Press release
Freiburg based Spindiag GmbH and Hahn-Schickard research institute receive six million euros from local government to develop a SARS-CoV-2 rapid test
The Economic Committee of Baden-Wuerttemberg's Ministry for Economic Affairs, Labour and Housing Construction approved of funding in fast track authorisationNested PCR test for results in about 35 minutes directly at the point of care
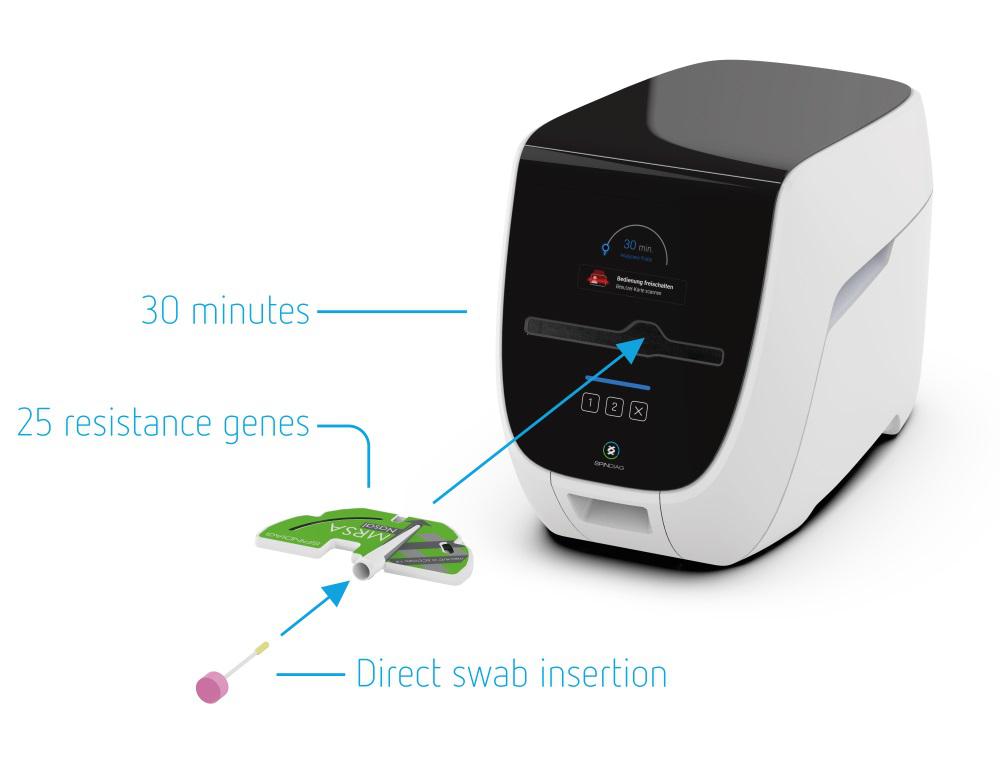
Freiburg im Breisgau, April 7, 2020 - Spindiag GmbH, based in Freiburg im Breisgau, is a medical technology start-up that arose out of the renowned Hahn-Schickard Institute for Microanalytical Systems four years ago. Together with Hahn-Schickard, the company announced today that it will receive a total of six million euros in funding from the State of Baden-Wuerttemberg for the development of a SARS-CoV-2 rapid test. This so-called nested PCR test can be used directly at the point of care, meaning at the place where samples are taken, thereby reducing time to diagnosis. The Economic Committee of the Ministry of Economics, Labour and Housing Construction approved the funding in a fast track authorisation on April 7th. It will enable the company to establish a rapid test for the SARS-CoV-2 virus on its existing test platform, which was developed for the detection of multidrug-resistant bacteria. The test will enable the reliable detection of SARS-CoV-2 in about 35 minutes. It is also planned to expand the platform for differential diagnosis of other respiratory diseases including seasonal influenza. The test system is currently undergoing analytical testing. The aim is to launch with a CE-IVD declaration of conformity by the third quarter of 2020.
Dr. Daniel Mark, CEO of Spindiag GmbH, states: "The corona pandemic is currently posing major challenges for the German health care system and health care systems worldwide. We can assume that there will continue to be a high demand for SARS-CoV-2 test systems, especially at the point of sample taking so that doctors can immediately order quarantine and initate further measures if necessary. We are very pleased that we were able to convince the state of Baden-Wuerttemberg of the potential and sustainability of our platform."
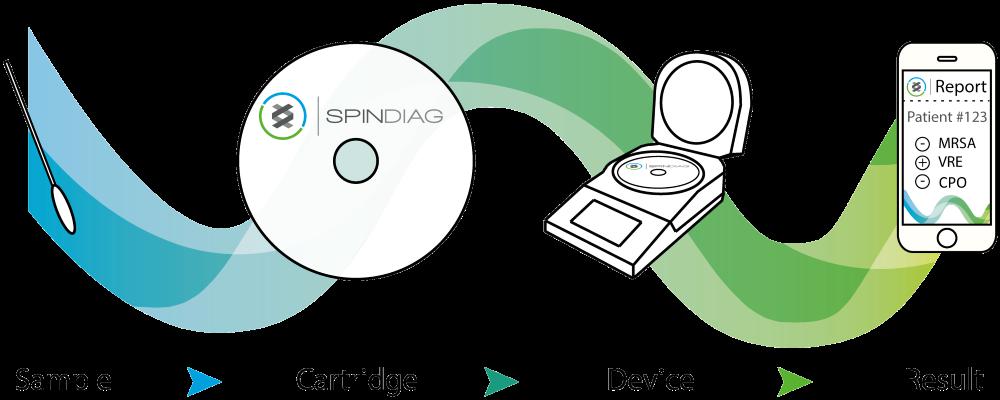
The Spindiag platform consists of an analyzer with an integrated test cartridge that can be intuitively integrated into the clinical routine. Without any preparation, swab samples from the nose or throat can be inserted directly into the test cartridge, which contains all the reagents required for the test. Similar to a CD, the cartridge is then inserted into the device and processed fully automatically. Results are available in about 35 minutes. A further advantage: two cartridges can be processed simultaneously in the device, which enables higher test capacities. Thanks to proprietary technology, manual pipetting of the sample into the test cartridge is no longer necessary. The risk of infection for users can thus be reduced to a minimum.
Test results can be read directly from the device and, and if necessary, the data can be transmitted anonymously via the mobile network or a direct cloud connection to relevant institutions.
About the nested PCR test method
The nested PCR test method is a further development of the common standard laboratory analysis called polymerase chain reaction (PCR), with which the virus can be directly detected in a sample. In the nested PCR approach, two stages of the standard PCR are connected in series in the test cartridge. This enables particularly sensitive, specific and therefore reliable analyses.
Spindiag GmbH
Dr. Daniel Mark, CEO
+49 761 203 73246
info@spindiag.de
Spindiag GmbH, based in Freiburg im Breisgau, Germany, was founded in 2016 by a team of internationally renowned experts in the fields of microfluidics, lab-on-a-chip technology and life sciences. Based on a proprietary microfluidic technology researched at the Hahn-Schickard Institute for Microanalytical Systems, Spindiag is developing a sustainable platform for the diagnosis of infectious diseases and plans to offer a rapid test for the SARS-CoV-2 at the point of care as first product. Since its foundation, the company has received a total of 8.6 million euros in three financing rounds to prepare for market entry.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Freiburg based Spindiag GmbH and Hahn-Schickard research institute receive six million euros from local government to develop a SARS-CoV-2 rapid test here
News-ID: 2005367 • Views: …
More Releases from SpinDiag GmbH

Spindiag seeks strategic investors for 15-min POC multiplex PCR system amid prel …
Spindiag GmbH invites strategic investors to join in on its innovation of a 15-minute point-of-care multiplex PCR system amid preliminary insolvency proceedings
Freiburg/Breisgau, Germany, July 10, 2023 - Spindiag GmbH, a start-up company in the field of molecular in-vitro diagnostics, has announced the opening of preliminary insolvency proceedings as a result of challenging market conditions caused by the rapid decline of COVID-19 testing. The company remains operational and is actively seeking…
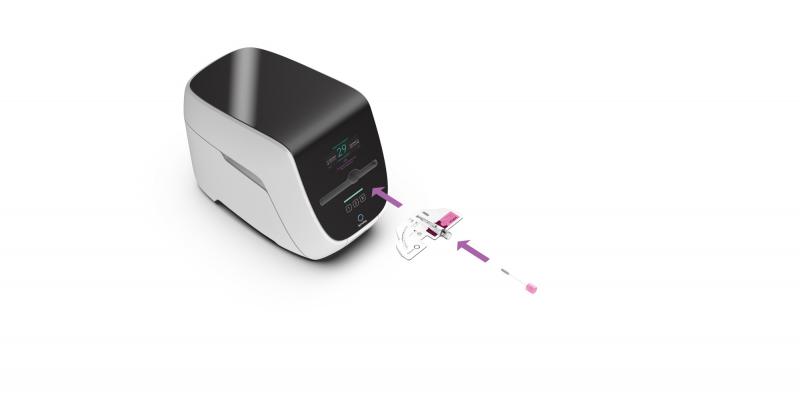
Spindiag’s continued road to success: Expansion of Series A with an additional …
- With new and current investors, the medtech startup in Freiburg takes the next big step in launching its innovative technology for the rapid detection of multidrug-resistant bacteria.
- The additional money will enable industrializing the production and securing CE-IVD approval for the EU market of an MRSA (methicillin-resistant Staphylococcus aureus) rapid test planned for launch in 2020.
- A recent study on the usability of Spindiag’s point-of-care system confirms its easy,…
SpinDiag closes second financing round of EUR 3.0 million (USD 3.4 million) to c …
Freiburg im Breisgau, Germany / November 2018 – One year after its seed financing, SpinDiag GmbH closes a second financing round of EUR 3 million (USD 3.4 million) as planned. The financing will enable SpinDiag to complete the product development of its first product for screening for antibiotic-resistant bacteria and to start clinical trials. This will pave the way for regulatory approval in the EU.
To finance the current Series A,…
SpinDiag Raises 1.6 Mio. € To Bring Point-of-Care Screening For Antibiotic Res …
The Freiburg-based startup SpinDiag GmbH recently closed a 1.6 Mio. EUR seed-round with three private investors. The team developed a revolutionary point-of-care screening system for testing patients for antibiotic-resistant bacteria at their admission to hospitals and almost instantly so. The seed-capital will make it feasible to bring SpinDiag’s system from its current laboratory environment to first tests in hospitals. “Testing in hospitals will not only be an important milestone for…
More Releases for PCR
Emerging Trends to Reshape the Global Real-Time PCR, Digital PCR And End-Point P …
Use code ONLINE20 to get 20% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
Real-Time PCR, Digital PCR And End-Point PCR Market Size Valuation Forecast: What Will the Market Be Worth by 2025?
The market encompassing real-time PCR, digital PCR, and end-point PCR has experienced robust expansion over the past few years, projecting a rise from $38.37 billion in 2024 to $41.55 billion…
Role Of Real-Time Rt-Pcr In Accelerating Covid-19 Diagnosis And Driving Pcr And …
The PCR And Realtime PCR Testing Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].
What Is the Current PCR And Realtime PCR Testing Market Size and Its Estimated Growth Rate?
The PCR and real-time PCR testing market has grown strongly, rising from $10.55 billion in 2024…
Role Of Real-Time Rt-Pcr In Accelerating Covid-19 Diagnosis And Driving Pcr And …
How Will the PCR And Realtime PCR Testing Market Grow, and What Is the Projected Market Size?
There has been a significant growth in the market size of PCR and realtime PCR testing in the last few years. Its size is expected to increase from $10.55 billion in 2024 to $11.21 billion in 2025, with a Compound Annual Growth Rate (CAGR) of 6.3%. The growth observed in the historic period is…
Role Of Real-Time Rt-Pcr In Accelerating Covid-19 Diagnosis And Driving Pcr And …
How Will the PCR And Realtime PCR Testing Market Grow, and What Is the Projected Market Size?
There has been a significant growth in the market size of PCR and realtime PCR testing in the last few years. Its size is expected to increase from $10.55 billion in 2024 to $11.21 billion in 2025, with a Compound Annual Growth Rate (CAGR) of 6.3%. The growth observed in the historic period is…
Q-PCR and D-PCR Devices Market: Revolutionizing Molecular Diagnostics
The Q-PCR and D-PCR devices market has experienced significant growth in recent years, driven by advancements in molecular biology and the increasing demand for accurate and sensitive diagnostic tools. These technologies have revolutionized the field of molecular diagnostics, enabling researchers and clinicians to detect and quantify specific DNA or RNA sequences with high precision.
Market Size and Growth
The global Q-PCR and D-PCR devices market is estimated to be worth billions of…
Real-Time PCR, Digital PCR And End-Point PCR Global Market Analysis, Trends, Gro …
The new report published by The Business Research Company, titled "Real-Time PCR, Digital PCR And End-Point PCR Global Market Report 2024 - Market Size, Trends, And Global Forecast 2024-2033", delivers an in-depth analysis of the leading size and forecasts, investment opportunities, winning strategies, market drivers and trends, competitive landscape, and evolving market trends.
As per the report, the real-time pcr, digital pcr and end-point pcr market size has grown strongly…
