Press release
Optimization of Three Elements of ADC Drugs
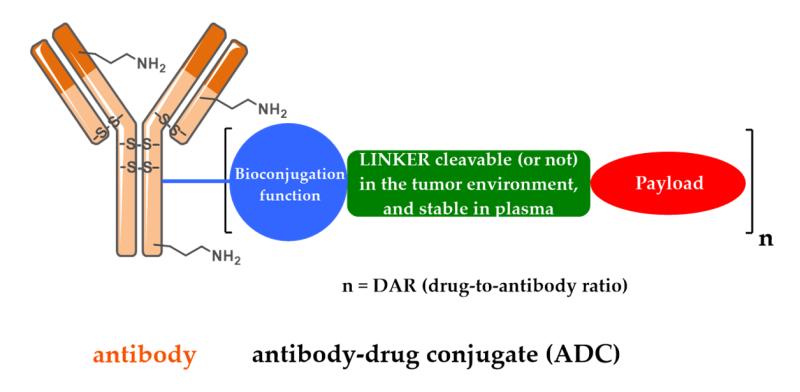
Structurally, an ADC consists of monoclonal antibodies (Antibody) that target specific antigens on tumor surfaces, cytotoxic drugs (drugs) that kill tumor cells, and linkers (Conjugate/ linkers) that conjugate cytotoxic drugs to antibodies.The link between the antibody and linker is controlled by the coupling method, which determines the DAR value of the drug and affects the stability and PK characteristics of the ADC. The connection of Linker to the drug is controlled by a shearable/non-shearable technique that determines the properties of the active payload, the rate at which the payload is released, and affects the solubility, stability, and titer of the ADC.
Optimization of Antibody: Development of new targets and dual antibody ADCs
The selection of antibodies is the starting point for the design of an ADC, and the antigenic target is the vehicle through which the ADC recognizes tumor cells. Considering that targets play a key role in delivering drugs to cancer cells, it is important to develop new targets or ADCs to expand new indications and gain a larger market space. Currently, a number of innovative ADC drug targets have entered the clinical stage worldwide.
In the process of research and development of innovative targets, it should be noted that the antigen targets suitable for ADC drugs should have the following characteristics:
It is highly expressed in tumor cells and low or no expression in normal tissues.
It is expressed on the cell surface and can access antibodies in the circulatory system.
It should internalize the antigen to allow the ADC drug to enter the cell after binding.
In addition, unlike traditional monoclonal ADCs, newly developed bi-specific ADCs can simultaneously target two different antigens, potentially increasing the rate of antibody internalization or improving tumor specificity (Figure 5). For example, the novel bi-specific ADC ZanidatamabZovodotin (ZW49) uses the internalization of Zanidatamab-enhanced antibody HER2 to deliver a novel cytotoxic drug to tumor cells via the slicable linker, inducing tumor cell death. Studies have shown that its efficacy and safety are good.
Optimization of Drug: continuous optimization of payload drugs
The cytotoxic payload carried by an ADC is the most important effector component, also known as the payload. It binds to key targets such as microtubules or genomic DNA to inhibit tumor cell proliferation. In order to develop more effective payloads, a number of relevant studies are under way, which can be optimized from three aspects: physical and chemical properties, load quantity and new loads.
Optimizing the physical and chemical properties of the current payload, especially the polarity, can overcome some of the problems faced in ADC preparation and application. Studies have shown that attaching too many hydrophobic payload molecules changes the conformational stability of the antibody, which increases its tendency to aggregate and precipitate, and ultimately affects the maximum DAR. At the same time, hydrophobic payloads can easily penetrate cell membranes and kill surrounding antigen-negative tumor cells through bystander effects. By contrast, hydrophilic payloads spread cancer cells more slowly and reduced bystander influence. Therefore, the hydrophilic polyethylene glycol (PEG) is used to balance the hydrophobic load and improve the overall stability of the ADC.
Most cancer treatment regimens require the use of multiple drugs, and clinical ADCs contain only a single drug payload. Therefore, it is imperative to develop ADCs containing multiple payloads. A study of a dual-payload ADC targeting HER2, consisting of a DNA crosslinking agent PNU-159682 and a tubulin polymerization inhibitor monomethyllaurin F (MMAF), showed that PNU-159682 induces S-phase cell cycle arrest through DNA damaging activity. By inhibiting tubulin polymerization, MMAF also causes G2/ M phase cell cycle arrest, indicating that the dual-payload ADC has a dual killing mechanism of tumor cells. In addition, dual payloads with two unrelated mechanisms may achieve mechanism synergies and minimize cancer cell resistance.
The development of new payloads has always been a hot topic in ADC design and cancer chemotherapy research. Structural modifications to approved payloads or well-studied toxins are currently considered to allow faster identification of new payloads. In addition, some new compounds may be able to combine two targets as dual-targeted warheads.
Optimization of Conjugate: Active linker can create more drug configurations
Linkers are divided into cleavable linkers and non-cleavable linkers. Research focuses on the preparation of homogeneous ADCs using connector technology, developing connector technologies that can easily connect antibodies to their payloads, and finding new ways to improve DAR while maintaining homogeneity. At present, some new connector technologies are under test, including secondary bridging ADC, DNA bridging ADC, self-assembly ADC, high DAR ADC, and optical shear ADC.
In summary, many preclinical studies have innovated to improve therapeutic efficacy or overcome the shortcomings of existing ADCs, thus inspiring the field of antibody drug conjugates and expanding the forms of development.
These novel antibody drug conjugates include, but are not limited to, small molecule conjugates (SMDCS), antibody drug conjugates, peptide-drug conjugates (PDC), amphiphilic peptide-drug conjugates, amphiphilic inhibitor-drug conjugates, antibody-polymer-drug conjugates, antibody-photosensitizer conjugates (APCs), and ligand drug conjugates (LDCs).
As a reliable PEG supplier, Biopharma PEG provides GMP standard PEG derivatives and bulk orders via custom synthesis, offering the opportunity to match customers' special quality requirements. ADC linkers with molecular weights, branching, and functional groups not listed in our online catalog may be available by custom synthesis.
Biopharma PEG Scientific Inc.
Address: 108 Water Street, Room 4D, Watertown, MA 02472, USA
TEL: 1-857-366-6766
Fax: 617-206-9595
Email: sales@biochempeg.com
Website: https://www.biochempeg.com/
Biopharma PEG Scientific Inc. is a biotechnology-oriented company in Watertown, Massachusetts. We are dedicated to manufacturing and supplying high purity monodispersed and polydispersed polyethylene glycol (PEG) derivatives and PEG raw material, PEGylation services, and custom PEG derivative synthesis to clients worldwide. We continuously expand the capability to provide large-scale manufacture of high purity PEG derivatives with an extensive variety of functional groups, in both non-GMP and GMP grade. These PEG linkers have been widely used in bioconjugation, antibody-drug conjugates (ADCs) therapeutic, click chemistry, 3d bioprinting, drug delivery and diagnostics field, etc.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Optimization of Three Elements of ADC Drugs here
News-ID: 3063657 • Views: …
More Releases from Biopharma PEG Scientific Inc

Biopharma PEG Offers In-Stock Monodispersed PEGs at Competitive Prices
Biopharma PEG Scientific Inc. announced the availability of a broad range of in-stock monodispersed PEG derivatives at highly competitive prices, supporting researchers working in antibody-drug conjugates (ADCs), nanoparticle and LNP delivery systems, diagnostics, and biomaterials.
Biopharma PEG specializes in PEG derivatives, including monodispersed, polydispersed, and multi-arm PEGs, offered in both GMP and non-GMP grades. By keeping key monodispersed PEGs on the shelf and ready to ship, the company aims to help…
Biopharma PEG Highlights the Critical Role of PEG-Lipid Nanoparticles in RNA The …
Watertown, MA, September 26, 2025 - In the past decade, lipid nanoparticles (LNPs) have emerged as the premier delivery vehicle for RNA-based medicines, powering the success of both siRNA drugs and mRNA vaccines. A key component of these systems is PEG-lipids, specialized polyethylene glycol (PEG) derivatives that stabilize nanoparticles, extend circulation time, and enhance safety.
Why PEG-Lipids Are Essential
RNA molecules are inherently unstable in the bloodstream, where they are rapidly degraded…
BIOPHARMA PEG Receives FDA DMF Listings for Two Key PEG Derivatives
BIOPHARMA PEG, a leading developer and supplier of high-purity polyethylene glycol (PEG) derivatives, today announced that two of its proprietary products-mPEG-pALD 20K and HZ-PEG-HZ (1K)-have successfully passed FDA review and are now listed in the U.S. Drug Master File (DMF) database.
mPEG-pALD (20K) - DMF No. 040600
HZ-PEG-HZ (1K) - DMF No. 041864
These new listings mark a significant step in BIOPHARMA PEG's ongoing efforts to provide regulatory-ready, high-quality PEG solutions to the…
Monodispersed PEG Linkers Enhance Antibody-Drug Conjugates (ADCs)
Antibody-drug conjugates (ADCs) combine potent small-molecule drugs with monoclonal antibodies for targeted cancer therapy. A major challenge is loading enough drug onto each antibody without compromising solubility or circulation time. Introducing polyethylene glycol (PEG) linkers between the antibody and drug payload can address this. PEG is hydrophilic, biologically inert, and FDA‐recognized as safe. As a flexible spacer, PEG "shields" the hydrophobic drug, greatly improving ADC solubility and stability. In fact,…
More Releases for ADC
ADC Market: Transforming Cancer Treatment
The global market for antibody-drug conjugates (ADCs) was valued at US$ 11.32 billion in 2023 and is expected to reach US$ 27.37 billion by 2033, growing at a compound annual growth rate (CAGR) of 9.23% from 2024 to 2033. This growth is primarily driven by the increasing prevalence of cancer and the growing demand for safe and effective treatments.
Antibody Drug Conjugates: A Growing Force in Cancer Treatment and Market Expansion
Key…
With Knockout ADC Innovations, Creative Biolabs Took Center Stage at the 2024 Wo …
Creative Biolabs wrapped up a successful show at the 15th Annual World ADC San Diego in the beginning of November, where its third year of participation, highlighting its dedication to the advancement of antibody-drug conjugate (ADC) technology.
New York, USA - November 13, 2024 - Creative Biolabs seized the spotlight at Booth 313 by offering groundbreaking solutions across the entire ADC development spectrum, which demonstrate their unrivaled expertise and dedication to…
Showing Off Their ADC Game: Creative Biolabs Hits the 15th Annual World ADC Summ …
From November 4, Creative Biolabs will be back at the 15th Annual World ADC Summit in San Diego, marking its fourth year at the leading global gathering featuring innovative conjugates.
New York, USA - November 6, 2024 - Creative Biolabs invites all ADC enthusiasts, from new entrants to seasoned experts, to explore its comprehensive suite of ADC solutions and experience the latest in antibody-drug conjugate (ADC) innovation at Booth #313.
Image: https://www.getnews.info/uploads/0c59d26f78bf6dd58aafee5bc68d7d8c.jpg
"2023…
ADC Drugs For Breast Cancer Treatment
Breast cancer is the malignant tumor with the highest morbidity and mortality among women worldwide. At present, the main therapeutic methods include surgery, chemotherapy, radiotherapy, endocrine therapy and targeted therapy, etc. The development and marketing of new drugs have far-reaching significance in improving the survival of breast cancer patients and changing the pattern of breast cancer treatment.
On February 24, 2023, The NMPA approved Enhertu, an injectable drug developed jointly by…
Analog-to-Digital Converters Market by Product (Ramp ADC, Integrating ADC, Succe …
Analog-to-Digital Converters Market by Product (Ramp ADC, Integrating ADC, Successive Approximation ADC, Delta-sigma ADC, and Others (Pipelined/Flash ADC)), and Application (Consumer Electronics and Industrial) - Global Opportunity Analysis and Industry Forecast, 2017-2023
Full Report : https://www.alliedmarketresearch.com/analog-to-digital-converters-market
Increasing disposable income and technological advancements supplement the analog-to-digital converters market. However, complex design of the ADC hampers this market growth. On the other hand, encouragement in digitization of work processes by government in emerging economies,…
Global Data Converters Market Report 2017 (Analog to Digital Converter, Current …
The research report titled Global Data Converters Sales Market Report 2017 market size and forecast and overview on current market trends.
This report studies sales (consumption) of Global Data Converters Market 2017, especially in United States, China, Europe and Japan, focuses on top players in these regions/countries, with sales, price, revenue and market share for each player in these regions, covering
National Semiconductor
Nippon Precision Circuits Inc
Micro Analog systems
Microchip Technology.
TelCom Semiconductor, Inc
Vishay Siliconix
Texas…