Press release
Guest Post: Electronic Trial Master File (eTMF) Systems Market
IntroductionThe Electronic Trial Master File (eTMF) system is a digital platform designed to manage, store, and track clinical trial documentation in the pharmaceutical, biotechnology, and clinical research industries. Traditionally, clinical trial master files were paper-based systems used to store essential documents related to clinical trials, including protocols, informed consent forms, and regulatory submissions. With the rapid advancement of technology, the eTMF system has become an essential tool, enabling better organization, accessibility, and compliance for clinical trials. As the demand for faster, more efficient, and cost-effective clinical trials increases, eTMF systems have gained significant traction in the market. This guest post explores the Electronic Trial Master File (eTMF) Systems market, covering the market size, share, opportunities, challenges, demand, and trends.
For more information:
https://www.databridgemarketresearch.com/reports/global-electronic-trial-master-file-etmf-systems-market
Market Size
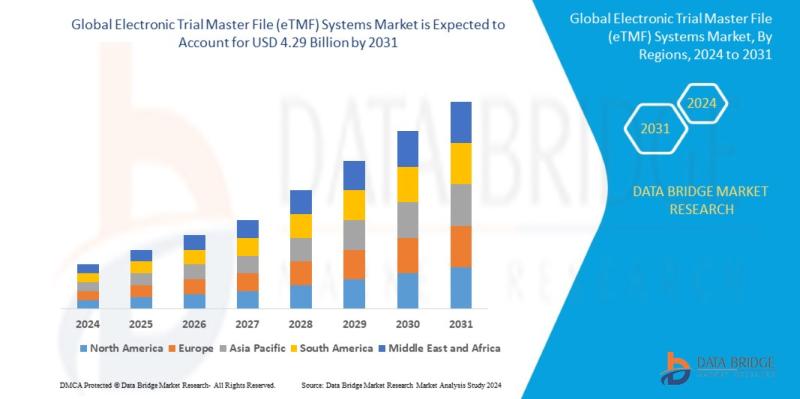
Global electronic trial master file (ETMF) systems market size was valued at USD 1.63 billion in 2023 and is projected to reach USD 4.29 billion by 2031, with a CAGR of 12.9% during the forecast period of 2024 to 2031. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, geographically represented company-wise production and capacity, network layouts of distributors and partners, detailed and updated price trend analysis and deficit analysis of supply chain and demand.
Market Share
The Electronic Trial Master File systems market is fragmented and consists of a mix of established companies and emerging players, each offering a range of solutions tailored to different clinical trial needs. Key players in the eTMF market include Veeva Systems, Parexel International Corporation, Medidata Solutions, and Pharmaceutical Product Development (PPD), among others. These companies offer cloud-based eTMF platforms that provide users with secure access to clinical trial documents and ensure compliance with regulatory standards.
Veeva Systems holds the largest market share in the eTMF systems market due to its strong presence in the cloud-based software space and its comprehensive suite of life sciences solutions. Veeva Vault QMS and Veeva Vault eTMF are widely adopted by pharmaceutical companies, contract research organizations (CROs), and clinical research sites for managing and maintaining trial documentation. Veeva's user-friendly interfaces, regulatory compliance features, and integration with other Veeva products have made it a preferred solution for large and small pharmaceutical companies alike.
Medidata Solutions, now a part of Dassault Systèmes, is another prominent player in the market, offering cloud-based eTMF solutions that improve the efficiency of clinical trials by providing seamless collaboration among stakeholders. Medidata's eTMF system is designed to support the complexities of global clinical trials and offers integration with other clinical trial management tools, improving overall trial operations.
The demand for cloud-based eTMF solutions has been growing rapidly due to their flexibility, scalability, and cost-effectiveness, which has contributed to the increasing market share of these companies. North America holds the largest share of the eTMF market due to the strong presence of pharmaceutical companies and CROs, while Europe and the Asia-Pacific region are witnessing steady growth driven by the expansion of the life sciences industry and the increasing adoption of digital solutions in clinical trial management.
Market Opportunities
The Electronic Trial Master File systems market offers several growth opportunities, driven by the need for more efficient and transparent clinical trial processes. The first opportunity is the increasing demand for end-to-end clinical trial management solutions. Pharmaceutical companies and contract research organizations are seeking integrated platforms that combine eTMF systems with other clinical trial management tools, such as clinical data management, monitoring, and reporting. The integration of eTMF systems with other software solutions helps streamline workflows, improve data consistency, and reduce the risk of errors.
Another opportunity exists in the growing trend toward decentralized clinical trials (DCTs). As clinical trials move away from traditional, centralized models and incorporate virtual or remote monitoring, the need for digital platforms like eTMF systems has become even more important. eTMF systems can facilitate remote access to trial documentation, allowing stakeholders to collaborate and make data-driven decisions in real time, regardless of location. The increasing adoption of decentralized clinical trials offers a significant opportunity for eTMF providers to expand their offerings.
The market is also seeing increasing interest in the use of artificial intelligence and machine learning within eTMF systems. AI and ML can be leveraged to automate document indexing, streamline data retrieval, and improve document compliance checks. These technologies can reduce the time and effort required for document management while improving the accuracy and quality of clinical trial documentation. As AI and ML continue to evolve, there is a growing opportunity for eTMF vendors to incorporate these capabilities into their platforms, providing more sophisticated and efficient solutions.
Moreover, the rising demand for regulatory compliance and real-time tracking of clinical trial documents presents an ongoing opportunity. Regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) require organizations to maintain accurate and up-to-date records throughout the clinical trial process. eTMF systems can help organizations comply with these requirements by providing an auditable, secure platform for storing and managing trial documentation.
Market Challenges
Despite the significant opportunities, the eTMF systems market faces several challenges. One of the primary challenges is the high cost of implementation and maintenance of eTMF systems, particularly for small and mid-sized organizations. While large pharmaceutical companies and CROs can afford to invest in comprehensive eTMF solutions, smaller organizations may face financial constraints. This disparity in affordability can limit the market's growth potential, especially in emerging markets.
Another challenge is the complexity of transitioning from paper-based systems to digital solutions. Organizations with long-established paper-based processes may find it difficult to implement eTMF systems, requiring substantial training, change management, and infrastructure upgrades. The transition to a digital system can be time-consuming and costly, and some organizations may hesitate to make the switch due to concerns about the disruption it could cause to ongoing clinical trials.
Data security and privacy concerns also pose a challenge to the eTMF market. Clinical trial data is highly sensitive and must be protected from unauthorized access or cyberattacks. Ensuring compliance with regulations such as the Health Insurance Portability and Accountability Act (HIPAA) in the U.S. and the General Data Protection Regulation (GDPR) in the European Union is critical for eTMF system providers. Any breaches in data security or failure to comply with regulatory requirements could result in severe penalties and damage to a company's reputation.
Additionally, there are challenges associated with the integration of eTMF systems with other clinical trial management tools. While integration is beneficial, it can be complex and may require customization to ensure compatibility with existing systems. This can increase the cost and time needed to implement the solution, which could deter potential customers from adopting eTMF platforms.
Market Demand
The demand for Electronic Trial Master File systems is driven by the increasing need for improved efficiency, transparency, and compliance in clinical trials. As clinical trials become more complex and global, organizations are seeking digital solutions that enable better collaboration, document tracking, and real-time access to trial data. The demand for eTMF systems is particularly strong in the pharmaceutical and biotechnology industries, where regulatory requirements are stringent, and the stakes of clinical trials are high.
The demand is also influenced by the growing trend toward digitalization in clinical trials. Pharmaceutical companies and contract research organizations are moving away from paper-based systems in favor of digital platforms that offer greater flexibility, scalability, and security. Cloud-based eTMF solutions are in high demand due to their ease of implementation, lower upfront costs, and the ability to support remote workforces.
In addition, the rising number of clinical trials being conducted, especially in emerging markets, contributes to the increasing demand for eTMF systems. These systems help organizations streamline trial processes, manage documentation more efficiently, and ensure compliance with regulatory standards.
Market Trends
Several key trends are shaping the Electronic Trial Master File systems market. One major trend is the shift toward cloud-based eTMF solutions. Cloud-based platforms offer benefits such as scalability, flexibility, and real-time collaboration, making them an attractive option for pharmaceutical companies and CROs. These solutions also allow for better data storage, management, and retrieval, ensuring that clinical trial documentation is secure and easily accessible.
Another trend is the growing adoption of decentralized clinical trials (DCTs), which has increased the demand for eTMF systems that can support remote collaboration and data access. As clinical trials move toward more flexible, patient-centric models, eTMF systems must adapt to support these new approaches.
The integration of artificial intelligence (AI) and machine learning (ML) into eTMF systems is another notable trend. These technologies can automate routine tasks, improve document indexing and retrieval, and enhance compliance monitoring, all of which contribute to greater efficiency in clinical trial management.
Browse Trending Reports:
https://newsresearch12.blogspot.com/2024/12/enzyme-linked-immunospot-assays-market.html
https://newsresearch12.blogspot.com/2024/12/hypoparathyroidism-treatment-market.html
https://newsresearch12.blogspot.com/2024/12/wire-rope-sling-market-size-share.html
https://newsresearch12.blogspot.com/2024/12/cardiac-rhythm-management-devices_17.html
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email: corporatesales@databridgemarketresearch.com"
About Data Bridge Market Research:
Data Bridge set forth itself as an unconventional and neoteric Market research and consulting firm with unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market. Data Bridge endeavors to provide appropriate solutions to the complex business challenges and initiates an effortless decision-making process.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Guest Post: Electronic Trial Master File (eTMF) Systems Market here
News-ID: 3791003 • Views: …
More Releases from Data Bridge Market Research

Scented Candle Market Shows Strong Growth Driven by Wellness and Home Décor Tr …
The global scented candle market is on track for significant expansion, increasing from an estimated USD 3.60 billion in 2024 to USD 6.00 billion by 2032, registering a strong CAGR of 6.60%. Rising consumer interest in home ambiance, wellness, and premium lifestyle products continues to drive market demand.
Get More Detail: https://www.databridgemarketresearch.com/reports/global-scented-candle-market
Market Growth Drivers
The scented candle market has evolved beyond being just a decorative item. Key growth factors include:
Home Fragrance &…
Water Treatment System Market: Sustaining the Future of Clean Water
Introduction
Understanding Water Treatment Systems
Water treatment systems are designed to purify and disinfect water for various uses-drinking, industrial processes, irrigation, and wastewater reuse. These systems eliminate contaminants such as bacteria, viruses, heavy metals, chemicals, and particulates, making water safe and sustainable for consumption and use.
Importance in Global Sustainability
Clean water is essential to life and industrial progress. With growing water demand and pollution, water treatment systems are now critical infrastructure across the…

Veterinary X-Ray Market Size, Analysis, Scope, Demand, Opportunities, Statistics
According to Data Bridge Market Research The global Veterinary X-Ray market size was valued at USD 915.19 million in 2024 and is projected to reach USD 1576.00 million by 2032, with a CAGR of 7.03 % during the forecast period of 2025 to 2032.
With increasing globalization and digital disruption, the Equine X-Ray Solutions Market is expanding across multiple industries, . Market research data indicates that businesses in the Companion Animal…

Veterinary X-Ray Market Size, Analysis, Scope, Demand, Opportunities, Statistics
According to Data Bridge Market Research The global Veterinary X-Ray market size was valued at USD 915.19 million in 2024 and is projected to reach USD 1576.00 million by 2032, with a CAGR of 7.03 % during the forecast period of 2025 to 2032.
With increasing globalization and digital disruption, the Equine X-Ray Solutions Market is expanding across multiple industries, . Market research data indicates that businesses in the Companion Animal…
More Releases for Electronic
Revolutionizing Electronic Warfare: The Power of Cognitive Electronic Warfare Sy …
According to the report published by Allied Market Research, " Cognitive Electronic Warfare System Market by Capability (Electronic Attack, Electronic Protection, Electronic Support, Electronic Intelligence) and by Platform (Naval, Airborne, Land, Space): Global Opportunity Analysis and Industry Forecast, 2023-2032 "Cognitive electronic warfare systems are based on learning action frameworks that use machine learning algorithms and artificial intelligence (AI) to mimic human perception of learning, memory, and judgement. Machine learning, being…
Electronic Cigarettes
Electronic Cigarettes are also known as Vaping, which consist of different type of devices that allow the users to inhale an aerosol that might be nicotine, flavoring and other species. Electronic Cigarettes market is expected to mark significant growth over forecasted period owing to increasing technological advancements, change in lifestyle and consists of various flavours and fragrance. There has been significant rise in number of prevalence of adults using e-cigarettes…
Electronic Voting Machine Market 2019-2025| Top key players: Gaurang Electronic …
This report titled as Electronic Voting Machine Market gives a brief about the comprehensive research and an outline of its growth in the market globally. Electronic voting is voting that uses electronic means to either aid or take care of casting and counting votes. Depending on the particular implementation, e-voting may use standalone electronic voting machines or computers connected to the Internet. It may encompass a range of Internet services,…
Electronic Voting Machine Market 2019-2025| Top key players: Gaurang Electronic …
The Electronic Voting Machine Market requires the prevailing regions in the market during the forecast period. Evidence on the region leading the market are some of the facets that are highlighted under this section of the report. A voting machine is a machine used to register and tabulate votes. The first voting machines were mechanical but it is increasingly more common to use electronic voting machines. Traditionally, a voting machine…
Electronic Wrapper Market Segmented By loading types semi-automatic electronic w …
A Revolutionary Product for the Packaging Industry
The electronic wrapper has been a revolutionizing product in the packaging industry. Electronic wrappers are easy to maintain and comfortable for size changes. The steel built electronic wrappers enable easy access for maintenance and cleaning, thus, gaining a considerable customer base from the packaging industries mainly for food and consumer products. An electronic wrapper is an automated machine, which is programmed to perform a…
Wearable Electronic Devices Market,Wearable Electronic Devices Industry, Global …
Latest industry research report on: Global Wearable Electronic Devices Market : Industry Size, Share, Research, Reviews, Analysis, Strategies, Demand, Growth, Segmentation, Parameters, Forecasts
This report studies the global Wearable Electronic Devices market status and forecast, categorizes the global Wearable Electronic Devices market size (value & volume) by manufacturers, type, application, and region. This report focuses on the top manufacturers in United States, Europe, China, Japan, South Korea and Taiwan and other…