Press release
Influenza Diagnostics Market is Projected to Accelerate at A US$ 4.5 Billion by 2032
The global influenza diagnostics market was valued at around US$ 2.62 Billion in 2021. Sales of influenza diagnostics tests are projected to accelerate at a CAGR of 5% which will reach US$ 4.5 Billion by 2032. A combination of poor socioeconomic conditions and poor hygiene is responsible for the highest prevalence of influenza in developing nations. Hospitals and diagnostic labs are always on the lookout for products that can help treat influenza. A and B strains of the flu are more likely to cause severe health problems, so testing will be more popular in the coming years.This portion of the offer has a lot of doors open, including the manufacture of the items, diverting them to retail, and managing the displays. Fact. The MR examiners have made great use of both required and voluntary research. to make various assessments and projections of demand for the Influenza Diagnostics market at both global and local levels.
𝗗𝗼𝘄𝗻𝗹𝗼𝗮𝗱 𝗮 𝘀𝗮𝗺𝗽𝗹𝗲 𝗰𝗼𝗽𝘆 𝗼𝗳 𝘁𝗵𝗶𝘀 𝗿𝗲𝗽𝗼𝗿𝘁:-https://www.factmr.com/connectus/sample?flag=S&rep_id=6682?PJ
𝗖𝘂𝗿𝗿𝗲𝗻𝘁 𝗠𝗮𝗿𝗸𝗲𝘁 𝗧𝗿𝗲𝗻𝗱𝘀:
Seasonal Flu Outbreaks: The annual occurrence of seasonal flu outbreaks continues to drive the demand for influenza diagnostics. These diagnostics are crucial for early detection and treatment to reduce the spread of the virus and mitigate its impact on public health.
Emergence of Novel Influenza Strains: The constant evolution of influenza viruses and the emergence of new strains, including pandemic threats, highlight the importance of robust diagnostics to identify and monitor these strains. The COVID-19 pandemic has underscored the need for preparedness in the face of viral outbreaks.
Point-of-Care (POC) Testing: Point-of-care testing for influenza is on the rise, enabling rapid and accurate diagnosis at the patient's bedside or in healthcare settings. POC testing allows for quicker treatment decisions, reducing the risk of disease transmission.
Technological Advancements: Ongoing technological advancements have led to the development of more sensitive and specific diagnostic tests. Molecular techniques, such as polymerase chain reaction (PCR) and nucleic acid amplification tests (NAATs), are increasingly used to detect influenza viruses.
Vaccination Programs and Public Health Initiatives: Government-led vaccination programs and public health initiatives aimed at controlling influenza outbreaks are further driving the need for efficient diagnostic tools to monitor the effectiveness of vaccination campaigns and understand disease prevalence.
𝗞𝗲𝘆 𝗙𝗮𝗰𝘁𝗼𝗿𝘀 𝗜𝗻𝗳𝗹𝘂𝗲𝗻𝗰𝗶𝗻𝗴 𝗠𝗮𝗿𝗸𝗲𝘁 𝗚𝗿𝗼𝘄𝘁𝗵
Several factors are driving the growth of the influenza diagnostics market:
Outbreak Preparedness: The recurrent nature of influenza outbreaks, along with the threat of new strains emerging, has highlighted the need for robust diagnostic capabilities. Governments and healthcare organizations are investing in diagnostic infrastructure to better respond to outbreaks and pandemics.
Increased Testing Awareness: The COVID-19 pandemic has raised awareness about the importance of diagnostic testing, leading to a broader acceptance of testing as a critical tool in disease management. This change in perception is likely to have a lasting impact on the demand for influenza diagnostics.
Technological Advancements: Ongoing advancements in diagnostic technologies have improved the accuracy and speed of influenza testing, making it more convenient and reliable for both healthcare providers and patients.
Global Aging Population: With an aging global population, the susceptibility to influenza-related complications increases. This demographic trend is likely to contribute to the sustained demand for influenza diagnostics and vaccination campaigns.
Government Initiatives: Governments around the world are investing in public health programs and initiatives aimed at preventing and controlling infectious diseases like influenza. These efforts often include funding for influenza surveillance and diagnostic infrastructure.
𝗞𝗲𝘆 𝗦𝗲𝗴𝗺𝗲𝗻𝘁𝘀 𝗖𝗼𝘃𝗲𝗿𝗲𝗱 𝗶𝗻 𝘁𝗵𝗲 𝗜𝗻𝗳𝗹𝘂𝗲𝗻𝘇𝗮 𝗗𝗶𝗮𝗴𝗻𝗼𝘀𝘁𝗶𝗰𝘀 𝗜𝗻𝗱𝘂𝘀𝘁𝗿𝘆 𝗦𝘁𝘂𝗱𝘆
𝗯𝘆 𝘁𝗲𝘀𝘁
Conventional influenza diagnostic test
Rapid Influenza Diagnostic Test (RIDT)
Direct Fluorescent Antibody (DFA) Testing
virus culture
Serological assay
Molecular influenza diagnostic test
Reverse transcription polymerase chain reaction (RT-PCR)
Isothermal Nucleic Acid Amplification Assay (INAAT)
Loop-mediated isothermal-based amplification assay
Transcription-mediated isothermal-based amplification assay
Transcription-mediated isothermal-based amplification assay
𝗕𝘆 𝗲𝗻𝗱 𝘂𝘀𝗲𝗿
hospital and clinical laboratories
Diagnostic Criteria Institute
Academic/research institution
𝗠𝗮𝗿𝗸𝗲𝘁 𝗣𝗮𝗿𝘁𝗶𝗰𝗶𝗽𝗮𝗻𝘁:-
thermo fisher scientific
Hologic
Kidel Corporation
F. Hoffmann-La Roche AG
Abbott Laboratories
Becton
Dickinson and Company
Danaher Corporation
Meridian Bioscience
bioMérieux SA
Luminex Co., Ltd.
Siemens Healthineers AG
genmark diagnosis
𝗥𝗲𝗴𝗶𝗼𝗻𝗮𝗹 𝗮𝗻𝗮𝗹𝘆𝘀𝗶𝘀 𝗶𝗻𝗰𝗹𝘂𝗱𝗲𝘀
North America (USA, Canada)
Latin America (Brazil, Mexico, Argentina, Chile, Peru, Rest of LATAM)
EU - (Germany, France, Italy, Spain), UK, BENELUX (Belgium, Netherlands, Luxembourg), NORDIC (Norway, Denmark, Iceland, Sweden), Eastern Europe (Poland, Ukraine, Czech Republic, etc.), Rest of Europe
CIS & Russia
Japan
Asia Pacific Excluding Japan (Greater China, India, Korea, ASEAN Countries, Rest of APEJ)
𝗜𝗻𝗱𝘂𝘀𝘁𝗿𝘆 𝗡𝗲𝘄𝘀 𝗮𝗻𝗱 𝗗𝗲𝘃𝗲𝗹𝗼𝗽𝗺𝗲𝗻𝘁𝘀:
Multiplex Testing Platforms: Multiplex testing platforms that can simultaneously detect multiple respiratory viruses, including influenza, are gaining prominence. These platforms enhance diagnostic efficiency and aid in distinguishing between different viral infections.
Antigen-Detection Tests: Rapid antigen-detection tests have seen improvements in sensitivity and specificity, making them valuable tools for quick and reliable influenza diagnosis. These tests are particularly useful in point-of-care settings.
Global Influenza Surveillance: The World Health Organization (WHO) and other global health agencies continue to invest in influenza surveillance systems to monitor the spread of the virus, identify potential pandemics, and inform vaccine development efforts.
AI and Machine Learning: Artificial intelligence (AI) and machine learning are being applied to influenza diagnostics to enhance data analysis and prediction capabilities. These technologies are expected to play a significant role in early outbreak detection and response.
Influenza Diagnostics in Telehealth: The integration of influenza diagnostics into telehealth services is becoming more common, allowing individuals to access testing and consultations remotely. This trend gained traction during the COVID-19 pandemic and is expected to continue.
𝗜𝗺𝗽𝗿𝗼𝘃𝗲𝗱 𝗗𝗶𝗮𝗴𝗻𝗼𝘀𝘁𝗶𝗰 𝗥𝗮𝗽𝗶𝗱 𝗧𝗲𝘀𝘁𝘀 𝗳𝗼𝗿 𝗜𝗻𝗳𝗹𝘂𝗲𝗻𝘇𝗮 𝗜𝗻𝗳𝗲𝗰𝘁𝗶𝗼𝗻 𝘄𝗶𝗹𝗹 𝘀𝘁𝗶𝗺𝘂𝗹𝗮𝘁𝗲 𝗦𝗮𝗹𝗲𝘀
Recent developments have made influenza diagnostic tests more useful, allowing for greater use of these tests in the healthcare industry. A variety of tests are available as part of influenza diagnostics, such as virus culture tests, RT-PCR, serology tests, rapid antigen tests, and so on.
With rapid advances in diagnostic technology, influenza diagnostic tests were previously used only in large medical facilities and diagnostic centers, but their availability in smaller clinics and research centers has made them a potentially transformative diagnostic tool for influenza.
𝗚𝗲𝘁 𝗖𝘂𝘀𝘁𝗼𝗺𝗶𝘇𝗮𝘁𝗶𝗼𝗻 𝗼𝗻 𝘁𝗵𝗶𝘀 𝗥𝗲𝗽𝗼𝗿𝘁 𝗳𝗼𝗿 𝗦𝗽𝗲𝗰𝗶𝗳𝗶𝗰 𝗥𝗲𝘀𝗲𝗮𝗿𝗰𝗵 𝗦𝗼𝗹𝘂𝘁𝗶𝗼𝗻𝘀:
https://www.factmr.com/connectus/sample?flag=RC&rep_id=6682
US Sales Office:
11140 Rockville Pike
Suite 400
Rockville, MD 20852
United States
Tel: +1 (628) 251-1583
E Mail : sales@factmr.com
Fact.MR is a market research and consulting agency with deep expertise in emerging market intelligence. Spanning a wide range - from automotive & industry 4.0 to healthcare, technology, chemical and materials, to even the most niche categories.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Influenza Diagnostics Market is Projected to Accelerate at A US$ 4.5 Billion by 2032 here
News-ID: 3286322 • Views: …
More Releases from Fact.MR
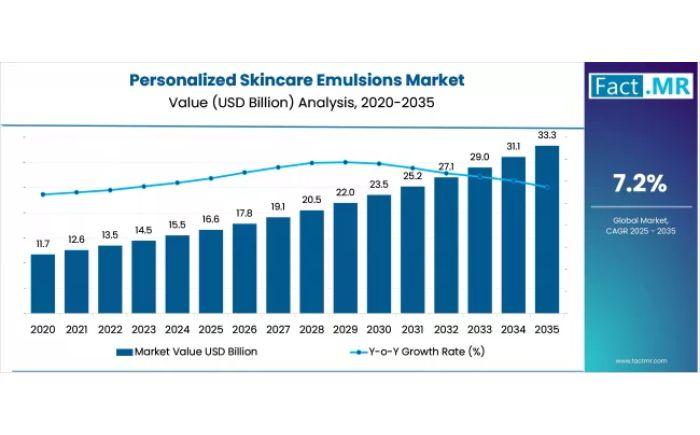
Personalized Skincare Emulsions Market Forecast 2026-2036: Size Analysis, Compet …
The global personalized skincare emulsions market is projected to grow from USD 16.6 billion in 2025 to USD 33.3 billion by 2035, expanding at a steady 7.2% CAGR. This growth is primarily driven by the "hyper-personalization" trend, where brands move away from mass-market products to focus on formulations tailored to individual DNA, microbiomes, and specific lifestyles.
Quick Stats:
Market size 2025? USD 16.6 billion.
Market size 2035? USD 33.3 billion.
CAGR? 7.2% (2025-2035).
Leading emulsion…
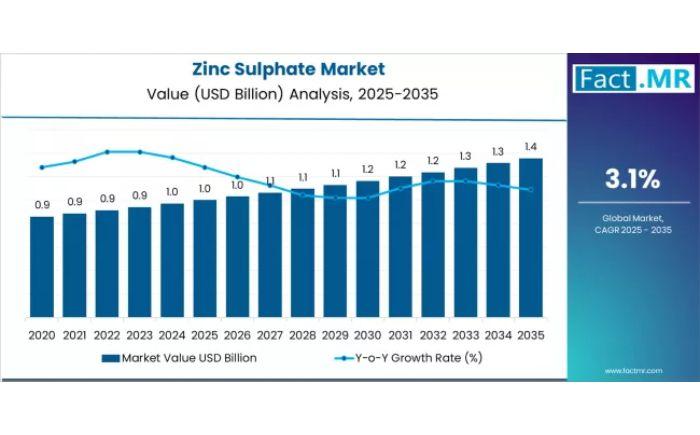
Zinc Sulphate Market Outlook to 2036: Innovation Trends, Growth Drivers & Strate …
The global zinc sulphate market is entering a decade of sustained growth, projected to expand from USD 1.0 billion in 2025 to USD 1.4 billion by 2035. This trajectory represents a steady 3.1% CAGR, fueled by the accelerating adoption of advanced micronutrient solutions in animal nutrition, specialized fertilizers, and pharmaceutical manufacturing.
Request for Sample Report | Customize Report | Purchase Full Report - https://www.factmr.com/connectus/sample?flag=S&rep_id=3663
Quick Stats:
Market size 2025? USD 1.0 billion.
Market size…
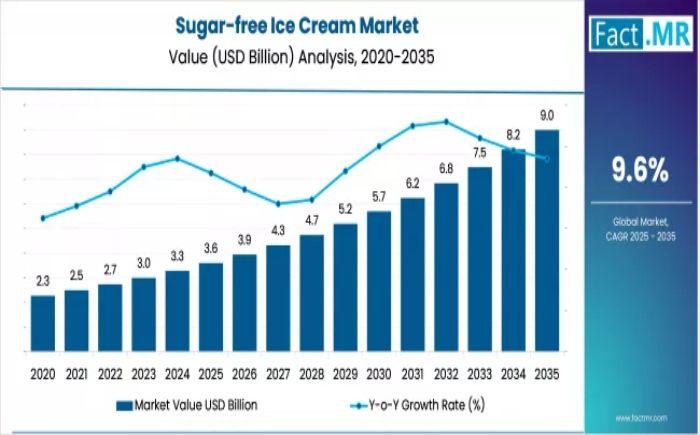
Sugar-free Ice Cream Market Strategic Report 2026-2036: Actionable Insights for …
The global sugar-free ice cream market is undergoing a rapid transformation, with its valuation expected to surge from USD 3.6 billion in 2025 to USD 9.0 billion by 2035. This represents a robust CAGR of 9.6%, making it one of the fastest-growing sub-sectors within the frozen dessert industry. The growth is fueled by a dual-engine of medical necessity-driven by rising global diabetes rates-and a lifestyle shift toward "clean label" and…
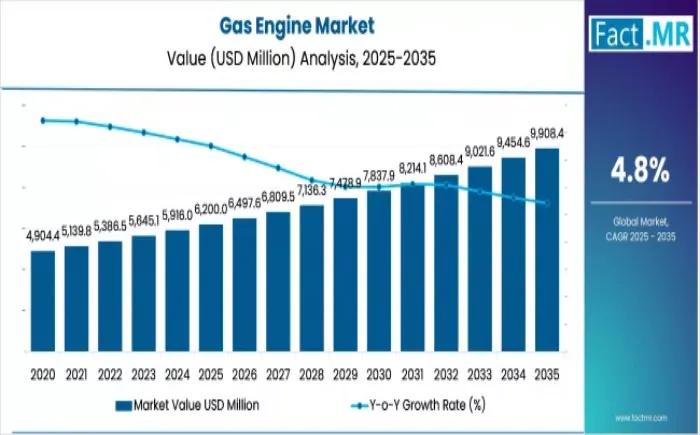
Gas Engine Market Study to 2036: Technology Evolution, Regional Demand & Forecas …
The global gas engine market is entering a period of significant long-term growth, projected to rise from USD 6.2 billion in 2025 to approximately USD 9.9 billion by 2035. This steady trajectory represents a 4.8% CAGR, driven by a global transition toward high-efficiency natural gas engines and the increasing integration of biogas and renewable gases into the energy mix.
Quick Stats:
Market size 2026? Following the 2025 valuation of USD 6.2 billion,…
More Releases for Test
Key Differences Between Megger Test, Tan Delta Test, and Hi-Pot Test for Electri …
Electrical insulation plays a critical role in ensuring the safety and efficiency of electrical systems. To assess the condition of insulation and identify potential issues, three common tests are used: the Megger test, Tan Delta test, and Hi-Pot test. Each test serves a unique purpose and provides valuable insights into the state of electrical insulation. Here's a closer look at the differences between these three essential tests.
Megger Test: Insulation Resistance…
Vitamin Test Market: Global Vitamin Test Analysis and Forecast (2023-2029)Vitami …
12.04.2024: Vitamin Test Market Overview
The development of companion diagnostic tools and advances in personalised treatment are driving considerable growth and revolution in the oncology Vitamin Test market. In the era of precision medicine, where healthcare is increasingly customised for individual individuals based on their own genetic and molecular profiles, this market segment is essential. Ongoing innovation and development define the oncology Vitamin Test market. To find particular biomarkers, genetic mutations,…
CAGR 8.1% Homecare Pregnancy Test Kits Market By Type of Test (Urine Test For H …
The Homecare Pregnancy Test Kits market report by Reports and Data provides an extensive overview of the vital elements of the Homecare Pregnancy Test Kits market and factors such as the drivers, restraints, latest trends, supervisory scenario, competitive landscape, technological advancements, and others. Further, it mentions the market shares associated with the market in terms of both value and volume along with the segmentation. Space-age industrial and digitalization tools are…
Home Safety Test Kits Market, Home Safety Test Kits Market Trends, Home Safety T …
“Home Safety Test Kits Market” 2020-2025 Research Report is a professional and in-depth study on the current state of the market. Global Home Safety Test Kits market containing a complete view of the market size, business share, profit estimates, SWOT analysis and the regional landscape of the Industry. The report explains key challenges and future development prospects of the market. The Global Home Safety Test Kits analysis is provided for…
Test Data Management (TDM) Market - test data profiling, test data planning, tes …
The report categorizes the global Test Data Management (TDM) market by top players/brands, region, type, end user, market status, competition landscape, market share, growth rate, future trends, market drivers, opportunities and challenges, sales channels and distributors.
This report studies the global market size of Test Data Management (TDM) in key regions like North America, Europe, Asia Pacific, Central & South America and Middle East & Africa, focuses on the consumption…
Hearing Screening and Diagnostic Devices Market Demands with Major Tests: pure T …
New Market Research Reports Title "Hearing Screening And Diagnostic Devices Market 2018" Has Been Added to Crystal Market Research Report database.
Hearing Screening and Diagnostic Devices - Competitive Insights:
The leading players in the market are Gn Otometrics A/S, Otodynamics, Nashua Hearing Group, Siemens Healthineers, Natus Medical Incorporated, Interacoustics A/S, Neurosoft S.A, Accent Hearing Pty Ltd, MAICO Diagnostics GmbH and IntriCon Corporation. The major players in the market are profiled in detail…