Press release
The contract manufacturing services market for stem cells is expected to be worth over USD 2.5 billion by 2030, claims Roots Analysis
Roots Analysis has announced the addition of the “Stem Cell Contract Manufacturing Market, 2019-2030” report to its list of offerings.Owing to a highly regulated production environment and the need for state-of-the-art technologies and equipment, it is difficult for innovators to establish in-house expertise for the large-scale manufacturing of stem cell therapies. As a result, stem cell therapy developers are increasingly relying on contract manufacturing organizations (CMOs) for their product development and manufacturing needs.
To order this 300+ page report, which features 120+ figures and 145+ tables, please visit this link https://www.rootsanalysis.com/reports/view_document/the-contract-manufacturing-services-market-for-stem-cells-is-expected-to-be-worth-over-usd-25-billion-by-2030-claims-roots-analysis/271.html
Key Market Insights
Over 80 CMOs presently claim to provide manufacturing services for stem cells therapies
- The market landscape is fragmented, featuring a mix of companies of all sizes; however, small and mid-sized companies represent 70% of the overall number of industry stakeholders. It is worth highlighting that over 50% of the CMOs claim to be capable of accommodating both clinical and commercial scales of operation.
100+ strategic alliances have been inked since 2015
- In fact, nearly 60% of the abovementioned deals were established post 2016. Majority of these agreements were observed to be focused on the manufacturing of various types of stem cells. It is worth highlighting that the maximum number of partnerships related to stem cell therapies were reported in 2018.
More than 80,000+ patients were reported to have been enrolled in stem cell therapy related trials, since 2010
- As a result, the manufacturing demand for such therapies can be anticipated to grow significantly over the next decade. The report features detailed projections of the future clinical and commercial demand for stem cell manufacturing, based on parameters, such as target patient population, dosing frequency, dose strength, source of stem cells, type of stem cells and key geographies.
Currently, there are more than 100 facilities dedicated to stem cell manufacturing
- The maximum share of the installed capacity belongs to large (more than 1,000 employees) and very large (more than 5,000 employees) companies. The report provides a detailed capacity analysis, taking into consideration the reported manufacturing capacities of industry stakeholders, and offering estimates on the distribution of the global contract manufacturing capacity for stem cell therapies, by company size, scale of operation and geography.
By 2030, North America and Europe are anticipated to capture over 70% of the market share
- Overall, the market is anticipated to witness an annualized growth rate of more than 20% over the next decade. In the long-term, the opportunity is expected to be well distributed across key stakeholder companies, which offer services for a diverse range of allogenic and autologous stem cell therapies.
To request a sample copy / brochure of this report, please visit this link https://www.rootsanalysis.com/reports/view_document/the-contract-manufacturing-services-market-for-stem-cells-is-expected-to-be-worth-over-usd-25-billion-by-2030-claims-roots-analysis/271.html
Key Questions Answered
- Who are the leading CMOs with expertise in stem cell production?
- What is the annual clinical and commercial demand for stem cell therapies?
- What is the current installed manufacturing capacity for stem cell therapies?
- What are the key challenges faced by stem cell manufacturers?
- What kind of partnership models (manufacturing agreements, licensing, process development, product development & commercialization, acquisitions, and others) are commonly adopted by stakeholders in this industry?
- Which type of stem cells are anticipated to be responsible for generating the maximum revenues in this domain?
- What fraction of stem cell manufacturing operations are presently outsourced to CMOs?
- How is the current and future market opportunity likely to be distributed across key market segments?
The USD 2.5 billion (by 2030) financial opportunity within the stem cells contract manufacturing market has been analyzed across the following segments:
Scale of Operation
- Preclinical
- Clinical
- Commercial
Size of Contract Manufacturer
- Small-sized
- Mid-sized
- Large / Very Large
Source of Stem Cells
- Autologous
- Allogenic
Type of Stem Cells
- Adult
- Embryonic
- Induced Pluripotent
Key geographical regions
- North America (US)
- Europe (UK, France, Germany, Italy and Spain)
- Asia (Japan)
- Rest of the World
The report features inputs from eminent industry stakeholders, according to whom the contract manufacturing opportunity related to stem cell therapies can be expected to witness substantial growth due to the rising demand for regenerative medicine across a number of therapeutic areas. The report includes detailed transcripts of discussions held with the following experts:
- Brian Dattilo (Manager of Business Development, Waisman Biomanufacturing)
- David Mckenna (Professor and American Red Cross Chair in Transfusion Medicine, University of Minnesota)
- Fiona Bellot (Business Development Manager, Roslin Cell Therapies)
- Gilles Devillers (General Manager, Bio Elpida)
- Mathilde Girard (Department Leader, Cell Therapy Innovation and Development, YposKesi)
The research covers detailed profiles of key players (illustrative list provided below) from across key global markets (North America, -Europe and Asia-Pacific); each profile provides an overview of the company, information on its service portfolio, manufacturing facilities, financial performance (if available), details on recent developments, as well as an informed future outlook.
- BioNTech Innovative Manufacturing Services
- Cell and Gene Therapy Catapult
- FUJIFILM Cellular Dynamics
- GenCure
- Lonza
- MEDINET
- Nikon CeLL innovation
- Progenitor Cell Therapy (PCT)
- Roslin Cells
- WuXi Advanced Therapeutics
For additional details, please visit https://www.rootsanalysis.com/reports/view_document/stem-cell-therapy-contract-manufacturing-market-2019-2030/271.html or email sales@rootsanalysis.com
You may also be interested in the following titles:
1. Global Stem Cell Therapy Market: Focus on Cardiovascular and Metabolic Disorders, 2019-2030
2. Vaccine Contract Manufacturing Market (2nd-edition), 2019-2030
3. Biopharma Contract Manufacturing Market (3rd Edition), 2019 - 2030
Roots Analysis
A430, 4th Floor, Bestech Business Towers, Sector 66, Mohali, India. PIN - 160059
gaurav.chaudhary@rootsanalysis
+1 (415) 800 3415
Leaders in Market Research focused on Biopharmaceutical and Medical Devices Industry
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release The contract manufacturing services market for stem cells is expected to be worth over USD 2.5 billion by 2030, claims Roots Analysis here
News-ID: 1845387 • Views: …
More Releases from Roots Analysis
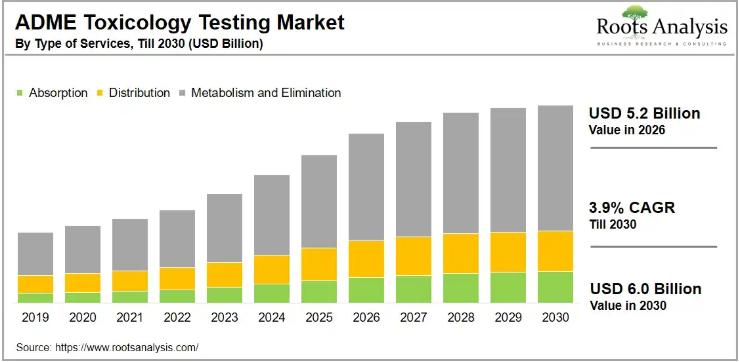
ADME Toxicology Testing Market to Reach USD 6.0 Billion by 2030 at 3.9% CAGR | C …
The global in vitro ADME toxicology testing market, valued at USD 4.5 billion in 2025, is on track to reach USD 5.2 billion in 2026 and USD 6.0 billion by 2030, growing at a compound annual growth rate of 3.9% over the forecast period. Rising pharmaceutical R&D investment, tightening regulatory requirements for drug approvals, and a growing industry-wide shift toward outsourcing preclinical testing services are all pushing demand higher at…
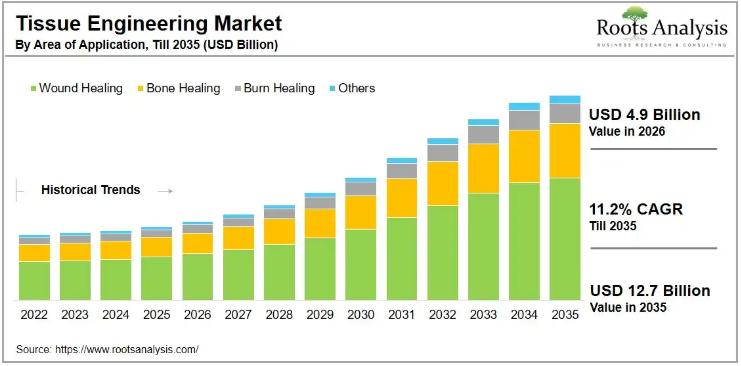
Tissue Engineering Market to Reach USD 12.7 Billion by 2035, Growing at 11.2% CA …
The global tissue engineering market, valued at USD 4.6 billion in 2025, will grow to USD 4.9 billion in 2026 and reach USD 12.7 billion by 2035, advancing at a compound annual growth rate of 11.2% through the forecast period. That trajectory reflects accelerating demand for regenerative therapies that can address chronic wounds, bone defects, and organ damage without the cost and complication burden that accompanies conventional transplantation. With more…
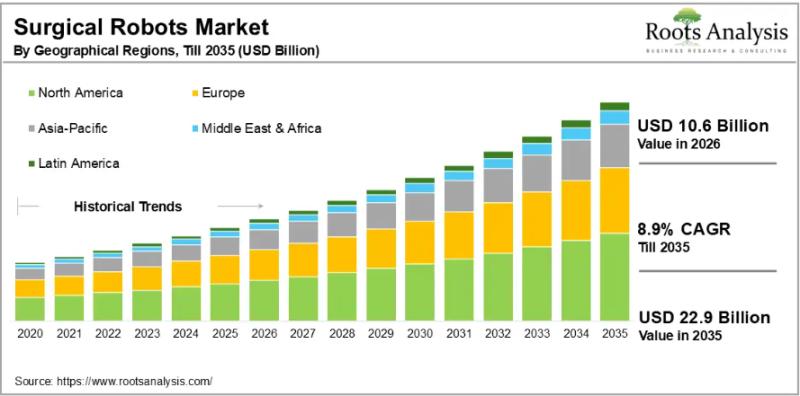
Surgical Robots Market to Reach $22.9 Billion by 2035 at 8.9% CAGR | Intuitive S …
The global surgical robots market, valued at USD 9.8 billion in 2025, will reach USD 10.6 billion in 2026 and climb to USD 22.9 billion by 2035, advancing at a compound annual growth rate (CAGR) of 8.9% across the 2026-to-2035 forecast window. That trajectory reflects sustained investment in robotic-assisted surgery platforms, a broad shift away from open surgical procedures toward minimally invasive techniques, and the accelerating integration of artificial intelligence…
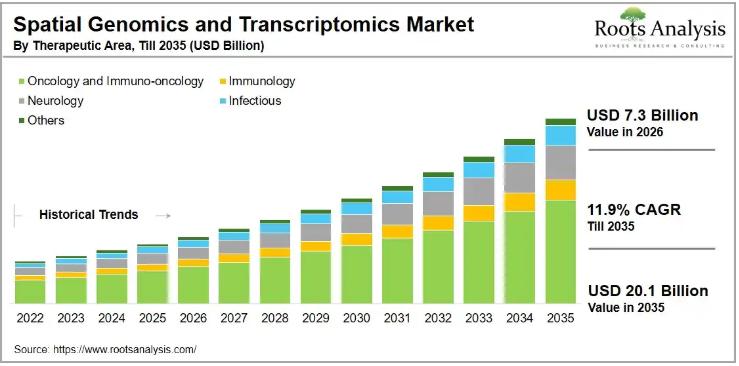
Spatial Genomics and Transcriptomics Market to Reach $20.1 Billion by 2035 at 11 …
The global spatial genomics and transcriptomics market, valued at USD 6.5 billion in 2025, is on course to reach USD 7.3 billion in 2026 and USD 20.1 billion by 2035, expanding at a compound annual growth rate (CAGR) of 11.9% over the forecast period 2026 to 2035. This growth trajectory reflects accelerating demand for high-resolution tissue analysis technologies that preserve spatial context, a capability conventional molecular profiling methods have historically…
More Releases for Cell
Cell Sorting Market Accelerates as Cell Therapy, Immuno-Oncology & Single-Cell R …
The rising focus on precision medicine, immunotherapy, and advanced cell-based research is driving the global cell sorting market into a high-growth phase. With expanding applications in stem cell therapy, CAR-T manufacturing, cancer immunology, and single-cell genomics, demand for accurate, high-purity cell isolation systems is stronger than ever. This release highlights key market trends, segmentation insights, technological innovations, and the factors shaping the future of cell sorting.
Download Full PDF Sample Copy…
Cell Isolation Cell Separation Market Size Analysis by Application, Type, and Re …
According to Market Research Intellect, the global Cell Isolation Cell Separation market under the Internet, Communication and Technology category is expected to register notable growth from 2025 to 2032. Key drivers such as advancing technologies, changing consumer behavior, and evolving market dynamics are poised to shape the trajectory of this market throughout the forecast period.
The market for cell isolation and separation is expanding rapidly as a result of sophisticated biotechnological…
Cell Free Protein Synthesis Market Beyond the Cell: Revolutionizing Protein Prod …
Cell-Free Protein Synthesis Market to reach over USD 457.13 Mn by the year 2031 - Exclusive Report by InsightAce Analytic
"Cell-Free Protein Synthesis Market" in terms of revenue was estimated to be worth $265.94 Mn in 2023 and is poised to reach $457.13 Mn by 2031, growing at a CAGR of 7.20% from 2024 to 2031 according to a new report by InsightAce Analytic.
Request for free Sample Pages: https://www.insightaceanalytic.com/request-sample/1445
Current…
Cell Expansion Market - Expand the Boundaries of Cell Therapy: Redefine Cell Exp …
Newark, New Castle, USA: The "Cell Expansion Market" provides a value chain analysis of revenue for the anticipated period from 2022 to 2030. The report will include a full and comprehensive analysis of the business operations of all market leaders in this industry, as well as their in-depth market research, historical market development, and information about their market competitors
Cell Expansion Market: https://www.growthplusreports.com/report/cell-expansion-market/7939
This latest report researches the industry structure, sales, revenue,…
Global GMP Cell Banking Market By Type - Mammalian Cell, Microbial Cell, Insect …
Researchmoz added Most up-to-date research on "Global GMP Cell Banking Market By Type - Mammalian Cell, Microbial Cell, Insect Cell and Others" to its huge collection of research reports.
This report researches the worldwide GMP Cell Banking market size (value, capacity, production and consumption) in key regions like North America, Europe, Asia Pacific (China, Japan) and other regions.
This study categorizes the global GMP Cell Banking breakdown data by manufacturers, region, type…
Cell Culture Market Size, Cell Culture Market Share, Cell Culture Market Trends …
According to a new research published by Polaris Market Research the global cell culture market is anticipated to reach more than USD 49 billion by 2026. Cell culture is a rapidly emerging as an implement for analyzing and treating various disease such as Alzheimer’s and cancer.
Request for Sample of This Research Report @ https://bit.ly/2D7pZ5u
Top Key Players: -
Becton,
Dickinson and Company
Biospherix
EMD Millipore
Eppendorf AG
Merck KGaA
Sartorius AG
VWR International
Cell culture is a rapidly emerging…