Press release
Decentralized Clinical Trials Market to Reach US$ 34.13 Billion by 2035 Driven by Patient-Centric Trial Models, Remote Monitoring Technologies, and AI-Enabled Clinical Research Platforms
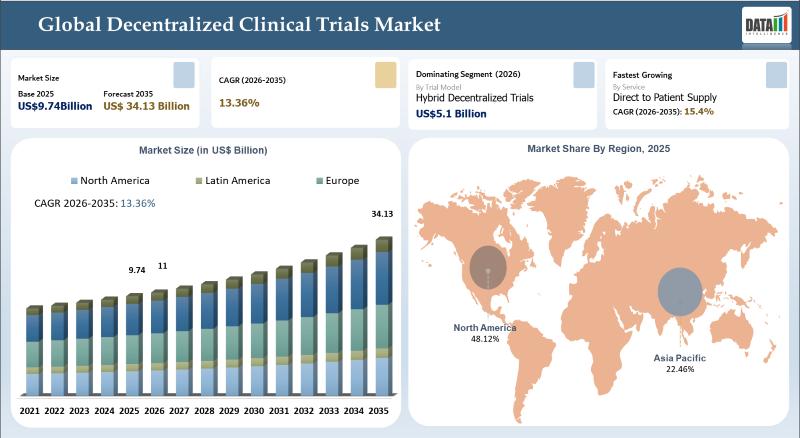
The Global Decentralized Clinical Trials Market was valued at US$ 9.74 billion in 2025 and is expected to reach US$ 34.13 billion by 2035, growing at a CAGR of 13.36% during the forecast period 2026-2035.Growth is driven by the increasing adoption of patient-centric and technology enabled clinical trial models that improve patient recruitment, retention, and accessibility. Decentralized clinical trials leverage digital tools, telemedicine, wearable devices, and remote monitoring technologies to conduct studies outside traditional clinical settings. Additionally, the rising demand for cost efficient and faster trial execution, along with the growing use of real world data and virtual trial platforms, is accelerating market expansion. Supportive regulatory frameworks and advancements in AI, data analytics, and cloud based clinical systems are further fueling the global growth of the decentralized clinical trials market.
Download your exclusive sample report(corporate email gets priority access): https://www.datamintelligence.com/download-sample/decentralized-clinical-trials-market?praneetha
✦ Decentralized Clinical Trials (DCT) Market: Competitive Intelligence
The major global players in the market include IQVIA Inc., ICON plc, Labcorp Holdings Inc., Parexel International Corporation, Thermo Fisher Scientific Inc., Oracle Corporation, Dassault Systèmes SE, Medable Inc., Science 37 Holdings, Inc., Signant Health, Syneos Health, Inc., Medpace Holdings, Inc., Veeva Systems Inc., Clario, Castor, Viedoc Technologies AB, THREAD Research, Medrio, Inc., OpenClinica, LLC, and Clinion IT Services Pvt. Ltd., among others.
The Decentralized Clinical Trials (DCT) Market is driven by contract research organizations (CROs), technology providers, and life sciences companies such as IQVIA, ICON, Parexel, Medable, and Veeva Systems, which offer virtual trial platforms, remote patient monitoring, eConsent, and data management solutions. These technologies enable clinical trials to be conducted outside traditional sites, improving patient recruitment, retention, and accessibility.
These companies' complementary strengths including IQVIA and Labcorp's extensive clinical research capabilities, Medable and Science 37's expertise in decentralized trial platforms, Oracle and Dassault Systèmes' robust data management systems, and Veeva's cloud-based clinical solutions are strengthening competitive positioning. Continuous investments in digital health technologies, remote monitoring tools, AI-driven trial analytics, and patient centric trial designs are driving market growth amid increasing demand for efficient, cost effective, and accessible clinical research solutions.
Ready to scale in this Market ? Connect with the right partners and unlock new growth opportunities :- https://www.datamintelligence.com/partner-identification-enquiry//decentralized-clinical-trials-market?praneetha
✦ New Product Launches
Medable Inc.
Medable expanded its decentralized clinical trial platform with enhanced remote patient monitoring, eConsent, and telemedicine capabilities. The solutions enable seamless virtual trial execution and improved patient engagement. These innovations support faster and more efficient clinical research.
Science 37 Holdings, Inc.
Science 37 introduced advanced DCT solutions with integrated mobile healthcare services and digital patient recruitment tools. The platform focuses on improving accessibility and diversity in clinical trials. These developments enhance trial participation and data collection.
✦ R&D Developments
IQVIA Inc.
IQVIA continued R&D in decentralized trial technologies, focusing on real-world data integration and AI-driven patient analytics. The company is developing solutions that improve trial design and execution. These advancements enhance efficiency and reduce trial timelines.
Parexel International Corporation
Parexel advanced research in hybrid and decentralized trial models, emphasizing digital tools and patient-centric approaches. The company is focusing on improving compliance and data accuracy. These developments support the evolution of modern clinical trials.
✦ Technological Advancements
Remote Patient Monitoring & Telemedicine Integration
The market is witnessing strong adoption of remote monitoring devices and telehealth solutions in clinical trials. These technologies enable continuous data collection and patient engagement. This improves trial efficiency and patient convenience.
AI & Real-World Data (RWD) Integration
Technological progress is enabling AI-driven analytics and integration of real-world data into clinical trial processes. These systems improve patient recruitment, retention, and outcome analysis. This enhances decision making and trial success rates.
✦ M&A / Strategic Developments
Thermo Fisher Scientific Inc.
Thermo Fisher continued strategic expansions in clinical research services, including decentralized trial capabilities. The company is focusing on integrating digital tools into its CRO services. These initiatives strengthen its position in the DCT market.
Oracle Corporation (Oracle Health Sciences)
Oracle pursued strategic developments in clinical trial management systems with decentralized capabilities. The company is focusing on cloud based platforms and data integration. This supports efficient and scalable clinical trial operations.
Request Custom Intelligence Report: https://www.datamintelligence.com/customize//decentralized-clinical-trials-market?praneetha
✦ Market Segmentation
By Study Type
The market is segmented into Interventional Trials 50%, Observational Trials 30%, Expanded Access Trials 10%, and Others 10%, with interventional trials dominating due to their extensive use in drug development and regulatory approvals.
By Component
The market includes Software Solutions 70% and Hardware Solutions 30%, with software dominating due to increasing adoption of virtual trial platforms, data management systems, and remote monitoring tools.
By Therapeutic Area
The market is segmented into Oncology 25%, Cardiovascular Diseases 15%, Infectious Diseases 14%, Neurology 12%, Respiratory Diseases 10%, Metabolic Disorders 10%, Rare Diseases 8%, and Others 6%, with oncology dominating due to high number of clinical trials and increasing focus on personalized medicine.
By End-User
The market includes Pharmaceutical & Biotechnology Companies 55%, Contract Research Organizations (CROs) 25%, Academic & Research Institutes 15%, and Others 5%, with pharmaceutical and biotechnology companies dominating due to high investment in drug development and clinical research.
✦ Regional Analysis
North America - 40% Share
North America leads with 40% share driven by advanced healthcare infrastructure, strong clinical research ecosystem, and early adoption of decentralized trial models.
Europe - 25% Share
Europe holds 25% share supported by regulatory support and increasing adoption of digital health technologies in clinical trials.
Asia-Pacific - 22% Share
Asia-Pacific accounts for 22% share due to growing clinical research activities, large patient pool, and cost advantages.
Purchase Corporate License | Market Intelligence:
https://www.datamintelligence.com/buy-now-page?report=decentralized-clinical-trials-market?praneetha
✅ Competitive Landscape
✅ Technology Roadmap Analysis
✅ Sustainability Impact Analysis
✅ KOL / Stakeholder Insights
✅ Consumer Behavior & Demand Analysis
✅ Import-Export Data Monitoring
✅ Live Market & Pricing Trends
Fabian
DataM Intelligence 4market Research LLP
6th Floor, M2 Tech Hub, DataM Intelligence 4market Research LLP, Lalitha Nagar, Habsiguda, Secunderabad, Hyderabad, Telangana 500039
USA: +1 877-441-4866
UK: +44 161-870-5507
Email: fabian@datamintelligence.com
About Us -
DataM Intelligence is a Market Research and Consulting firm that provides end-to-end business solutions to organizations from Research to Consulting. We, at DataM Intelligence, leverage our top trademark trends, insights and developments to emancipate swift and astute solutions to clients like you. We encompass a multitude of syndicate reports and customized reports with a robust methodology.
Our research database features countless statistics and in-depth analyses across a wide range of 6300+ reports in 40+ domains creating business solutions for more than 200+ companies across 50+ countries; catering to the key business research needs that influence the growth trajectory of our vast clientele.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Decentralized Clinical Trials Market to Reach US$ 34.13 Billion by 2035 Driven by Patient-Centric Trial Models, Remote Monitoring Technologies, and AI-Enabled Clinical Research Platforms here
News-ID: 4485770 • Views: …
More Releases from DataM Intelligence 4Market Research LLP

Luxury Car Market (2026-2033) | Premium Vehicles, High-End EVs, ADAS, Smart Inte …
DataM Intelligence has released a new research report titled "Luxury Car Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…

Smart Thermometers Market (2026-2033) |Digital Health, Connected Devices, Real-T …
DataM Intelligence has released a new research report titled "Smart Thermometers Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…

Cleanroom Technologies Market (2026-2033) | Contamination Control, HEPA Filters, …
DataM Intelligence has released a new research report titled "Cleanroom Technologies Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…

Agentic AI Market (2026-2035) | Autonomous AI, Real-Time Decisioning, Cognitive …
DataM Intelligence has released a new research report titled "Agentic AI Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…
More Releases for Trials
Clinical Trials Management System Market Trends: How Decentralized Clinical Tria …
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Clinical Trials Management System Market Size, Share & Trends Analysis Report By Solution Type (Enterprise and Site based), By Delivery Mode (Web & Cloud-based, On-premise), By Component (Software, Services), By End-user (Pharmaceutical and Biotechnology Firms, Medical Device Firms, CROs)- Market Outlook And Industry Analysis 2031"
The global Clinical Trials Management System market is estimated to reach…
Brazil Clinical Trials Market ANVISA Brazil Guidelines Brazil Clinical Trials Re …
Brazil Cancer Drugs Clinical Trials Insight 2024 Report Offering:
• Brazil Clinical Trials Market Opportunity 2024 and 2030 (In US$ Billion)
• Clinical Trials Regulatory Framework In Brazil
• Total Number of Cancer Drugs In Clinical Trials In Brazil
• Total Number Of Cancer Drugs Approved In Brazil
• 400 Pages Clinical Trials Insight On All Cancer Drugs In Clinical Trials By Company, Indication and Phase
• 80 Pages Clinical Insight On All Cancer Drugs Approved in Market By Company and Indication
• Insight…
Clinical Trials Management System Market Clinical Trials Management System Marke …
InsightAce Analytic announces the release of a market assessment report on the "Global Clinical Trials Management System Market Size, Share & Trends Analysis Report By Solution Type (Enterprise and Site based), By Delivery Mode (Web & Cloud-based, On-premise), By Component (Software, Services), By End-user (Pharmaceutical and Biotechnology Firms, Medical Device Firms, CROs)- Market Outlook And Industry Analysis 2031.
The global Clinical Trials Management System market is estimated to reach over USD…
Revolutionizing Clinical Trials: The Evolution of eCOA, eSource, and the Clinica …
The global eCOA, eSource & clinical trials market is valued at US$ 48 billion in 2023 and is expected to reach a market size of US$ 104 billion by the end of 2033, expanding rapidly at a CAGR of 8% over the next ten years. Worldwide demand for eCOA (electronic clinical outcome assessment) solutions is predicted to rise at a CAGR of 8.2% over the forecast period.
In the dynamic landscape…
Virtual Clinical Trials Market - Bridging the Gap: Virtual Clinical Trials Revol …
Newark, New Castle, USA - The latest report from Growth Plus Reports analyzes the production, potential applications, demand, major manufacturers, and SWOT analysis of the global Virtual Clinical Trials Market.
Virtual Clinical Trials Market: https://www.growthplusreports.com/report/virtual-clinical-trials-market/9106
The Virtual Clinical Trials Market Report assists in determining the optimum distribution methods for certain products as well as possible markets for future product launches. The report also analyses the purchase and supply trends that influence the…
Clinical Trials Market: Coronavirus Pandemic Pushes Sponsors, Patients to Adopt …
Clinical trials Market: Introduction
According to the report, the global clinical trials market was valued over US$ 46.7 Bn in 2019 and is projected to expand at a CAGR of ~5% from 2020 to 2030. High prevalence and increase in incidence rate of chronic diseases, and rise in R&D activities in biotechnology & pharmaceuticals industries are anticipated to drive the global clinical trials market from 2020 to 2030. North America is projected to…