Press release
Key Factors and Emerging Trends Shaping the Pediatric Clinical Trials Market Landscape
The pediatric clinical trials sector is on track for significant expansion as the demand for child-specific medical research intensifies. Innovations such as digital and artificial intelligence tools are reshaping trial methodologies, while growing collaborations and regulatory support further accelerate the market's development. This overview explores the expected market size, key driving factors, major participants, and segmentation trends shaping the industry through 2030.Pediatric Clinical Trials Market Size and Growth Outlook
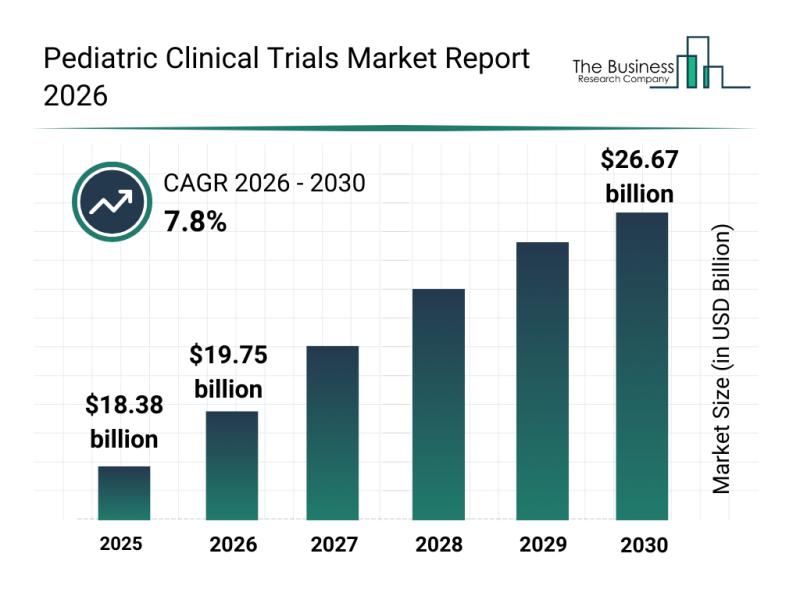
Forecasts indicate that the pediatric clinical trials market will expand to $26.67 billion by 2030, growing at a compound annual growth rate (CAGR) of 7.8%. This robust growth is driven by increasing requirements for therapies tailored specifically to children, wider adoption of AI and digital technologies in clinical processes, enhanced regulatory incentives encouraging pediatric research, and stronger partnerships between pharmaceutical companies and research bodies. Additionally, there is a growing emphasis on patient-centered approaches and individualized clinical trial designs for pediatric populations. Key trends expected to influence the market include the uptake of precision medicine, integration of real-world evidence, and rising investments dedicated to pediatric drug development.
Download a free report of the pediatric clinical trials market report:
https://www.thebusinessresearchcompany.com/report/pediatric-clinical-trials-global-market-report?utm_source=OpenPR&utm_medium=Paid&utm_campaign=Apr_PR
Primary Factors Fueling Pediatric Clinical Trials Market Expansion
The rising demand for therapies that address unique pediatric health needs is a fundamental driver for this market. As pediatric diseases require specialized treatment protocols, clinical trials focused on this group are becoming increasingly essential.
Simultaneously, the integration of digital tools and AI technologies in clinical trial design and data analysis is enhancing efficiency and accuracy. These advancements help optimize study protocols and accelerate drug development timelines, making pediatric clinical trials more feasible and effective.
Key Players Leading the Pediatric Clinical Trials Industry
The pediatric clinical trials field includes numerous influential companies such as Medpac Inc., Pharmaceutical Product Development Inc., ICON plc, Syneos Health Inc., QPS Holdings LLC, Pfizer Inc., IQVIA Inc., Premier Research, Labcorp, The Emmes Company LLC, Synteract Inc., Charles River Laboratories International Inc., Covance Inc., Bristol-Myers Squibb Company, GlaxoSmithKline plc, BioNTech SE, Moderna Inc., Parexel International Corporation, PRA Health Sciences, Wuxi AppTec, Merck & Co. Inc., AstraZeneca plc, Sanofi S.A., Johnson & Johnson, Eli Lilly and Company, Takeda Pharmaceutical Company Limited, C.H. Boehringer Sohn AG & Co. KG, AbbVie Inc., Teva Pharmaceutical Industries Ltd., and Oracle Health Sciences.
View the full pediatric clinical trials market report:
https://www.thebusinessresearchcompany.com/report/pediatric-clinical-trials-global-market-report?utm_source=OpenPR&utm_medium=Paid&utm_campaign=Apr_PR
Recent Strategic Moves in the Pediatric Clinical Trials Sector
In March 2024, PCM Trials, known for providing mobile research nurse visits for decentralized clinical trials, acquired EmVenio Research in an effort to enhance patient-centric and decentralized clinical trial models. This acquisition aims to improve recruitment and retention, especially among underrepresented minority groups. EmVenio Research specializes in expanding access to pediatric clinical trials for diverse and underserved populations within the United States.
Collaborations Shaping Pediatric Clinical Trials Development
Industry players are increasingly engaging in partnerships to enhance service delivery and research outcomes. An example is the September 2023 collaboration between Strados Labs, a US-based smart biosensor provider for respiratory monitoring, and Ann & Robert H. Lurie Children's Hospital, a pediatric acute care hospital. Their joint clinical trial focuses on the RESP Biosensor, a wearable device that detects lung sounds associated with asthma symptoms such as coughing and wheezing. The trial aims to validate the device's effectiveness in monitoring asthma exacerbations in children.
Breakdown of Pediatric Clinical Trials Market Segments
This market is analyzed through several key segments:
1) By Phase: Phase I, Phase II, Phase III, and Phase IV trials
2) By Study Design: Treatment studies and observational studies
3) By Therapeutic Areas: Including infectious diseases, oncology, autoimmune or inflammatory diseases, respiratory disorders, mental health disorders, and other therapeutic categories
Detailed subsegments within each phase include:
- Phase I: First-in-pediatrics trials, dose escalation studies, safety and tolerability assessments
- Phase II: Efficacy studies, dose optimization trials, pharmacokinetic studies
- Phase III: Large-scale efficacy trials, comparative studies, long-term safety evaluations
- Phase IV: Post-marketing surveillance, long-term safety monitoring, and real-world effectiveness studies
These classifications provide a comprehensive view of the pediatric clinical trials market landscape and its areas of focus for drug development and regulatory validation.
Reach out to us:
The Business Research Company: https://www.thebusinessresearchcompany.com/,
Americas +1 310-496-7795,
Europe +44 7882 955267,
Asia & Others +44 7882 955267 & +91 8897263534,
Email us at info@tbrc.info.
Follow Us On:
LinkedIn: https://in.linkedin.com/company/the-business-research-company,
Twitter: https://twitter.com/tbrc_info,
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Learn More About The Business Research Company
With over 17500+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead.Our flagship product, the Global Market Model (GMM), is a premier market intelligence platform delivering comprehensive and updated forecasts to support informed decision-making.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Key Factors and Emerging Trends Shaping the Pediatric Clinical Trials Market Landscape here
News-ID: 4487440 • Views: …
More Releases from The Business Research Company
Emerging Sub-Segments Transforming the Pelvic Cancer Induced Hemorrhagic Cystiti …
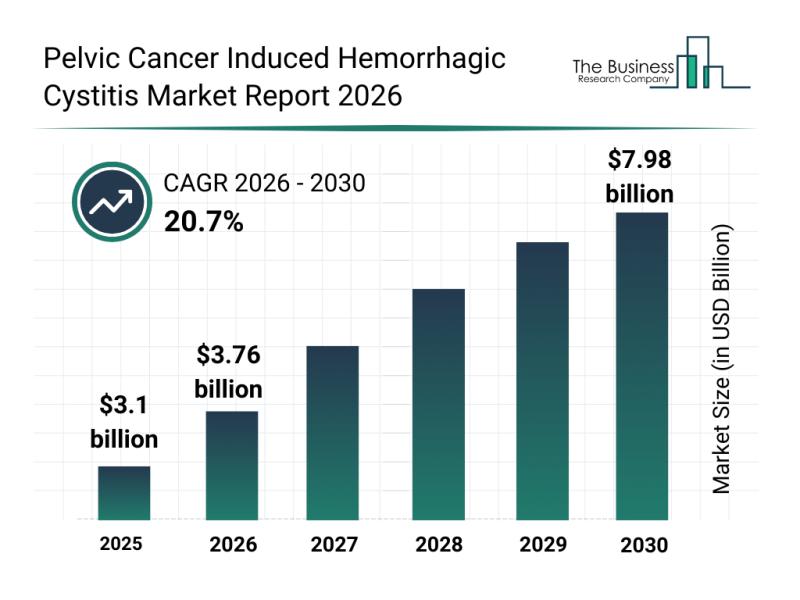
The pelvic cancer induced hemorrhagic cystitis market is poised for remarkable expansion in the coming years. With ongoing advancements in medical research and technology, this field is set to experience substantial growth driven by innovations and improved treatment methods. Let's explore the market size, key players, emerging trends, and important segments shaping the future of this specialized healthcare sector.
Projected Market Expansion for Pelvic Cancer Induced Hemorrhagic Cystitis by 2030
The…
Competitive Landscape: Leading Companies and New Entrants in the Pediatric Imagi …
The pediatric imaging sector is poised for significant advancement as healthcare providers increasingly prioritize precise and safe diagnostic tools for children. With technological innovations and growing investments, this market is set to expand rapidly, offering enhanced solutions tailored specifically for pediatric care.
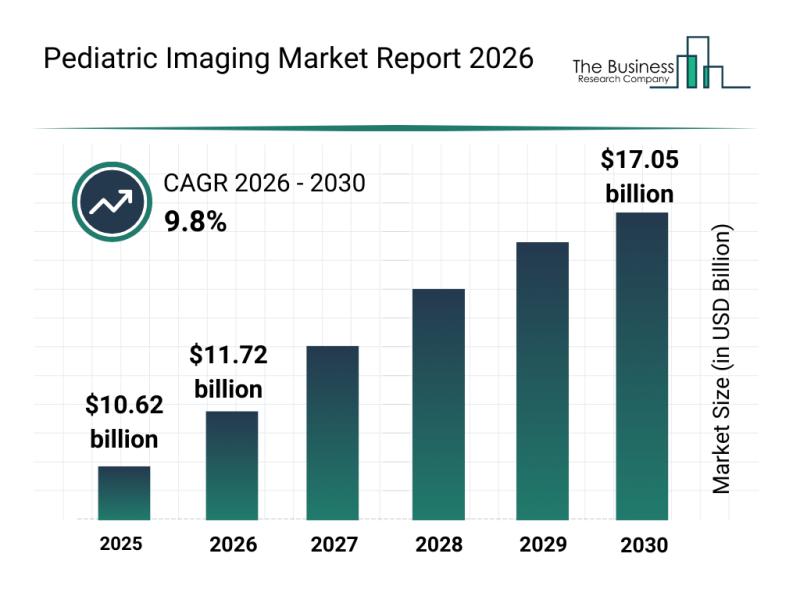
Projected Pediatric Imaging Market Value by 2030
The pediatric imaging market is anticipated to experience robust growth, reaching a value of $17.05 billion by 2030. This expansion corresponds to…
Emerging Sub-Segments Transforming the Urgent Care Apps Market Landscape
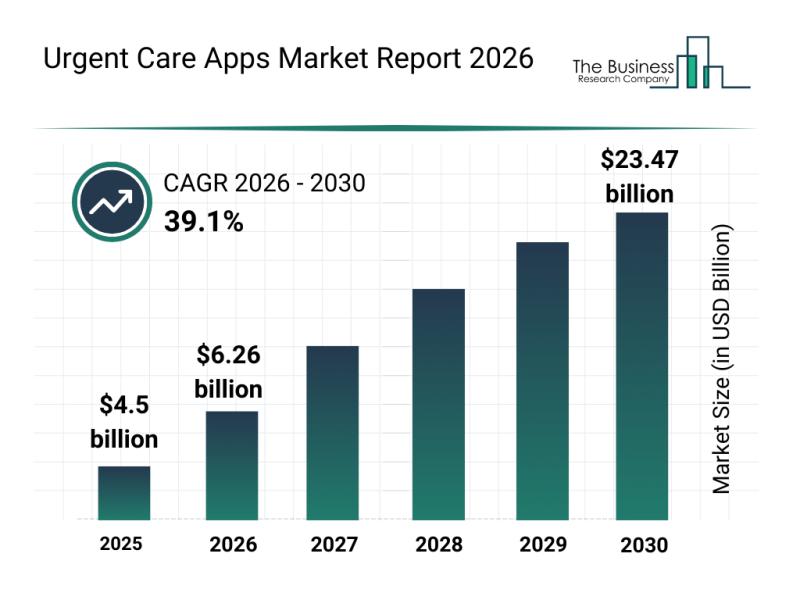
The urgent care apps industry is on the verge of remarkable expansion, driven by rapid advancements in digital health technology and growing demand for accessible medical services. As healthcare increasingly shifts towards remote and AI-assisted platforms, urgent care apps are becoming vital tools for efficient patient management and timely treatment. Let's explore the current market size, primary drivers, leading companies, emerging trends, and key segments shaping this fast-evolving sector.
Projected Market…
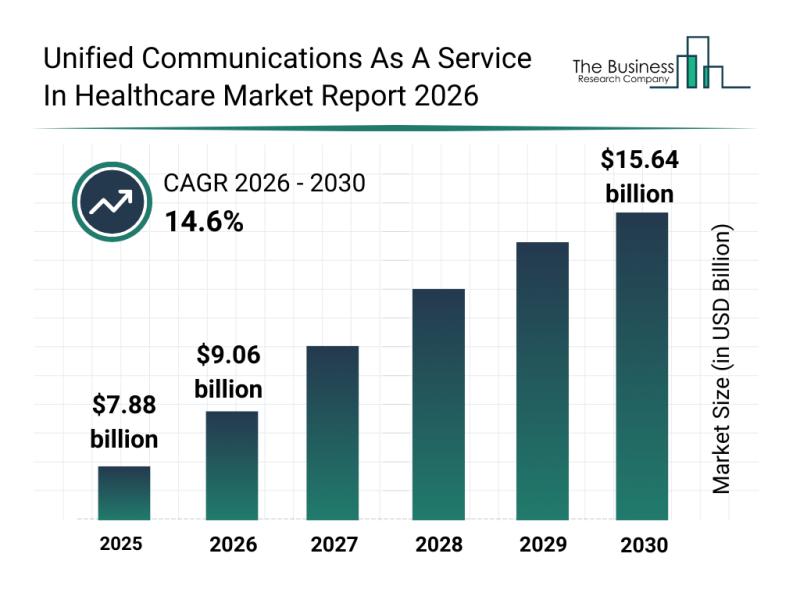
Competitive Landscape: Key Market Leaders and New Entrants in the Unified Commun …
The unified communications as a service (UCaaS) sector within healthcare is set to experience remarkable growth in the upcoming years, driven by evolving technological needs and enhanced care delivery models. As healthcare providers increasingly rely on seamless, secure communication tools, this market is positioned for substantial expansion by 2030, supported by several key factors shaping its trajectory.
Forecasted Market Growth and Size of Unified Communications As A Service in Healthcare …
More Releases for Trials
Clinical Trials Management System Market Trends: How Decentralized Clinical Tria …
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Clinical Trials Management System Market Size, Share & Trends Analysis Report By Solution Type (Enterprise and Site based), By Delivery Mode (Web & Cloud-based, On-premise), By Component (Software, Services), By End-user (Pharmaceutical and Biotechnology Firms, Medical Device Firms, CROs)- Market Outlook And Industry Analysis 2031"
The global Clinical Trials Management System market is estimated to reach…
Brazil Clinical Trials Market ANVISA Brazil Guidelines Brazil Clinical Trials Re …
Brazil Cancer Drugs Clinical Trials Insight 2024 Report Offering:
• Brazil Clinical Trials Market Opportunity 2024 and 2030 (In US$ Billion)
• Clinical Trials Regulatory Framework In Brazil
• Total Number of Cancer Drugs In Clinical Trials In Brazil
• Total Number Of Cancer Drugs Approved In Brazil
• 400 Pages Clinical Trials Insight On All Cancer Drugs In Clinical Trials By Company, Indication and Phase
• 80 Pages Clinical Insight On All Cancer Drugs Approved in Market By Company and Indication
• Insight…
Clinical Trials Management System Market Clinical Trials Management System Marke …
InsightAce Analytic announces the release of a market assessment report on the "Global Clinical Trials Management System Market Size, Share & Trends Analysis Report By Solution Type (Enterprise and Site based), By Delivery Mode (Web & Cloud-based, On-premise), By Component (Software, Services), By End-user (Pharmaceutical and Biotechnology Firms, Medical Device Firms, CROs)- Market Outlook And Industry Analysis 2031.
The global Clinical Trials Management System market is estimated to reach over USD…
Revolutionizing Clinical Trials: The Evolution of eCOA, eSource, and the Clinica …
The global eCOA, eSource & clinical trials market is valued at US$ 48 billion in 2023 and is expected to reach a market size of US$ 104 billion by the end of 2033, expanding rapidly at a CAGR of 8% over the next ten years. Worldwide demand for eCOA (electronic clinical outcome assessment) solutions is predicted to rise at a CAGR of 8.2% over the forecast period.
In the dynamic landscape…
Virtual Clinical Trials Market - Bridging the Gap: Virtual Clinical Trials Revol …
Newark, New Castle, USA - The latest report from Growth Plus Reports analyzes the production, potential applications, demand, major manufacturers, and SWOT analysis of the global Virtual Clinical Trials Market.
Virtual Clinical Trials Market: https://www.growthplusreports.com/report/virtual-clinical-trials-market/9106
The Virtual Clinical Trials Market Report assists in determining the optimum distribution methods for certain products as well as possible markets for future product launches. The report also analyses the purchase and supply trends that influence the…
Clinical Trials Market: Coronavirus Pandemic Pushes Sponsors, Patients to Adopt …
Clinical trials Market: Introduction
According to the report, the global clinical trials market was valued over US$ 46.7 Bn in 2019 and is projected to expand at a CAGR of ~5% from 2020 to 2030. High prevalence and increase in incidence rate of chronic diseases, and rise in R&D activities in biotechnology & pharmaceuticals industries are anticipated to drive the global clinical trials market from 2020 to 2030. North America is projected to…