Press release
GMP Storage Market Predicted to Reach US$ 11,019.9 Million at a CAGR of 5.3% by 2034
The GMP storage industry in the United States is projected to have a market value of approximately US$ 2,844 million in 2024, representing a dominant share of 87.7% in the North American region. The sales of GMP storage are expected to demonstrate a Compound Annual Growth Rate (CAGR) of 5.3% until the year 2034.The worldwide GMP storage market is approximated to reach a valuation of US$ 6,568.3 million in 2024. Forecasts suggest a noteworthy expansion at a Compound Annual Growth Rate (CAGR) of 5.3% until 2034. The expected growth trajectory is poised to elevate the market to an impressive scale of US$ 11,019.9 million by 2034. The surge in demand for pharmaceutical and biotechnology products, combined with stringent regulatory standards, has been instrumental in driving this exponential growth in the market.
Download a Sample Copy of This Report:
https://www.factmr.com/connectus/sample?flag=S&rep_id=9300
The pharmaceutical industry is experiencing unprecedented growth, driven by advancements in medical science, increased demand for innovative therapies, and a global focus on healthcare. As pharmaceutical manufacturing expands to meet these demands, the need for stringent quality control measures becomes paramount. Good Manufacturing Practice (GMP) storage facilities have emerged as a critical component in ensuring the integrity and compliance of pharmaceutical products.
Pharmaceutical Manufacturing on the Rise:
The pharmaceutical sector is witnessing a surge in manufacturing activities as the industry responds to the global demand for life-saving medications, vaccines, and biotechnological products. This growth is spurred by factors such as an aging population, the prevalence of chronic diseases, and heightened awareness of health and well-being. Pharmaceutical companies are scaling up production capacities to meet the increasing need for a diverse range of medical treatments.
Importance of GMP Compliance:
GMP guidelines are a set of quality assurance principles that ensure the consistent production and control of pharmaceutical products according to established quality standards. Compliance with GMP regulations is not only a regulatory requirement but also a fundamental aspect of ensuring the safety, efficacy, and quality of pharmaceutical products. GMP storage facilities play a pivotal role in maintaining the integrity of raw materials, intermediates, and finished products throughout the manufacturing process.
Preserving Product Integrity:
GMP storage facilities are designed to meet strict regulatory standards, providing controlled environments for storing pharmaceutical materials under optimal conditions. These facilities help prevent contamination, degradation, and cross-contamination, which are critical factors in maintaining the efficacy and safety of pharmaceutical products. The controlled storage conditions also contribute to extending the shelf life of products, reducing waste, and ensuring a reliable supply chain.
Global Expansion of GMP Storage Market:
The growing emphasis on GMP compliance has led to an increased demand for specialized storage solutions worldwide. The GMP storage market is witnessing substantial growth as pharmaceutical manufacturers recognize the importance of maintaining a robust quality management system. Regions such as North America, Europe, and Asia-Pacific are witnessing significant investments in state-of-the-art GMP storage facilities to support the expanding pharmaceutical manufacturing landscape.
Read More: https://www.factmr.com/report/gmp-storage-market
Competitive Landscape
The global GMP storage market is filled with a multitude of companies, such as ThermoGenesis Holdings, Inc., ThermoFisher Scientific Inc., BioLife Solutions, Inc., Danaher (Cytiva), MEDIPOST, etc. These companies have a strong foothold in the international marketplace due to their prolonged presence in the industry. There is a presence of smaller companies in the market. However, these companies have to face substantial challenges due to the dominance of the industry giants.
Almac Sciences, a member of the Almac Group, officially opened a new custom-built GMP warehouse and dispatch hub at its global headquarters in Craigavon, UK in February 2023. This facility consolidated existing warehouses and supply chain offices, providing support for all active pharmaceutical ingredient (API) manufacturing. Belgian testing solutions provider and one of the leading companies in the GMP storage market, Eurofins has inaugurated its largest biopharma services campus in Genome Valley, Hyderabad, India. The facility will cater to the life sciences, agrisciences, and environmental sectors.
In August 2023, Avantor inaugurated its expanded Singapore Hub, which included newly added cGMP manufacturing and a world-class quality control laboratory. The hub served as a one-stop facility, meeting the needs of customers across Asia, the Middle East, and Africa (AMEA)
Key Segments Covered in GMP Storage Industry Research
By Product & Service:
Products
Refrigerators and Freezers
Cryogenic Storage
Services
By Application:
Cell & Gene Therapy
Cell Banking
Biologics
Small Molecules
Others
By End User:
Biopharmaceutical Companies
Contract Manufacturing Organizations
Contract Research Organizations
Research & Academic Institutes
By Region:
North America
Latin America
Europe
Asia Pacific
Middle East & Africa
The surging demand for pharmaceutical products globally is reshaping the landscape of pharmaceutical manufacturing. In this dynamic environment, GMP storage facilities are emerging as essential components in ensuring the quality, safety, and compliance of pharmaceutical products. As the industry continues to expand, the demand for advanced and compliant GMP storage solutions is expected to grow, making it a pivotal segment in the pharmaceutical supply chain and contributing to the overall advancement of global healthcare.
Check Out More Related Reports:
Thermal Ablation Device Market
https://www.factmr.com/report/thermal-ablation-device-market
Wireless Fetal Monitoring System Market
https://www.factmr.com/report/wireless-fetal-monitoring-system-market
Chromatography System Market
https://www.factmr.com/report/chromatography-system-market
Contact:
US Sales Office
11140 Rockville Pike
Suite 400
Rockville, MD 20852
United States
Tel: +1 (628) 251-1583, +353-1-4434-232
Email: sales@factmr.com
About Fact.MR
We are a trusted research partner of 80% of fortune 1000 companies across the globe. We are consistently growing in the field of market research with more than 1000 reports published every year. The dedicated team of 400-plus analysts and consultants is committed to achieving the utmost level of our client's satisfaction.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release GMP Storage Market Predicted to Reach US$ 11,019.9 Million at a CAGR of 5.3% by 2034 here
News-ID: 3373382 • Views: …
More Releases from Fact.MR
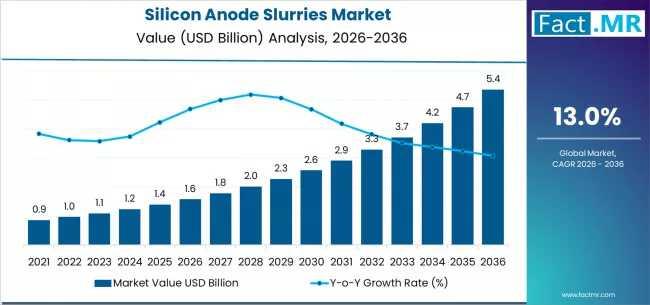
Silicon Anode Slurries Market Forecast 2026-2036: Market Size, Share, Competitiv …
The global silicon anode slurries market is set for significant expansion between 2026 and 2036, fueled by the rising adoption of high-energy-density lithium-ion batteries across electric vehicles (EVs), consumer electronics, and grid-scale energy storage. As battery manufacturers increasingly transition from graphite to silicon-enhanced anodes, the demand for high-performance, scalable silicon anode slurries is projected to grow sharply.
To access the complete data tables and in-depth insights, request a Discount On The…

Silicon Anode Slurries Market Forecast 2026-2036: Market Size, Share, Competitiv …
The global silicon anode slurries market is set for significant expansion between 2026 and 2036, fueled by the rising adoption of high-energy-density lithium-ion batteries across electric vehicles (EVs), consumer electronics, and grid-scale energy storage. As battery manufacturers increasingly transition from graphite to silicon-enhanced anodes, the demand for high-performance, scalable silicon anode slurries is projected to grow sharply.
To access the complete data tables and in-depth insights, request a Discount On The…
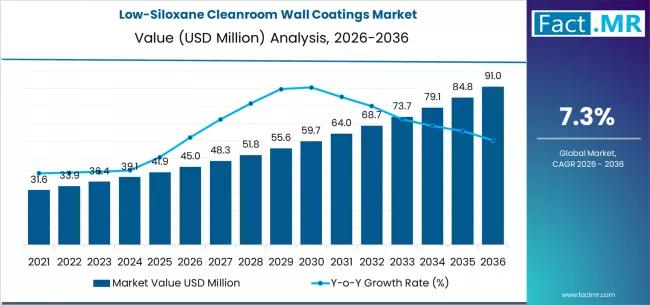
Low-Siloxane Cleanroom Wall Coatings Market Deep-Dive 2026-2036: Strategic Forec …
The low-siloxane cleanroom wall coatings market is poised for steady growth over the next decade, driven by rising contamination-control requirements across semiconductor, pharmaceutical, biotechnology, and precision manufacturing industries. These coatings are specifically engineered to minimize siloxane outgassing and volatile organic compound emissions, helping maintain ultra-clean environments where even trace contamination can disrupt production quality.
By 2036, the market for low-siloxane cleanroom wall coatings is expected to grow to USD 91.04 million.…

Low-Siloxane Cleanroom Wall Coatings Market Deep-Dive 2026-2036: Strategic Forec …
The low-siloxane cleanroom wall coatings market is poised for steady growth over the next decade, driven by rising contamination-control requirements across semiconductor, pharmaceutical, biotechnology, and precision manufacturing industries. These coatings are specifically engineered to minimize siloxane outgassing and volatile organic compound emissions, helping maintain ultra-clean environments where even trace contamination can disrupt production quality.
By 2036, the market for low-siloxane cleanroom wall coatings is expected to grow to USD 91.04 million.…
More Releases for GMP
Creative Peptides Released GMP Synthesis Service
Located in Shirley, New York, the world’s leading peptide supplier Creative Peptides announced the launch of its GMP synthesis (https://www.creative-peptides.com/services/custom-gmp-peptide-synthesis-services.html ) business on August 29, 2018. Now this company is focused on the development and GMP manufacturing of pharmaceutical grade peptides.
As the demand of pharmaceutical market continues to grow, more and more pharmas and research institutions choose the CMO and CRO models to expand their businesses, which is more…
Diapharm implements European GMP guidelines in China
Münster (DE), London (UK), Ningbo (CN), 20 December 2013 – Pharmaceutical service provider Diapharm (diapharm.com) is increasing its business activities in China: Diapharm has now implemented a “European” quality management system for Neptune Pharma Ltd (www.neptunepharma.com) in their Joint Venture Partner’s factory in Ningbo, Zhejiang Province. And it has done so successfully: The veterinary medicinal product Trident 500mg/g Powder for Suspension for Fish Treatment (www.trident-50.com), is manufactured onsite under EU…
ECA Foundation releases free GMP WebApp
The ECA Foundation has been providing advanced training and information services in the pharmaceutical industry and especially with regard to pharmaceutical Quality Assurance and GMP compliance for more than 10 years. Now the organisation took advantage of its extensive experience to develop a further free of charge service – the new GMP WebApp.
This new GMP WebApp runs on all smartphones and tablet PCs (Apple and Android platforms) and allows users…
GMP Friction Products Awarded ISO 9001:2008
Internationally Recognized Certification Measures Consistency in Process, Procedure and Quality Performance in Manufacture of Friction Materials
AKRON, OH (March 23, 2011) -- GMP Friction Products, a world leader manufacturing powdered metal friction products for clutch plates and brake pads, recently received certification for ISO 9001:2008.
“ISO 9001:2008 signifies we have taken the extra measure of documenting the policies and standards to ensure consistent compliance with our manufacturing processes,” said Jerry Lynch,…
GMP MANUAL Volume 2 - Validation Procedures by Maas & Peither AG – GMP Publish …
GMP Publishing is launching its new GMP MANUAL Volume 2 – Validation Procedures.
The compendium on validation procedures was written by Dr. Doris Borchert, Dr. Peter Bosshard, Dr. Ralph Gomez, Dr. Michael Hiob, Dr. Christine Oechslein, Max Lazar, Ulrike Reuter, Michael Schulte, Uwe Schwarzat – all international experts and key opinion leaders. They share their detailed understanding of the various aspects of the validation process in clear and comprehensive style…
blue inspection body celebrates 50 GMP audits
Münster (Germany), 20 November 2009. Two years after founding the company and just 18 months after gaining the accreditation blue inspection body GmbH announced today the successful execution of its 50th GMP audit. Further audit trips to China, India, Israel and various European countries have been scheduled already, meaning that in the first quarter 2010 the 75th audit is targeted to be completed. Blue, as a privately organised inspection body,…
