Press release
Urinary Tract Infection Treatment Market Size to Expand Significantly by the End of 2027
The global urinary tract infection treatment market is projected to amass a large amount of revenues in the coming years. The healthcare industry has shown a deft level of earnestness in dealing with infectious diseases and disorders. Besides, availability of improved research facilities has helped medical scientists in diagnosing the various reasons responsible for urinary tract infection. It is safe to expect that the global urinary tract infection treatment market would become a safe haven for investment in the years to follow. Rapid advancements in the field of nephrology have also played a part in driving market demand.Read Report Overview: https://www.transparencymarketresearch.com/urinary-tract-infection-treatment-market.html
Transparency Market Research (TMR), in one of its reports, predicts that the global urinary tract infection treatment market would expand at a sluggish CAGR of 3% over the period between 2019 and 2027. Furthermore, the total market value is expected to touch US$ 7.5 Bn in 2027, growing from a value of US$ 5.9 Bn in 2018. The slow rate of growth can be attributed to the delays in approval of several drugs. However, the presence of a seamless healthcare industry shall continue to complement market growth. Moreover, new research initiatives pertaining to medicine and healthcare shall also drive market demand.
Request a PDF Brochure - https://www.transparencymarketresearch.com/sample/sample.php?flag=B&rep_id=72872
Approvals from FDA as a Forerunner to Market Growth
The Food and Drug Administration (FDA) has been the harbinger of multiple improvements in the healthcare industry. As the need for improved treatment mechanisms gathers momentum across the world, FDA is playing a defining role in modern medicine. In its latest, FDA approved the use of FETROJA® (cefiderocol) drug for the treatment of urinary tract infections. The occurrence of certain complicated forms of urinary tract infections in adults could be deadly. These types of infections do not have an alternative line of treatment, except the use of heavy drugs. FDA’s new approval is expected to aid the growth of the global urinary tract infection treatment market.
Request for Analysis of COVID19 Impact on Urinary Tract Infection Treatment Market - https://www.transparencymarketresearch.com/sample/sample.php?flag=covid19&rep_id=72872
Availability of Improved Medical Facilities to Drive Demand
The adverse effects of insufficient water consumption are widely discussed by medical practitioners and doctors. Furthermore, urinary infections are often an outcome of low water consumption. Therefore, medications for treatment of urinary tract infection are widely distributed across hospitals and pharmacies. Therefore, the global urinary tract infection treatment market is set to undergo revolutionary growth in the years to follow. The advent of better healthcare facilities within nephrology have played a vital role in driving market demand. The geriatric population is highly vulnerable to the incidence of urinary tract infection. Therefore, improvements in geriatric care have also given a thrust to market growth.
Buy now Urinary Tract Infection Treatment Market Report - https://www.transparencymarketresearch.com/checkout.php?rep_id=72872
Some of the leading vendors operating in the global urinary tract infection market are AstraZeneca plc, Pfizer, Inc., Johnson & Johnson, F. Hoffmann La-Roche Ltd., Teva Pharmaceutical Industries Ltd., Boehringer Ingelheim GmbH, Novartis AG.
More Trending Reports by Transparency Market Research:
https://www.prnewswire.com/news-releases/rise-in-the-demand-for-personalized-medicines-to-propel-the-drug-discovery-outsourcing-market-says-tmr-301326948.html
https://www.prnewswire.com/news-releases/rising-demand-for-needle-free-vaccines-to-drive-innovation-in-the-respiratory-virus-vaccines-market-tmr-301332746.html
https://www.prnewswire.com/news-releases/surgical-microscopes-market-revenue-to-reach-us-1-3-billion-by-2027--manufacturers-focus-on-performance-and-portability-of-products-for-innovation-transparency-market-research-301336425.html
Contact Us
Transparency Market Research
State Tower,
90 State Street,
Suite 700,
Albany NY - 12207
United States
USA - Canada Toll Free: 866-552-3453
Website: https://www.transparencymarketresearch.com
About Us
Transparency Market Research is a next-generation market intelligence provider, offering fact-based solutions to business leaders, consultants, and strategy professionals.
Our reports are single-point solutions for businesses to grow, evolve, and mature. Our real-time data collection methods along with ability to track more than one million high growth niche products are aligned with your aims. The detailed and proprietary statistical models used by our analysts offer insights for making right decision in the shortest span of time. For organizations that require specific but comprehensive information we offer customized solutions through adhoc reports. These requests are delivered with the perfect combination of right sense of fact-oriented problem solving methodologies and leveraging existing data repositories.
TMR believes that unison of solutions for clients-specific problems with right methodology of research is the key to help enterprises reach right decision.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Urinary Tract Infection Treatment Market Size to Expand Significantly by the End of 2027 here
News-ID: 2368144 • Views: …
More Releases from Transparency Market Research

Electric Wheelchair Market Expanding at 9.2% CAGR Through 2036 - By Control Type …
The global electric wheelchair market continues to demonstrate strong and sustained growth, fueled by demographic transitions, technological innovation, and expanding healthcare access worldwide. Valued at US$ 5.8 billion in 2025, the market is projected to reach US$ 15.3 billion by 2036, expanding at a compound annual growth rate (CAGR) of 9.2% from 2026 to 2036.
Discover essential conclusions and data from our Report in this sample -
https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=4198
This robust trajectory reflects rising…
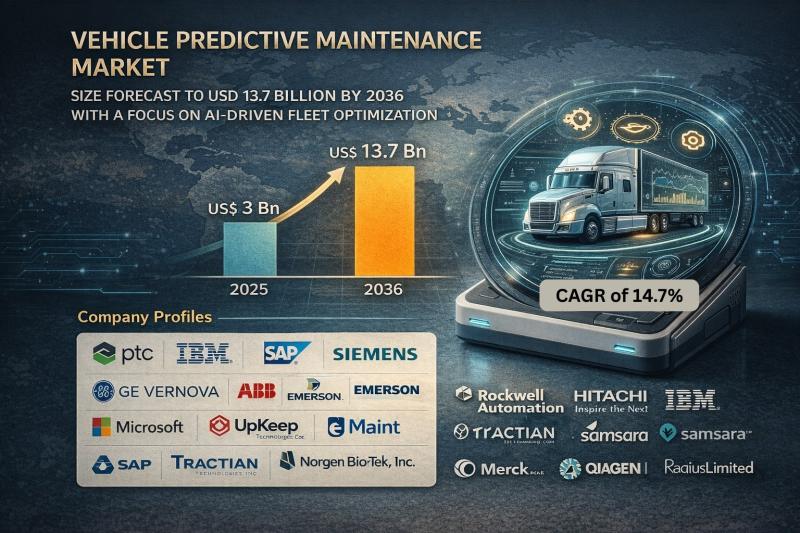
Vehicle Predictive Maintenance Market Size Forecast to USD 13.7 Billion by 2036 …
Vehicle Predictive Maintenance Market Outlook 2036
The global vehicle predictive maintenance market was valued at USD 3 Billion in 2025 and is projected to reach USD 13.7 Billion by 2036, expanding at a robust CAGR of 14.7% from 2026 to 2036. Market growth is driven by increasing adoption of connected vehicles, rising fleet digitalization, advancements in AI-driven analytics, and growing emphasis on minimizing vehicle downtime and maintenance costs.
👉 Get your sample…
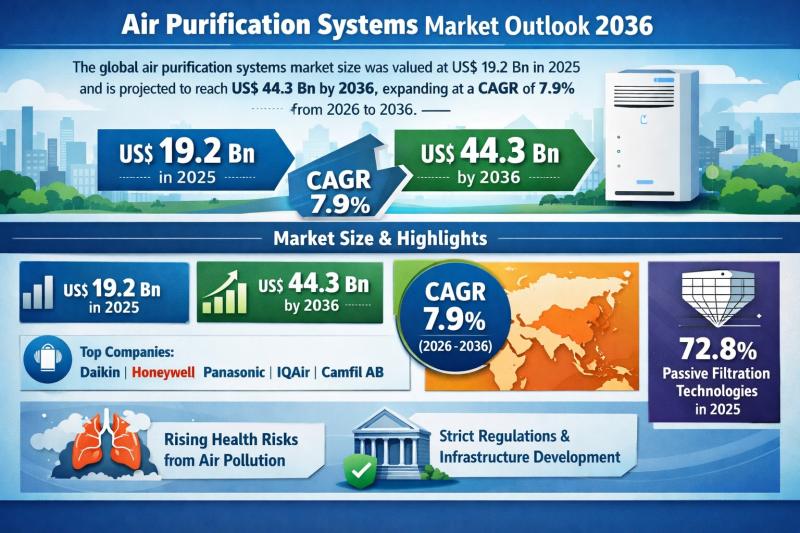
Global Air Purification Systems Market to Reach USD 44.3 Billion by 2036 at 7.9% …
The global Air Purification Systems Market was valued at US$ 19.2 Bn in 2025 and is projected to expand to US$ 44.3 Bn by 2036, registering a compound annual growth rate (CAGR) of 7.9% from 2026 to 2036. The market's upward trajectory reflects the structural shift in indoor air quality (IAQ) management, moving from discretionary consumer spending to mission-critical infrastructure investment.
With historical data available from 2021 to 2024, the industry…
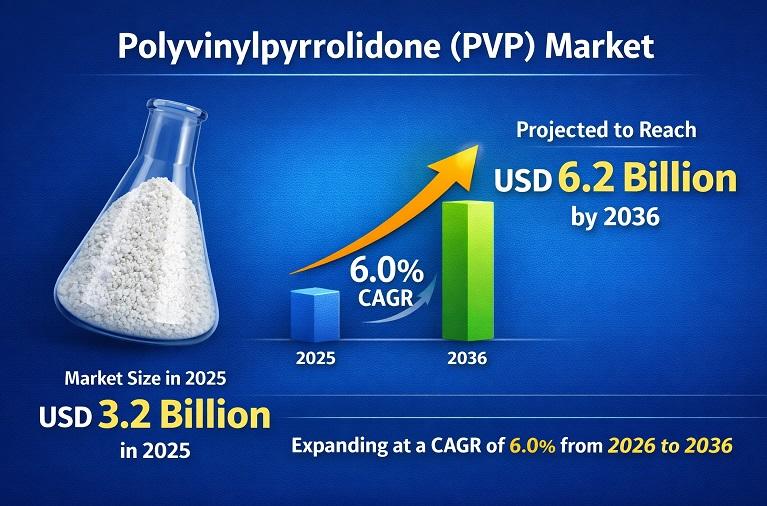
Polyvinylpyrrolidone (PVP) Market to Reach USD 6.2 Billion by 2036 Driven by Pha …
The Polyvinylpyrrolidone (PVP) Market was valued at around US$ 3.2 billion in 2025 and is projected to reach approximately US$ 6.2 billion by 2036, expanding at a steady CAGR of about 6.0% during the forecast period. This growth is primarily driven by rising demand from the pharmaceutical industry, where PVP is widely used as a tablet binder, solubilizer, and stabilizer, along with increasing consumption in cosmetics and personal care products…
More Releases for FDA
DreaMed receives 5th FDA Clearance
TEL AVIV, Israel: DreaMed Diabetes LTD. ("DreaMed" or the "Company"), developer of the endo.digital Clinical Decision Support System announced today that it has received its 5th U.S Food and Drug Administration (FDA) clearance that expands the scope of AI enhanced treatment recommendations to patients on fixed meal insulin regimens. endo.digital is the first decision support system that has been cleared to assist healthcare providers in the management of diabetes…
FDA Compliant Blood Storage and Preservation
Accsense Monitoring System Automates Data Archive and Alarming
CAS DataLoggers provided the temperature alarming and monitoring system to a hospital blood bank looking to replace their old paper chart recorders as they became unreliable and spare parts were harder to find. For proper blood storage and preservation, the lab’s medical units needed to maintain storage temperatures between 2°C to 6°C (36°F to 43°F), given the perishability of blood components. The facility…
FDA grants orphan drug status to Vicore
US Food and Drug Administration has awarded Vicore Pharmaceuticals with orphan Drug designation for the treatment of Idiopathic Pulmonary Fibrosis (IPF). FDA’s Orphan Drug Designation program provides certain incentives for companies developing therapeutics to treat rare diseases or conditions, defined as those affecting less than 200,000 individuals in the U.S. A drug candidate and its sponsor must meet several key criteria in order to qualify for, and obtain, orphan drug…
New FDA Design Control Training Courses
Salt Lake City, Utah - February 23 2017 - Procenius Consulting is a medical device consulting firm specializing solely in medical device design controls regulation (21 CFR 820.30).
Announcing New Design Control Training Courses
Procenius Consulting has just launched two new training courses covering basic and advanced topics of medical device design control regulation. These courses focus on compliance, practical implementation and industry best practices techniques for developing or improving a…
fda online training
GRC Training Solutions provides end-to-end FDA compliance solutions for those companies who want to maximize security, minimize operational costs, improve staff productivity and stay on top of all their compliance documentation.
GRC Training Solutions boasts a team of experts and specialists who have a proven track record in working with the biotechnology, medical device, diagnostic and pharmaceutical fields. Our team will work with you closely and develop solutions that meet…
FDA online training
Description:
Device firms, establishments or facilities that are involved in the production and distribution of medical devices intended for use in the U.S are required to register annually. Most establishments that are required to register with the FDA are also required to list the devices that are made there and the activities that are performed on those devices. Initially, FDA issued a 28-page Proposed Rule that would amend its regulations regarding…