Press release
Global Coagulation Factor VII Treatment Market - Industry Trends, Growth Analysis, and Forecast 2026-2033 (CAGR of 9.80%)
As per Data Bridge Market Research analysis, the global coagulation factor VII treatment market was estimated at USD 1.80 billion in 2025. The market is expected to grow from USD 1.98 billion in 2026 to USD 2.90 billion in 2030, at a CAGR of 9.80% during the forecast period (2026-2030), driven by the rising demand for advanced hemophilia therapies, increasing prevalence of rare bleeding disorders, and growing adoption of recombinant coagulation factor products.Key growth drivers include technological advancements in biologics, rising healthcare expenditure, strong regulatory support for orphan drugs, and increased awareness and diagnosis of rare coagulation disorders globally.
Get the full PDF sample copy of the report: https://www.databridgemarketresearch.com/request-a-sample/?dbmr=global-coagulation-factor-vii-treatment-market
Market Size & Forecast
2025 Market Size: USD 1.80 Billion
2026 Projected Market Size: USD 1.98 Billion
2030 Projected Market Size: USD 2.90 Billion
CAGR (2026-2030): 9.80%
Largest Region: North America
Fastest Growing Region: Asia Pacific
Key Market Report Takeaways
North America accounts for approximately 42% of global market share due to advanced healthcare infrastructure and high adoption of recombinant therapies
Asia Pacific is projected to grow at the fastest CAGR exceeding 11% due to increasing patient pool and healthcare investments
Recombinant factor VII products dominate the market with the highest share due to superior safety and efficacy profiles
Congenital factor VII deficiency treatment represents the leading application segment
Hospitals remain the dominant end-use segment driven by specialized treatment requirements and access to advanced biologics
Key Market Trends & Highlights
North America leads the global market due to strong reimbursement frameworks, early adoption of biologics, and presence of leading pharmaceutical companies
Asia Pacific is the fastest-growing region driven by rising healthcare expenditure, improving diagnostic capabilities, and expanding access to rare disease treatments
Recombinant therapies remain the dominant segment due to reduced risk of pathogen transmission and increased clinical preference
Market growth is supported by increasing prevalence of bleeding disorders, favorable orphan drug policies, and advancements in biotechnology
Emerging technologies such as gene therapy and extended half-life biologics are reshaping treatment paradigms
Strategic collaborations, regulatory approvals, and investments in rare disease research are accelerating market expansion
Market Dynamics
Market Drivers
Rising Prevalence of Rare Bleeding Disorders
The increasing incidence of congenital factor VII deficiency and other coagulation disorders is a primary growth driver. Improved diagnostic capabilities in regions such as North America and Europe are leading to higher detection rates. Emerging economies are also witnessing better disease identification due to awareness programs. This expanding patient pool is directly contributing to sustained demand for treatment solutions globally.
Advancements in Recombinant Biologics
Technological innovations in recombinant DNA technology have significantly enhanced the safety and efficacy of factor VII treatments. Recombinant products eliminate the risk of blood-borne infections associated with plasma-derived therapies. Continuous R&D investments by pharmaceutical companies are improving product stability and half-life. These advancements are accelerating adoption across developed healthcare systems.
Favorable Regulatory Environment and Orphan Drug Incentives
Governments and regulatory bodies are offering incentives such as fast-track approvals, tax benefits, and exclusivity for orphan drugs. The U.S. and Europe have established strong frameworks supporting rare disease treatments. These policies encourage pharmaceutical companies to invest in niche therapeutic areas. As a result, the pipeline for coagulation factor therapies is expanding steadily.
Increasing Healthcare Expenditure and Access to Specialized Care
Rising healthcare spending, particularly in developed economies, is supporting the adoption of advanced therapies. Hospitals and specialty clinics are increasingly equipped to administer biologics. In Asia Pacific, government initiatives are improving access to rare disease treatments. This trend is driving market penetration in both developed and emerging regions.
Growing Awareness and Screening Programs
Awareness campaigns and screening initiatives by healthcare organizations are improving early diagnosis rates. Patient advocacy groups are playing a significant role in education and treatment accessibility. Early diagnosis enables timely intervention, increasing treatment demand. This factor is particularly impactful in emerging markets with historically low diagnosis rates.
Market Restraints
High Cost of Treatment
Coagulation factor VII therapies, particularly recombinant products, are associated with high costs. This creates affordability challenges, especially in low- and middle-income countries. Limited insurance coverage in some regions further restricts access. The high cost burden impacts overall market penetration and patient adherence.
Regulatory Complexities and Approval Delays
Stringent regulatory requirements for biologics can delay product approvals. Compliance with safety, efficacy, and quality standards requires extensive clinical trials. These processes increase development timelines and costs. Smaller companies often face barriers in navigating complex regulatory frameworks.
Limited Patient Population
Factor VII deficiency is a rare condition, resulting in a relatively small target population. This limits large-scale commercialization opportunities for manufacturers. Market growth is dependent on accurate diagnosis and patient identification. The niche nature of the disease restricts volume-driven revenue expansion.
Supply Chain and Manufacturing Challenges
Biologic production requires highly specialized infrastructure and strict quality control. Disruptions in supply chains, particularly for raw materials, can impact production. Maintaining cold chain logistics adds complexity to distribution. These challenges affect product availability in remote and developing regions.
Competition from Alternative Therapies
Emerging therapies such as gene therapy and bypassing agents are creating competitive pressure. These alternatives may offer long-term or curative potential. As innovation progresses, traditional factor replacement therapies may face reduced demand. This could impact long-term market growth dynamics.
Market Opportunities
Development of Gene Therapy Solutions
Gene therapy represents a transformative opportunity in the treatment of coagulation disorders. Ongoing research aims to provide long-term or permanent solutions. Successful commercialization could significantly improve patient outcomes. Companies investing in gene therapy pipelines are positioned for future growth.
Expansion in Emerging Markets
Asia Pacific, Latin America, and the Middle East & Africa offer untapped growth potential. Improving healthcare infrastructure and government initiatives are increasing treatment accessibility. Rising awareness and diagnostic capabilities are expanding the patient base. These regions present significant revenue opportunities for market players.
Strategic Collaborations and Partnerships
Pharmaceutical companies are increasingly engaging in partnerships to enhance R&D capabilities. Collaborations with biotech firms and research institutions accelerate innovation. Joint ventures also facilitate market entry in new regions. These strategies strengthen competitive positioning and expand product portfolios.
Advancements in Extended Half-Life Products
Development of extended half-life coagulation factors is improving treatment convenience. These products reduce dosing frequency and enhance patient compliance. Technological advancements in protein engineering are driving this segment. Increased adoption is expected to boost overall market growth.
Government Support and Rare Disease Funding
Governments are increasing funding for rare disease research and treatment programs. Initiatives supporting early diagnosis and treatment accessibility are expanding. Public-private partnerships are fostering innovation in niche therapeutic areas. These factors create a favorable environment for market expansion.
Get the full of the report: https://www.databridgemarketresearch.com/reports/global-coagulation-factor-vii-treatment-market/companies
Market Challenges
High Dependency on Specialized Healthcare Infrastructure
Administration of coagulation factor therapies requires specialized clinical settings. Limited availability of such infrastructure in developing regions restricts access. This creates disparities in treatment availability globally. Addressing infrastructure gaps remains a critical challenge for market expansion.
Complex Manufacturing Processes
Biologic production involves intricate processes requiring stringent quality control. Any deviation can impact product efficacy and safety. Scaling up production while maintaining consistency is challenging. These factors increase operational costs and limit production capacity.
Regulatory and Compliance Burden
Continuous updates in regulatory standards require companies to adapt frequently. Compliance with international guidelines adds complexity for global operations. Delays in approvals can hinder product launches. This impacts revenue generation and competitive positioning.
Market Fragmentation and Competitive Pressure
The presence of multiple global and regional players creates competitive intensity. Price competition and product differentiation challenges affect profitability. Smaller players may struggle to sustain operations. This fragmentation influences overall market dynamics.
Economic Constraints in Developing Regions
Limited healthcare budgets and reimbursement challenges affect treatment adoption. Patients in low-income regions often lack access to advanced therapies. Economic instability can impact healthcare investments. These factors restrict market growth in emerging economies.
Market Segmentation & Analysis
By Product Type
The market is segmented into plasma-derived factor VII and recombinant factor VII. Recombinant factor VII dominates the market with over 65% share due to its safety profile and reduced infection risk. This segment is also the fastest-growing, driven by technological advancements and clinical preference. Plasma-derived products remain relevant in cost-sensitive regions. Growth is supported by increasing adoption in developed markets.
By Application
Applications include congenital factor VII deficiency and acquired coagulation disorders. Congenital deficiency accounts for the largest share due to long-term treatment requirements. The segment benefits from early diagnosis and continuous therapy demand. Acquired disorders are growing steadily due to increasing surgical procedures and trauma cases. The congenital segment is expected to maintain dominance throughout the forecast period.
By End-User
End-users include hospitals, specialty clinics, and research institutes. Hospitals dominate the segment due to access to advanced infrastructure and skilled professionals. Specialty clinics are growing rapidly with increasing outpatient treatment models. Research institutes contribute to innovation and clinical trials. Hospitals remain the primary revenue-generating segment globally.
By Distribution Channel
The market is segmented into hospital pharmacies, retail pharmacies, and online pharmacies. Hospital pharmacies hold the largest share due to controlled drug administration. Retail pharmacies are expanding in developed regions with improved access. Online pharmacies are emerging as a fast-growing segment due to digital healthcare trends. Distribution efficiency is improving market accessibility.
Analytical Insights
Largest Segment: Recombinant factor VII (due to safety and efficacy advantages)
Fastest Growing Segment: Extended half-life recombinant therapies
Dominance driven by technological innovation, regulatory support, and clinical outcomes
Increasing adoption trends and patient preference for advanced biologics support segment growth
Regional Analysis
By geography, the market is categorized into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
North America
North America dominates the market, accounting for approximately 42% of global revenue. The region benefits from advanced healthcare infrastructure, strong reimbursement systems, and early adoption of biologics. The U.S. is the key contributor due to high healthcare spending and presence of leading pharmaceutical companies. Regulatory support for orphan drugs further strengthens market growth.
Europe
Europe represents a mature and steadily growing market driven by strong R&D investments and regulatory frameworks. Countries such as Germany, the U.K., and France lead regional growth. Increasing focus on rare disease treatment and government support enhances market expansion. Adoption of advanced therapies remains high across the region.
Asia Pacific
Asia Pacific is the fastest-growing region, with a CAGR exceeding 11%. Rapid industrialization, improving healthcare infrastructure, and government initiatives are driving growth. China, India, and Japan are key contributors due to large patient populations. Increasing awareness and diagnostic capabilities are expanding market demand.
Latin America
Latin America is an emerging market with gradual growth driven by improving healthcare systems. Brazil and Mexico lead regional development. Economic constraints and limited access to advanced therapies remain challenges. However, increasing investments are supporting market expansion.
Middle East & Africa
The Middle East & Africa region shows steady but slower growth. Government initiatives and private sector investments are improving healthcare access. Infrastructure gaps and limited awareness restrict growth potential. However, gradual improvements are expected to support long-term market development.
Key Insights:
Largest Region: North America
Fastest Growing Region: Asia Pacific
Competitive Landscape
Market Structure Overview
The coagulation factor VII treatment market is moderately consolidated, with a mix of global pharmaceutical leaders and regional players. Competition is driven by technological innovation, product efficacy, and regulatory approvals. Companies focus on strengthening their biologics portfolios and expanding geographic reach. Competitive landscape analysis provides insights into strategic positioning and market dynamics.
Key Industry Players
Leading companies operate with strong R&D capabilities and global distribution networks. They focus on innovation, product differentiation, and regulatory approvals. Market leaders maintain dominance through advanced recombinant technologies.
List of Key Industry Players:
Novo Nordisk A/S
Pfizer Inc.
F. Hoffmann-La Roche Ltd.
Bayer AG
CSL Limited
Takeda Pharmaceutical Company Limited
Octapharma AG
Grifols, S.A.
Competitive Strategies
Companies are investing in product innovation, particularly recombinant and gene therapies. Strategic partnerships and collaborations are enhancing research capabilities. Mergers and acquisitions are strengthening market presence. Geographic expansion into emerging markets is a key focus area. Firms aim to improve patient outcomes and gain competitive advantage through advanced solutions.
Emerging Players & Market Dynamics
Startups and biotech firms are introducing innovative therapies and cost-effective solutions. Increasing funding and investment activities are supporting new entrants. Emerging players focus on niche innovations such as gene therapy. This is intensifying competition and reshaping market dynamics.
Latest Developments
January 2025 - Novo Nordisk A/S: Launched an advanced recombinant factor VII therapy with extended half-life, improving dosing efficiency and patient compliance.
October 2024 - Pfizer Inc.: Announced strategic collaboration with a biotech firm to develop gene therapy solutions for rare bleeding disorders, strengthening pipeline innovation.
June 2024 - CSL Limited: Expanded manufacturing capacity in Europe to meet rising demand for coagulation therapies, enhancing supply chain efficiency.
March 2024 - Takeda Pharmaceutical Company: Received regulatory approval for a new factor VII treatment in Japan, expanding its Asia Pacific presence.
December 2023 - Bayer AG: Invested in R&D for next-generation biologics targeting rare coagulation disorders, reinforcing its innovation strategy.
September 2023 - Octapharma AG: Entered a partnership with research institutions to accelerate development of plasma-derived therapies, supporting product diversification.
July 2023 - Grifols, S.A.: Expanded distribution network in Latin America to improve market access and regional penetration.
For More Regional Related Reports @
https://www.databridgemarketresearch.com/reports/global-cyclodextrins-in-pharma-market
https://www.databridgemarketresearch.com/reports/global-cytokine-release-syndrome-drug-market
https://www.databridgemarketresearch.com/reports/global-dementia-alzheimer-disease-market
https://www.databridgemarketresearch.com/reports/global-gaucher-disease-market
https://www.databridgemarketresearch.com/reports/global-minocycline-hydrochloride-market
https://www.databridgemarketresearch.com/reports/global-otic-drugs-market
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email:- corporatesales@databridgemarketresearch.com
About Data Bridge Market Research:
An absolute way to forecast what the future holds is to comprehend the trend today!
Data Bridge Market Research set forth itself as an unconventional and neoteric market research and consulting firm with an unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market. Data Bridge endeavors to provide appropriate solutions to the complex business challenges and initiates an effortless decision-making process. Data Bridge is an aftermath of sheer wisdom and experience which was formulated and framed in the year 2015 in Pune.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Coagulation Factor VII Treatment Market - Industry Trends, Growth Analysis, and Forecast 2026-2033 (CAGR of 9.80%) here
News-ID: 4487265 • Views: …
More Releases from Data Bridge Market Research
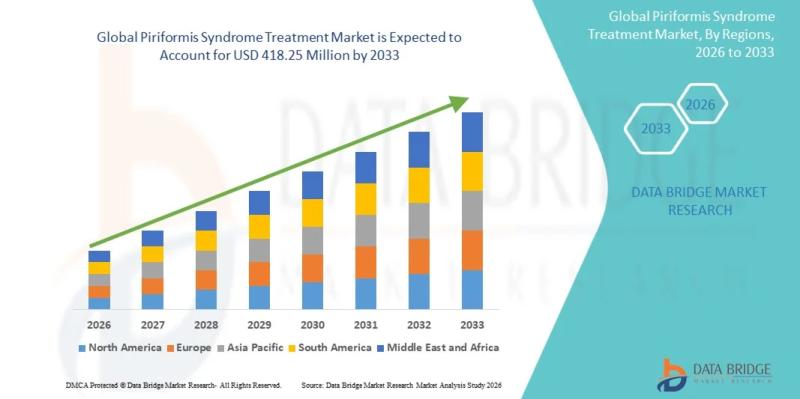
Piriformis Syndrome Treatment Market Size to Reach USD 418.25 million by 2033, G …
As per Data Bridge Market Research analysis, the Piriformis Syndrome Treatment Market was estimated at USD 0.26 billion in 2025. The market is expected to grow from USD 262.42 million in 2025 to USD 418.25 million by 2033, at a CAGR of 6.00% during the forecast period with driven by the rising demand for minimally invasive pain management therapies, increasing prevalence of musculoskeletal disorders, advancements in diagnostic imaging, and growing…
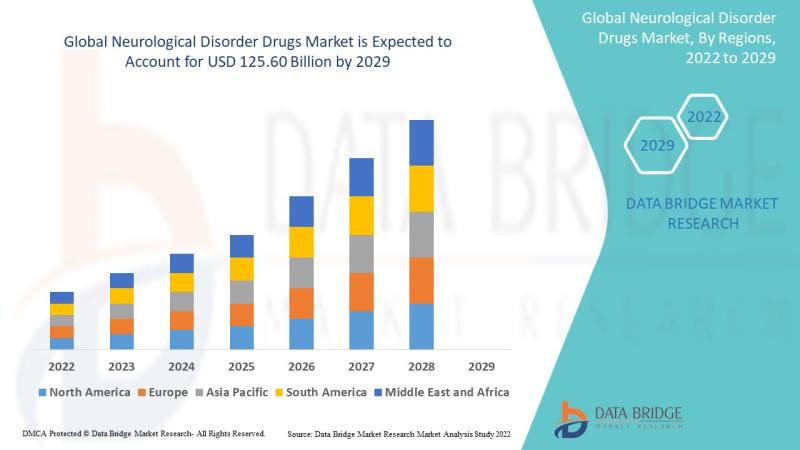
Neurological Disorder Drugs Market Outlook (2026-2032): 5.9% CAGR Driving Growth …
"As per Data Bridge Market Research analysis, the Neurological Disorder Drugs Market was estimated at USD 99.86 billion in 2025. The market is expected to grow from USD 105.76 billion in 2026 to USD 149.17 billion in 2032, at a CAGR of 5.9% during the forecast period with driven by the rising demand for advanced neurotherapeutics, increasing prevalence of neurological diseases, and expanding adoption of targeted biologics and precision medicine…
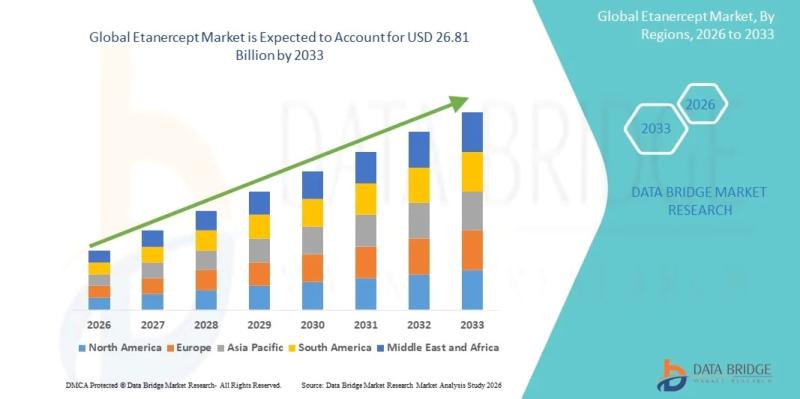
Etanercept Market Size, Share & Industry Trends Analysis Report, 2026-2033: Mark …
As per Data Bridge Market Research analysis, the Etanercept Market was estimated at USD 20.05 billion in 2025. The market is expected to grow from USD 20.79 billion in 2026 to USD 26.81 billion in 2033, at a CAGR of 3.7% during the forecast period with driven by the rising demand for biologic therapies for autoimmune disorders, expanding biosimilar adoption, increasing prevalence of rheumatoid arthritis and psoriasis, and continuous advancements…
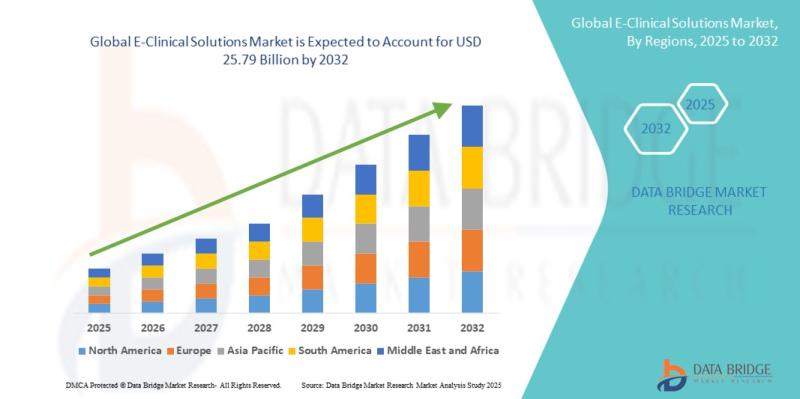
E-Clinical Solutions Market Size, Share & Industry Analysis, By Product, Deploym …
As per Data Bridge Market Research analysis, the E-Clinical Solutions Market was estimated at USD 10.62 billion in 2025. The market is expected to grow from USD 12.06 billion in 2026 to USD 25.79 billion in 2032, at a CAGR of 13.50% during the forecast period, driven by the rising demand for decentralized clinical trials, increasing adoption of cloud-based clinical data management platforms, growing pharmaceutical R&D investments, and expanding integration…
More Releases for VII
TeroAI Wins Lightning Round VII Pitch Competition Hosted by 1752vc
TeroAI was selected as the winner of 1752vc's Lightning Round VII after pitching alongside leading early-stage startups in front of a panel of investors and operators, with more than 1,000 people signing up to attend the event.
Santa Monica, CA - Feb 17, 2026 - 1752vc [http://1752.vc/?utm_source=abnews] (formerly Pegasus Angel Accelerator) successfully hosted Lightning Round VII, bringing together four exceptional early stage founders to pitch live in front of leading investors…
Human Coagulation Factor VII Market Analysis By Top Keyplayers - NovoNordisk, No …
The "Human Coagulation Factor VII Market" is expected to reach USD xx.x billion by 2031, indicating a compound annual growth rate (CAGR) of xx.x percent from 2024 to 2031. The market was valued at USD xx.x billion In 2023.
Growing Demand and Growth Potential in the Global Human Coagulation Factor VII Market, 2024-2031
Verified Market Research's most recent report, "Human Coagulation Factor VII Market: Global Industry Trends, Share, Size, Growth, Opportunity and…
Human Coagulation Factor VII Market 2022 | Detailed Report
Global Human Coagulation Factor VII Market 2022-2028, has been prepared based on an in-depth market analysis with inputs from industry experts. The report covers the market landscape and its growth prospects in the coming years. The report includes a discussion of the key vendors operating in this market. An exclusive data offered in this report is collected by research and industry experts team.
Download FREE Sample Report @ https://www.reportsnreports.com/contacts/requestsample.aspx?name=5558827
The report…
Global Human Coagulation Factor VII Market Report Forecast 2021 to 2027
Human coagulation factor VII is a single-chain plasma glycoprotein that starts the coagulation cascade by causing blood to clot. It's a member of the serine protease family of enzymes. Factor VII is made in the liver and subsequently circulates in the bloodstream, where it interacts with tissue factor, a membrane protein. Factor VII is transformed to factor VIIa after binding, which starts the coagulation process.
The global Human Coagulation Factor VII…
Human Coagulation Factor VII Market 2020 Demand Analysis – NovoNordisk
Global Human Coagulation Factor VII Market Growth 2020-2025
The report will make detailed analysis mainly on in-depth research on the development environment, Market size, development trend, operation situation and future development trend of Human Coagulation Factor VII Market on the basis of stating current situation of the industry in 2020.
This is a latest report, covering the current COVID-19 impact on the market. The pandemic of Coronavirus (COVID-19) has affected every aspect…
Global Human Coagulation Factor VII Market 2018 - NovoNordisk
Eminent Market, recently published a detailed market research study focused on the "Human Coagulation Factor VII Market" across the global, regional and country level. The report provides 360° analysis of "Human Coagulation Factor VII Market" from view of manufacturers, regions, product types and end industries. The research report analyses and provides the historical data along with current performance of the global Human Coagulation Factor VII industry, and estimates the future…