Press release
Global Drivers Analysis: The Rapid Evolution of the Human Immunodeficiency Viruses (HIV) Clinical Trials Market
The human immunodeficiency viruses (HIV) clinical trials market is positioned for significant growth over the coming years, driven by advancements in treatment options and innovative research methodologies. As the focus on developing effective therapies intensifies, this market is set to expand substantially, presenting new opportunities and challenges for stakeholders involved in HIV clinical research.Projected Expansion of the Human Immunodeficiency Viruses (HIV) Clinical Trials Market by 2030
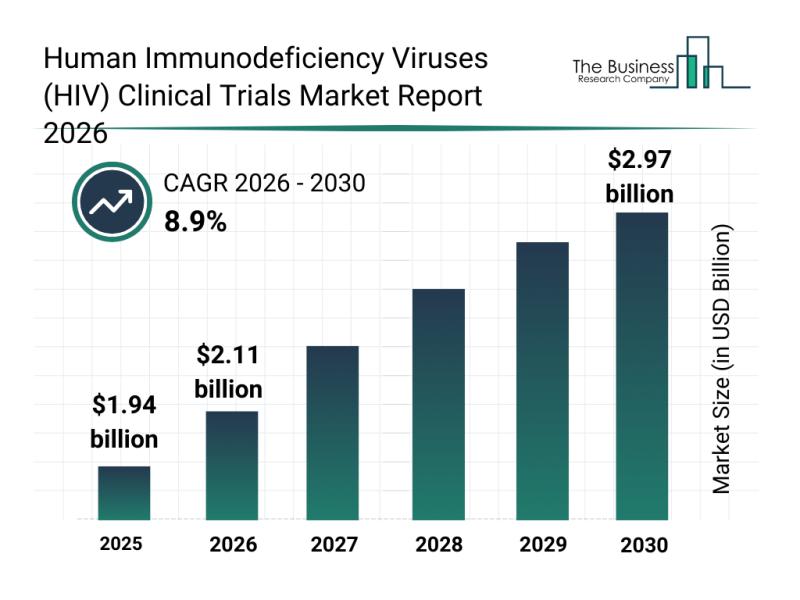
The HIV clinical trials market is forecasted to reach a valuation of $2.97 billion by 2030, growing at a compound annual growth rate (CAGR) of 8.9%. This upward trajectory is supported by several factors, including the ongoing development of both curative and preventive HIV treatments, increased adoption of adaptive trial designs that allow for more flexible research frameworks, and a surge in vaccine-related HIV trials. Furthermore, advancements such as the integration of artificial intelligence in clinical studies and a heightened focus on assessing long-term safety and efficacy continue to propel market growth. Key trends anticipated during this period encompass the rising use of decentralized clinical trial models, enhanced digital tools for patient recruitment, greater emphasis on long-acting HIV therapies, broader incorporation of real-world evidence, and more sophisticated data analytics for trial management.
Download a free report of the human immunodeficiency viruses (hiv) clinical trials market report:
https://www.thebusinessresearchcompany.com/report/human-immunodeficiency-viruses-hiv-clinical-trials-global-market-report?utm_source=OpenPR&utm_medium=Paid&utm_campaign=Apr_PR
Top Players Steering the Human Immunodeficiency Viruses (HIV) Clinical Trials Market
The HIV clinical trials sector features several influential companies shaping its landscape. Leading organizations in this market include Johnson & Johnson, F. Hoffmann-La Roche Ltd., Pfizer Inc., AbbVie Inc., Sanofi S.A., Thermo Fisher Scientific Inc., Amgen Inc., Gilead Sciences Inc., Merck & Co., IQVIA Holdings Inc., ICON plc, Moderna Inc., ViiV Healare Limited, HOOKIPA Pharma Inc., Excision BioTherapeutics Inc., American Gene Technologies International Inc., Bionor Holding AS, Aelix Therapeutics, Immuno Cure BioTech Private Limited, and ImmunityBio.
In a significant development in July 2025, Syneos Health, a US-based pharmaceutical commercial company specializing in HIV clinical trials, was acquired by a trio of investment firms-Elliott Investment Management L.P., Patient Square Capital LP, and Veritas Capital Fund Management L.L.C.-for $7.1 billion. This acquisition is aimed at accelerating Syneos Health's growth trajectory, improving service delivery to clients, and positioning the company as a technology-driven leader in biopharma solutions.
View the full human immunodeficiency viruses (hiv) clinical trials market report:
https://www.thebusinessresearchcompany.com/report/human-immunodeficiency-viruses-hiv-clinical-trials-global-market-report?utm_source=OpenPR&utm_medium=Paid&utm_campaign=Apr_PR
Key Factors Positively Influencing the Human Immunodeficiency Viruses (HIV) Clinical Trials Market
One important force behind growth in the HIV clinical trials market is the increasing use of cutting-edge technologies to enhance study design and execution. The incorporation of artificial intelligence and digital tools is streamlining procedures and improving patient recruitment accuracy, enabling trials to be more efficient and adaptive.
Another crucial driver is the focus on long-acting HIV therapies, which are gathering momentum and attracting more clinical research attention. These therapies aim to improve adherence and treatment outcomes, reflecting a shift in trial priorities toward innovations that address patient convenience and long-term disease management.
Important Developments in Related Technologies Impacting the Clinical Trials Sector
Companies involved in scientific instrumentation are also contributing indirectly to the HIV clinical trials market by developing advanced analytical tools. For example, in June 2023, Bruker Corporation introduced the EVOQ DART-TQ+, an innovative triple-quadrupole mass spectrometer designed for rapid, chromatography-free mass spectrometry. This instrument is tailored for sectors including food and beverage, forensic toxicology, industrial, and environmental analysis, offering simplified workflows with enhanced sensitivity and accuracy. Such technologies support the broader clinical research ecosystem by enabling faster, cost-effective, and environmentally sustainable analysis methods.
Detailed Segmentation of the Human Immunodeficiency Viruses (HIV) Clinical Trials Market
This market is categorized into various segments to comprehensively address the diversity of clinical trials:
1. By Phase: Phase I (including first-in-human and safety/tolerability studies), Phase II (dose-finding and efficacy studies), Phase III (pivotal and comparative efficacy trials), Phase IV (post-marketing surveillance and long-term safety studies), and Preclinical (in vitro and animal studies).
2. By Drug Type: Antiretroviral Therapy (ART), Pre-Exposure Prophylaxis (PrEP), Post-Exposure Prophylaxis (PEP), Long-Acting Injectable Antiretrovirals, and Experimental Drugs.
3. By Study Design: Interventional Studies, Observational Studies, and Expanded Access Studies.
4. By Patient Population: Adults, Pediatric Patients, Seniors, Pregnant Women, and High-Risk Populations.
5. By End User: Pharmaceutical Companies, Research Institutes, Hospitals, and Other End Users.
These segments provide a clear framework for understanding the various dimensions along which HIV clinical trials are conducted and managed, reflecting the complexity and specialization necessary to advance HIV treatment options.
Reach out to us:
The Business Research Company: https://www.thebusinessresearchcompany.com/,
Americas +1 310-496-7795,
Europe +44 7882 955267,
Asia & Others +44 7882 955267 & +91 8897263534,
Email us at info@tbrc.info.
Follow Us On:
LinkedIn: https://in.linkedin.com/company/the-business-research-company,
Twitter: https://twitter.com/tbrc_info,
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Learn More About The Business Research Company
With over 17500+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead.Our flagship product, the Global Market Model (GMM), is a premier market intelligence platform delivering comprehensive and updated forecasts to support informed decision-making.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Drivers Analysis: The Rapid Evolution of the Human Immunodeficiency Viruses (HIV) Clinical Trials Market here
News-ID: 4480313 • Views: …
More Releases from The Business Research Company
Market Driver Insights: The Impact of Recent Advances on the Sotrovimab or Xevud …
The Sotrovimab or Xevudy market is drawing significant attention as advancements in antibody therapies and pandemic preparedness shape its future. With growing investments and evolving clinical strategies, this market is expected to undergo considerable transformation by 2030. Let's explore the current market size, key players, driving factors, and relevant segments that define the Sotrovimab or Xevudy landscape.
Anticipated Market Growth and Drivers in the Sotrovimab or Xevudy Market
The projected…
Competitive Landscape: Key Market Leaders and New Entrants in the Acute Intermit …
The acute intermittent porphyria market is positioned for significant advancement in the coming years, driven by ongoing innovations and increasing awareness surrounding this rare condition. As research and treatment options evolve, the market is poised to offer more effective management solutions that improve patient outcomes and accessibility.
Forecasted Market Size and Growth Trajectory of the Acute Intermittent Porphyria Market
The acute intermittent porphyria market is expected to reach a valuation…
Segmentation, Major Trends, and Competitive Overview of the Skyrizi Market
The Skyrizi market is positioned for significant growth as it expands into new inflammatory disease areas and benefits from increased investment in advanced biologic therapies. With patients increasingly favoring convenient dosing schedules and healthcare providers emphasizing long-term remission, Skyrizi is set to gain wider adoption. Key trends driving this market include the growing use of IL-23 targeted treatments, the broadening of applications across various autoimmune conditions, the preference for biologics…
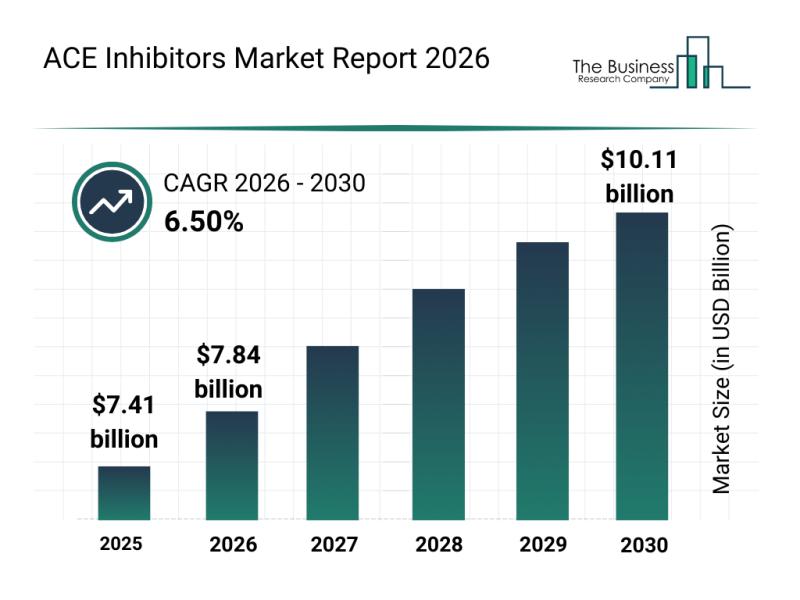
Analysis of the ACE Inhibitors Market: Industry Structure, Emerging Trends, and …
The ACE inhibitors market is set to experience significant growth in the coming years, driven by advancements in medical treatment and expanding healthcare access. With increasing demand for tailored therapies and digital health integration, this segment of the pharmaceutical industry is evolving rapidly. Let's explore the market's value projections, leading companies, emerging trends, and key segments shaping its future.
Projected Market Value and Growth Rate of the ACE Inhibitors Market …
More Releases for HIV
HIV Drugs Market - Defeating HIV Together: Advancing Treatment Options for a Bri …
Newark, New Castle, USA: The "HIV Drugs Market" provides a value chain analysis of revenue for the anticipated period from 2022 to 2030. The report will include a full and comprehensive analysis of the business operations of all market leaders in this industry, as well as their in-depth market research, historical market development, and information about their market competitors
HIV Drugs Market: https://www.growthplusreports.com/report/hiv-drugs-market/7792
This latest report researches the industry structure, sales, revenue,…
HIV-Associated Lipodystrophy Treatment Market - Increasing prevalence of HIV is …
HIV-associated lipodystrophy also known as lipodystrophy is a syndrome that occurs in HIV-infected patients. It is characterized by loss of subcutaneous fat from face, buttocks, arms and legs. Although the exact cause of HIV-associated lipodystrophy is not fully elucidated, some research evidence reported that it occurs in HIV-infected patients who are under antiretroviral medications. According to an article published in National Center for Biotechnology Information (NCBI) in 2014, prevalence of…
Global HIV Drugs Market | Global HIV Drugs Industry | Global HIV Drugs Market Re …
Human immunodeficiency Virus (HIV) could be a chronic and severe sickness which might be transferred from one person to a different through blood-to-blood and sexual contact. it's a deadly disease that attacks immune cells called CD-4 cells, creating body vulnerable to infections and alternative diseases. Over the years, the rising prevalence of HIV sickness worldwide has completely influenced the demand for HIV medicine. HIV medicine facilitate in preventing the multiplication…
HIV Therapeutics Market– South Africa's Aspen launches three-in-one HIV drug
Recent Developments
Aspen Pharma care, a South Africa’s drug maker has launched a triple combination of tablet for the treatment of HIV in the country where the HIV virus is the most prevalent. The company's new Emdolten drug is a once a day tablet which is in the form of dolutegravir, an antiretroviral medication that balances the drug’s resistance. The company has launched Aspen Stavudine which was its first generic ARV…
HIV Vaccine Market HIV Vaccine Clinical Pipeline Report 2022
For Report Sample Contact: neeraj@kuickresearch.com or +91-11-47067990
Report Table of Contents
Introduction to Human Immunodeficiency Virus (HIV) Vaccines
1.1 Overview
1.2 Antiquity of HIV Vaccine
Need for the Development of HIV Vaccine
Primer of HIV inside the Body
3.1 Inclusion of HIV Virus into the System
3.2 Interaction of HIV with Host
3.3 Eradication of HIV Virus
HIV Vaccine Development Process
4.1 Introduction
…
Global HIV Vaccine Market & HIV Vaccine Clinical Trial Outlook 2022
Worldwide, around the 35 Million of the people are currently infected with the HIV and about 30 Million of the people died because of the AIDS infection. There is no human example of clearing an HIV infection naturally. HIV virus makes copies of it very quickly, many types of HIV exist and new types of virus are continue to rise. Many scientists are still trying to understand the specific ways…