across developed markets are confronting an uncomfortable reality: the population most in need of urinary incontinence solutions is growing faster than the innovation pipeline can support. Artificial urinary sphincters, once considered a mature category, are now at the center…
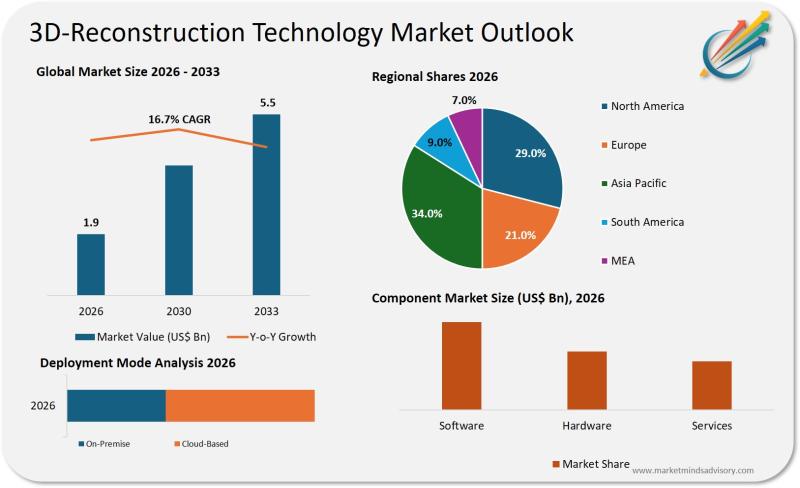
3D Reconstruction Technology Is No Longer Optional It's Infrastructure
Companies treating 3D reconstruction as a visualization tool rather than a strategic capability are already behind. The technology has crossed the threshold from experimental to mission-critical across industries where spatial intelligence determines competitive advantage.
The shift is structural. Organizations that once relied on manual measurement, 2D imaging, or legacy CAD workflows now face competitors deploying real-time 3D reconstruction to compress decision cycles, eliminate rework, and unlock entirely new revenue models. This…
More Releases for Automated
Automated Fiber Placements And Automated Tape Laying (AFP- ATL) Machines Market …
According to Market Research Intellect, the global Automated Fiber Placements And Automated Tape Laying (AFP- ATL) Machines market under the Internet, Communication and Technology category is expected to register notable growth from 2025 to 2032. Key drivers such as advancing technologies, changing consumer behavior, and evolving market dynamics are poised to shape the trajectory of this market throughout the forecast period.
The market for automated tape laying (ATL) and automated fiber…
Ace Automated Gates | Austin Enhances Security with Premium Automated Gate Solut …
Ace Automated Gates | Austin enhances property security with premium automated gate solutions. Specializing in gate installation, repair, and advanced access control systems, they provide high-quality electric and motorized gates for residential and commercial properties. Offering expert craftsmanship and durable materials, they ensure long-lasting security and convenience.
Austin, TX - Home and business owners seeking enhanced security, convenience, and style can now rely on Ace Automated Gates | Austin [https://aceautomatedgatesaustin.com/] for…
Automated Autoinjectors Market
The "Automated Autoinjectors Market" is expected to reach USD xx.x billion by 2031, indicating a compound annual growth rate (CAGR) of xx.x percent from 2024 to 2031. The market was valued at USD xx.x billion In 2023.
Growing Demand and Growth Potential in the Global Automated Autoinjectors Market, 2024-2031
Verified Market Research's most recent report, "Automated Autoinjectors Market: Global Industry Trends, Share, Size, Growth, Opportunity and Forecast 2023-2030," provides an in-depth examination…
Automated Journalism Market All Sets for Continued Outperformance: Automated Ins …
HTF MI introduces new research on Automated Journalism covering the micro level of analysis by competitors and key business segments (2024-2030). The Automated Journalism explores a comprehensive study of various segments like opportunities, size, development, innovation, sales, and overall growth of major players. The research is carried out on primary and secondary statistics sources and it consists of both qualitative and quantitative detailing. Some of the major key players profiled…
Automated Fiber Placements and Automated Tape Laying (AFP- ATL) Machines Market …
Automated fiber placement and automated tape laying machines are used for the fabrication of composites parts used in aerospace and defense industries. These machines help in precisely placing fiber tapes, tows and sheets on molds or cores to manufacture composite parts with high strength and lightweight properties.
Market Dynamics:
Increasing demand from aerospace and defense industry is one of the major factors estimated to boost the growth of the AFP- ATL machines…
Automated Microbiology Market - Efficiency Meets Accuracy: Redefining Microbiolo …
Newark, New Castle, USA: The "Automated Microbiology Market" provides a value chain analysis of revenue for the anticipated period from 2022 to 2030. The report will include a full and comprehensive analysis of the business operations of all market leaders in this industry, as well as their in-depth market research, historical market development, and information about their market competitors
Automated Microbiology Market: https://www.growthplusreports.com/report/automated-microbiology-market/7801
This latest report researches the industry structure, sales, revenue,…