Press release
Electronic Trial Master File (eTMF) Systems Market: An In-Depth Analysis
Introduction:An Electronic Trial Master File (eTMF) is a digital solution for managing and storing essential documents in clinical trials. It provides a centralized system that streamlines the process of document collection, organization, and management throughout the clinical trial lifecycle. eTMF systems enhance compliance, improve collaboration, and facilitate real-time tracking of trial documents. These systems are essential for ensuring the efficiency and accuracy of clinical trials, particularly in an era where regulatory requirements are becoming increasingly stringent. As pharmaceutical companies and contract research organizations (CROs) seek more efficient ways to conduct trials, eTMF systems have gained significant traction.
Market Size:
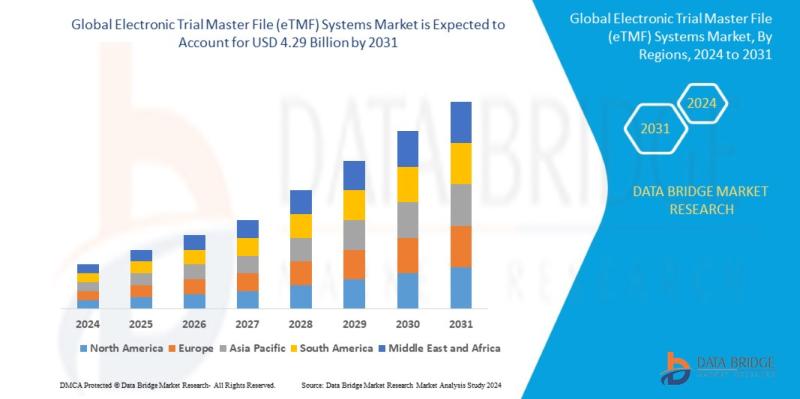
Global electronic trial master file (ETMF) systems market size was valued at USD 1.63 billion in 2023 and is projected to reach USD 4.29 billion by 2031, with a CAGR of 12.9% during the forecast period of 2024 to 2031. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, geographically represented company-wise production and capacity, network layouts of distributors and partners, detailed and updated price trend analysis and deficit analysis of supply chain and demand.
https://www.databridgemarketresearch.com/reports/global-electronic-trial-master-file-etmf-systems-market
Some of the major players operating in the market are:
IQVIA Inc.(U.S.)
Labcorp Drug Development (U.S.)
TransPerfect (U.S.)
Oracle (U.S.)
Phlexglobal (U.S.)
SureClinical Inc. (U.S.)
Aurea, Inc. (U.S.)
Veeva Systems (U.S.)
MasterControl Solutions, Inc. (U.S.)
Clinevo Technologies (India)
Mayo Foundation for Medical Education and Research (MFMER) (U.S.)
Montrium Inc. (U.S.)
NCGD Inc. (U.S.)
PharmaVigilance (U.S.)
Market Share:
The eTMF systems market is highly competitive, with several key players holding significant market shares. Veeva Systems, Phlexglobal, TransPerfect, and Oracle are among the leading companies in the market, offering comprehensive eTMF solutions to pharmaceutical companies and CROs. Veeva Systems holds a dominant position due to its extensive product portfolio, which includes cloud-based eTMF systems that are widely adopted by global life sciences organizations.
Geographically, North America holds the largest market share, accounting for over 45% of the global eTMF systems market. The dominance of this region is attributed to the presence of a large number of pharmaceutical companies and the high adoption of advanced technologies in clinical research. Europe follows closely, with significant market share driven by the increasing focus on regulatory compliance and the digitization of clinical trials. The Asia-Pacific region is expected to witness rapid growth in the coming years, as the clinical research industry continues to expand in countries such as China, India, and Japan.
Market Trends:
Several key trends are shaping the eTMF systems market. One of the most notable trends is the increasing adoption of cloud-based eTMF solutions. Cloud-based systems offer several advantages over traditional on-premise systems, including scalability, remote access, and cost-effectiveness. As clinical trials become more global and decentralized, the ability to access trial documents from any location is becoming increasingly important. Cloud-based eTMF systems facilitate real-time collaboration among stakeholders, making it easier to manage complex, multi-site trials.
Another significant trend is the integration of artificial intelligence (AI) and machine learning (ML) into eTMF systems. AI and ML technologies are being used to automate document classification, improve data accuracy, and enhance compliance monitoring. For example, AI-powered algorithms can identify discrepancies in trial documents, flag potential compliance issues, and suggest corrective actions. The use of AI and ML in eTMF systems is expected to increase efficiency and reduce the risk of human error in document management.
The growing focus on regulatory compliance is also shaping the eTMF systems market. Regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) require that trial master files be maintained in a way that ensures accuracy, completeness, and accessibility. eTMF systems help organizations meet these regulatory requirements by providing secure, compliant storage of trial documents and audit-ready records. As regulatory scrutiny increases, the demand for eTMF systems that ensure compliance with global standards is expected to grow.
Market Growth:
The eTMF systems market is expected to experience robust growth in the coming years, driven by several factors. One of the primary drivers of market growth is the increasing complexity of clinical trials. As clinical trials become more complex, with larger patient populations, multiple endpoints, and diverse trial sites, the need for efficient document management becomes critical. eTMF systems provide a centralized platform for managing the vast amount of documentation generated during clinical trials, ensuring that all documents are organized, accessible, and compliant with regulatory requirements.
The growing demand for faster drug development is also contributing to the growth of the eTMF systems market. Pharmaceutical companies are under increasing pressure to bring new drugs to market more quickly, particularly in the face of rising healthcare costs and the need for innovative treatments. eTMF systems streamline the clinical trial process by automating document management tasks, reducing administrative burdens, and improving collaboration among trial stakeholders. The ability to accelerate trial timelines is driving the adoption of eTMF systems across the pharmaceutical industry.
The rising adoption of decentralized and virtual clinical trials is another factor fueling market growth. Decentralized trials, which involve conducting trials at multiple locations or virtually, require robust digital infrastructure to manage trial documents and ensure compliance. eTMF systems are well-suited for decentralized trials, as they enable remote access to trial documents, real-time monitoring, and secure collaboration among geographically dispersed teams. The increasing popularity of decentralized trials is expected to drive demand for eTMF systems in the coming years.
Market Demand:
The demand for eTMF systems is growing rapidly, driven by several factors. One of the primary drivers of demand is the increasing number of clinical trials being conducted worldwide. The global pharmaceutical and biotechnology industries are experiencing a surge in clinical trial activity, driven by the need for new treatments and therapies. As the number of clinical trials increases, so does the need for efficient document management solutions. eTMF systems help organizations manage the growing volume of trial documents, ensuring that they are organized, accessible, and compliant with regulatory standards.
The growing trend towards digital transformation in the life sciences industry is also contributing to the demand for eTMF systems. As pharmaceutical companies and CROs increasingly adopt digital technologies to streamline their operations, the need for digital solutions that enhance the efficiency of clinical trials is becoming more pronounced. eTMF systems are an essential part of this digital transformation, providing a digital platform for managing trial documents and improving collaboration among trial stakeholders.
The increasing focus on data security and privacy is another factor driving demand for eTMF systems. Clinical trials generate a significant amount of sensitive data, and organizations are under growing pressure to ensure that this data is protected from unauthorized access and breaches. eTMF systems offer robust security features, including encryption, user authentication, and audit trails, ensuring that trial documents are stored securely and in compliance with data privacy regulations. The growing emphasis on data security is expected to drive demand for eTMF systems in the coming years.
Factors Driving Growth:
Several factors are driving the growth of the eTMF systems market. The increasing complexity of clinical trials is one of the most significant factors. As trials become more intricate, with larger patient populations, multiple endpoints, and diverse trial sites, the need for efficient document management solutions becomes critical. eTMF systems provide a centralized platform for managing trial documents, ensuring that all documents are organized, accessible, and compliant with regulatory requirements.
The growing demand for faster drug development is another key driver of market growth. Pharmaceutical companies are under increasing pressure to bring new drugs to market more quickly, and eTMF systems help streamline the clinical trial process by automating document management tasks and improving collaboration among trial stakeholders. The ability to accelerate trial timelines is driving the adoption of eTMF systems across the pharmaceutical industry.
The rising adoption of decentralized and virtual clinical trials is also contributing to market growth. Decentralized trials require robust digital infrastructure to manage trial documents and ensure compliance. eTMF systems enable remote access to trial documents, real-time monitoring, and secure collaboration among trial stakeholders, making them well-suited for decentralized trials.
The increasing focus on regulatory compliance is another factor driving the growth of the eTMF systems market. Regulatory agencies have stringent requirements for the collection, storage, and management of trial documents, and eTMF systems help organizations meet these requirements by providing secure, compliant storage of trial documents and audit-ready records. The growing emphasis on regulatory compliance is expected to drive demand for eTMF systems in the coming years.
Conclusion:
The eTMF systems market is poised for significant growth in the coming years, driven by the increasing complexity of clinical trials, the growing demand for faster drug development, and the rising adoption of decentralized trials. As the market continues to evolve, key trends such as the adoption of cloud-based solutions, the integration of AI and ML, and the increasing focus on regulatory compliance are expected to shape the future of the industry. With ongoing advancements in technology and increasing regulatory scrutiny, the eTMF systems market offers significant opportunities for growth and innovation.
Browse Trending Reports:
https://indexsep.blogspot.com/2024/09/picks-disease-treatment-market-size.html
https://indexsep.blogspot.com/2024/09/johanson-blizzard-syndrome-market-size.html
https://indexsep.blogspot.com/2024/09/gelatin-substitutes-market-size-share.html
https://indexsep.blogspot.com/2024/09/functional-confectionery-market-size.html
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email: corporatesales@databridgemarketresearch.com
About Data Bridge Market Research:
Data Bridge set forth itself as an unconventional and neoteric Market research and consulting firm with unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market. Data Bridge endeavors to provide appropriate solutions to the complex business challenges and initiates an effortless decision-making process.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Electronic Trial Master File (eTMF) Systems Market: An In-Depth Analysis here
News-ID: 3650878 • Views: …
More Releases from Data Bridge Market Research

Scented Candle Market Shows Strong Growth Driven by Wellness and Home Décor Tr …
The global scented candle market is on track for significant expansion, increasing from an estimated USD 3.60 billion in 2024 to USD 6.00 billion by 2032, registering a strong CAGR of 6.60%. Rising consumer interest in home ambiance, wellness, and premium lifestyle products continues to drive market demand.
Get More Detail: https://www.databridgemarketresearch.com/reports/global-scented-candle-market
Market Growth Drivers
The scented candle market has evolved beyond being just a decorative item. Key growth factors include:
Home Fragrance &…
Water Treatment System Market: Sustaining the Future of Clean Water
Introduction
Understanding Water Treatment Systems
Water treatment systems are designed to purify and disinfect water for various uses-drinking, industrial processes, irrigation, and wastewater reuse. These systems eliminate contaminants such as bacteria, viruses, heavy metals, chemicals, and particulates, making water safe and sustainable for consumption and use.
Importance in Global Sustainability
Clean water is essential to life and industrial progress. With growing water demand and pollution, water treatment systems are now critical infrastructure across the…

Veterinary X-Ray Market Size, Analysis, Scope, Demand, Opportunities, Statistics
According to Data Bridge Market Research The global Veterinary X-Ray market size was valued at USD 915.19 million in 2024 and is projected to reach USD 1576.00 million by 2032, with a CAGR of 7.03 % during the forecast period of 2025 to 2032.
With increasing globalization and digital disruption, the Equine X-Ray Solutions Market is expanding across multiple industries, . Market research data indicates that businesses in the Companion Animal…

Veterinary X-Ray Market Size, Analysis, Scope, Demand, Opportunities, Statistics
According to Data Bridge Market Research The global Veterinary X-Ray market size was valued at USD 915.19 million in 2024 and is projected to reach USD 1576.00 million by 2032, with a CAGR of 7.03 % during the forecast period of 2025 to 2032.
With increasing globalization and digital disruption, the Equine X-Ray Solutions Market is expanding across multiple industries, . Market research data indicates that businesses in the Companion Animal…
More Releases for Master
Online Master of Social Work: St. Bonaventure University Unveils Flexible, Conve …
Image: https://www.globalnewslines.com/uploads/2025/02/1739277381.jpg
St. Bonaventure University recently launched its clinically-focused online Master in Social Work program, combining academic excellence with flexible, industry-oriented education.
Building on its Franciscan heritage of community values, St. Bonaventure University sets a new benchmark in higher education with its online Master in Social Work [https://online.sbu.edu/] program. Designed for individuals without a social work degree, the program offers a flexible format, allowing students to work while preparing for a career…
Why Master Data Management Matters
In today's data-driven world, businesses are drowning in information. Customer records sprawl across siloed systems, product details differ between departments, and supplier data lacks consistency. This fragmented data landscape leads to errors, inefficiencies, and ultimately, poor decision-making.
Enter Master Data Management (MDM), the hero businesses need to conquer their data chaos. MDM is a strategic approach that ensures the uniformity, accuracy, and completeness of core business data, often referred to…
Performance of a great master
Havana, February 21, 2020 - The researcher in charge of the project "Fidel Mic� Catalogue Raisonn�" is pleased to announce the public performance of the great master from February 21 to 28. Mic� will be painting at the private gallery "Humidores Habana" (Bulevar de San Rafael, Centro Habana, La Habana, Cuba) to show his exquisite technique to create high quality landscape paintings. Access will be free immediately after inauguration ceremony…
Master Data Management Market: Master Data Management Investments in BFSI Sector …
As the amount of data generated by organizations multiples rapidly, organizations can no longer afford to delay investments in master data management solutions. This will help the opportunity in the global master data management market to spiral to US$37.97 bn by 2024, rising at a 27.25% CAGR from 2016 to 2024. The market had a valuation of US$4.35 bn in 2015. Consolidation among companies is helping this market blaze ahead,…
Global Master Control Switchers Market
Key players in the master control switchers market include Evertz Microsystems, Ltd, Harris Corporation, Grass Valley USA, LLC, PESA, Miranda Technologies, Inc., Pixel Power, Inc., Utah Scientific, Inc and Snell Group.
Master control switchers are an essential part of television and video broadcast that aggregate programming feeds from different sources such as audio and video. Global emergence of broadcasting industry forced operational cost reduction and technological enhancements in the same,…
Master Equipment Appraisers now has 5 Master Certified Machinery & Equipment App …
Master Equipment Appraisers, a reputable appraisal service provider unveiled that they now have 5 of the 23 master certified machinery and equipment appraisers in the US. Having such industry-best appraisers, the company offers the best appraisals one could ever get in the area.
Master Equipment Appraisers is a leading firm offering marketing and consulting services such as business buy/sell brokerage, commercial real estate appraisal, business valuation and equipment appraisal services. They…