Press release
ADME Toxicology Testing Market to Reach USD 6.0 Billion by 2030 at 3.9% CAGR | Charles River Laboratories, WuXi AppTec, Eurofins Scientific
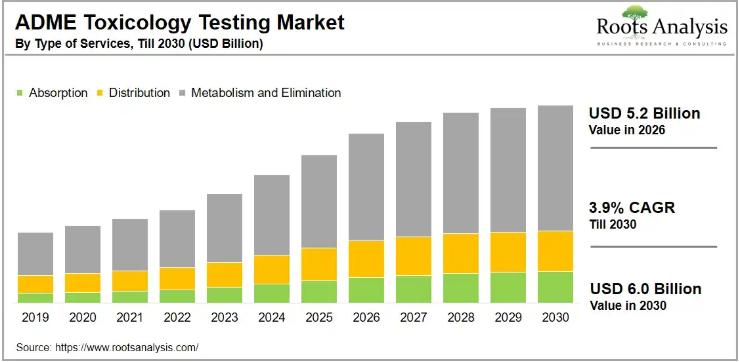
The global in vitro ADME toxicology testing market, valued at USD 4.5 billion in 2025, is on track to reach USD 5.2 billion in 2026 and USD 6.0 billion by 2030, growing at a compound annual growth rate of 3.9% over the forecast period. Rising pharmaceutical R&D investment, tightening regulatory requirements for drug approvals, and a growing industry-wide shift toward outsourcing preclinical testing services are all pushing demand higher at a steady, measurable pace.To explore the complete findings, request a free sample of the report at https://www.rootsanalysis.com/reports/adme-toxicology-testing-market/request-sample.html
Market Overview
ADME toxicology testing covers the study of how a drug is absorbed into the body, how it distributes across tissues, how it is metabolized, and how it is excreted. These tests serve as a critical filter during early drug discovery, identifying candidates with viable pharmacokinetic profiles before companies commit to expensive in vivo and clinical programs. Without reliable ADME data, pharmaceutical sponsors risk advancing molecules that will fail later, at far higher cost.
The pressure to reduce late-stage drug attrition is real and quantifiable. Industry studies on terminated drug programs have consistently identified poor pharmacokinetic profiles and ADME-related failures as primary causes of clinical trial dropout. Meanwhile, pharmaceutical R&D spending climbed from approximately USD 128 billion in 2008 to USD 165 billion in 2018, and the trajectory has continued upward since. That growing R&D base, combined with a persistent failure rate in clinical development, is creating durable demand for early-stage testing that can derisk the pipeline.
Over the past decade, the ADME toxicology testing sector has consolidated considerably. Larger contract research organizations (CROs) have systematically acquired smaller, specialized players to broaden their service portfolios and extend their global footprint. Today, more than 90 companies worldwide offer some form of in vitro ADME testing services, with 42% of providers classified as small companies. This mix of scale players and specialists creates a market that competes on breadth of assay capabilities, turnaround time, regulatory expertise, and geographic access.
Key Growth Drivers
Rising pharmaceutical R&D investment and drug development activity. Pharmaceutical and biotechnology companies are running larger, more complex pipelines than at any prior point in the industry's history. The growing incidence of chronic diseases, including oncological, neurological, and cardiovascular conditions, has intensified the push for new therapeutic candidates. ADME toxicology testing sits at the front end of that pipeline, ensuring compounds have the safety and metabolic profiles necessary to justify further development spend.
Stringent regulatory requirements from the FDA, EMA, and international bodies. Regulatory agencies including the US Food and Drug Administration and the European Medicines Agency have made ADME toxicology profiling a mandatory component of drug evaluation submissions. Compliance is not optional. These regulatory mandates act as a structural demand floor for testing services, regardless of broader economic conditions. Companies that want their drug candidates to advance through the approval process must generate this data.
Accelerating outsourcing of preclinical testing to specialized CROs. The high capital cost of ADME testing equipment and the difficulty of maintaining in-house expertise at scale have driven mid-sized and large pharmaceutical companies to rely on specialized contract service providers. Outsourcing partners that offer an integrated suite covering design, synthesis, safety pharmacology, and in vitro ADME testing under a single engagement provide significant operational advantages. This consolidation of services, rather than point-by-point procurement, is becoming the preferred operating model.
Technological advances in high-throughput screening and advanced in vitro systems. High-throughput screening tools and combinatorial chemistry have made ADME profiling faster and more cost-efficient at earlier stages of discovery. Beyond that, significant research investment is flowing into next-generation in vitro systems, including 3D cell culture assays, organ-on-chip platforms, and organoids, all designed to replace or supplement traditional 2D cell culture and animal testing. In April 2025, both Thermo Fisher Scientific and WuXi AppTec launched new high-throughput ADME toxicology platforms featuring advanced automation and data analytics, signaling that the technology investment cycle is accelerating.
Strategic acquisitions and funding rounds expanding service capacity. In January 2025, Evotec SE completed a EUR 150 million funding round to expand its ADME and toxicology testing infrastructure globally. In April 2025, Eurofins Scientific acquired XenoTech, a specialist in ADME and toxicology testing, to deepen its European capabilities in preclinical drug safety evaluations. These moves reflect a broader race among major CROs to secure capacity ahead of anticipated demand growth.
Market Segmentation
The in vitro ADME toxicology testing market segments across four primary service categories: absorption, distribution, metabolism, and excretion. Metabolism and excretion together account for the largest slice of current revenue, with metabolism and elimination alone projected to hold 60.3% of total revenue by 2030. Absorption, meanwhile, is the fastest-growing sub-segment, forecast to expand at a CAGR of 9.3% through 2030 as early-stage permeability and bioavailability screening commands greater attention from drug developers.
By molecule type, small molecules dominate and are projected to account for 56% of revenue by 2030, supported by their ease of administration and the volume of small-molecule programs in active development. The biologics segment is smaller today but growing faster, at a CAGR of 9.0% through 2030, as monoclonal antibodies, bispecifics, and peptide therapeutics move deeper into development pipelines. On the end-user side, pharmaceutical and biotechnology companies hold 67% of market share, while academic and research institutes, though a smaller contributor today, are the faster-growing end-user segment at a CAGR of 9.9% through 2030. By therapeutic area, oncology disorders command the largest share at 42.4% of revenue, with neurological disorders growing fastest at a CAGR of 6.7%.
To request quote of this report, please visit:
https://www.rootsanalysis.com/reports/adme-toxicology-testing-market/request-quote.html
Regional Insights
North America is the dominant geography in the ADME toxicology testing market, projected to hold 43% of global market share by 2030. The region benefits from the concentration of pharmaceutical and biotechnology headquarters, proximity to the FDA regulatory framework, and the presence of more than 40 established CROs offering ADME services. A senior scientific officer at a US-based CRO noted that while North American clients historically preferred domestic service providers, cross-border outsourcing is becoming more accepted, though the bulk of activity remains centered in this region.
Asia-Pacific is the fastest-growing region, forecast to expand at a CAGR of 8.7% through 2030. Rising interest in outsourcing from North American and European sponsors to lower-cost, high-capability providers in China and India is a primary driver. Companies such as WuXi AppTec, Pharmaron, Shanghai Medicilon, Syngene International, and GVK Biosciences have built substantial in vitro ADME capabilities in the region, enabling clients to access quality preclinical services at competitive pricing. Europe holds meaningful share as well, anchored by CROs with deep regulatory expertise for EMA-compliant submissions.
Competitive Landscape
Key service providers active in the in vitro ADME toxicology testing market include Albany Molecular Research (AMRI), Charles River Laboratories, Eurofins Scientific, Evotec, Galapagos, GVK Biosciences, Pharmaron, Pharmaceutical Product Development (PPD), RTI International, Sai Life Sciences, Shanghai Medicilon, Syngene International, Tecan Group, and WuXi AppTec.
The competitive dynamic in this market is shaped primarily by consolidation and capability breadth. Large CROs are acquiring smaller specialists to broaden their assay menus and geographic reach, while sponsors increasingly prefer integrated partners over point-solution providers. More than 35 partnership deals have been recorded among stakeholders in this field, underscoring how heavily the market is moving toward alliance-based and preferred-provider models. For smaller entrants, differentiation through proprietary technology platforms, particularly in advanced in vitro systems, remains the most viable path to competing against well-resourced incumbents.
Browse Full Report Description + Research Methodology + Table of Content + Infographics here:
https://www.rootsanalysis.com/reports/adme-toxicology-testing-market/239.html
Contact Details
Gaurav Chaudhary
Email: Gaurav.chaudhary@rootsanalysis.com or sales@rootsanalysis.com
Website: https://www.rootsanalysis.com
About Roots Analysis
Roots Analysis is a global leader in the pharma / biotech market research. Having worked with over 750 clients worldwide, including Fortune 500 companies, start-ups, academia, venture capitalists and strategic investors for more than a decade, we offer a highly analytical / data-driven perspective to a network of over 450,000 senior industry stakeholders looking for credible market insights. All reports provided by us are structured in a way that enables the reader to develop a thorough perspective on the given subject. Apart from writing reports on identified areas, we provide bespoke research / consulting services dedicated to serve our clients in the best possible way.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release ADME Toxicology Testing Market to Reach USD 6.0 Billion by 2030 at 3.9% CAGR | Charles River Laboratories, WuXi AppTec, Eurofins Scientific here
News-ID: 4514383 • Views: …
More Releases from Roots Analysis
Tissue Engineering Market to Reach USD 12.7 Billion by 2035, Growing at 11.2% CA …
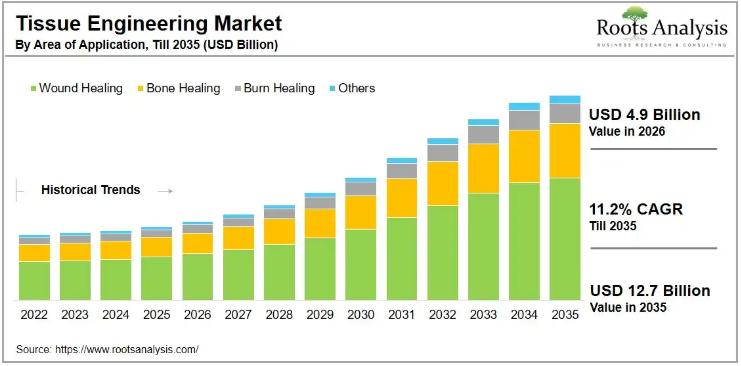
The global tissue engineering market, valued at USD 4.6 billion in 2025, will grow to USD 4.9 billion in 2026 and reach USD 12.7 billion by 2035, advancing at a compound annual growth rate of 11.2% through the forecast period. That trajectory reflects accelerating demand for regenerative therapies that can address chronic wounds, bone defects, and organ damage without the cost and complication burden that accompanies conventional transplantation. With more…
Surgical Robots Market to Reach $22.9 Billion by 2035 at 8.9% CAGR | Intuitive S …
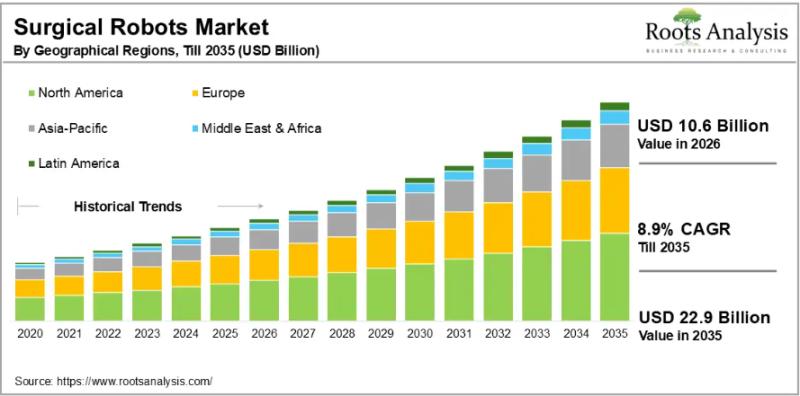
The global surgical robots market, valued at USD 9.8 billion in 2025, will reach USD 10.6 billion in 2026 and climb to USD 22.9 billion by 2035, advancing at a compound annual growth rate (CAGR) of 8.9% across the 2026-to-2035 forecast window. That trajectory reflects sustained investment in robotic-assisted surgery platforms, a broad shift away from open surgical procedures toward minimally invasive techniques, and the accelerating integration of artificial intelligence…
Spatial Genomics and Transcriptomics Market to Reach $20.1 Billion by 2035 at 11 …
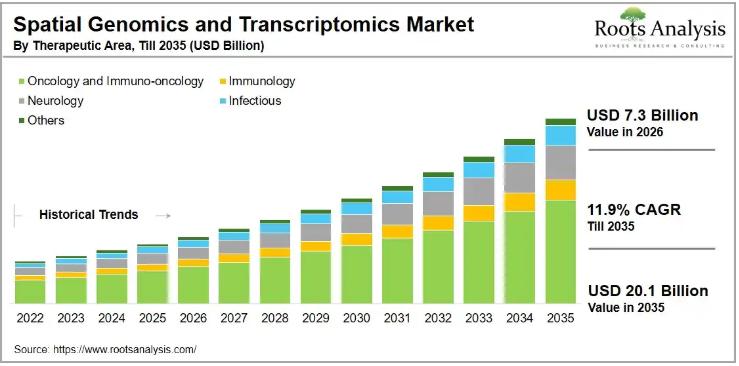
The global spatial genomics and transcriptomics market, valued at USD 6.5 billion in 2025, is on course to reach USD 7.3 billion in 2026 and USD 20.1 billion by 2035, expanding at a compound annual growth rate (CAGR) of 11.9% over the forecast period 2026 to 2035. This growth trajectory reflects accelerating demand for high-resolution tissue analysis technologies that preserve spatial context, a capability conventional molecular profiling methods have historically…
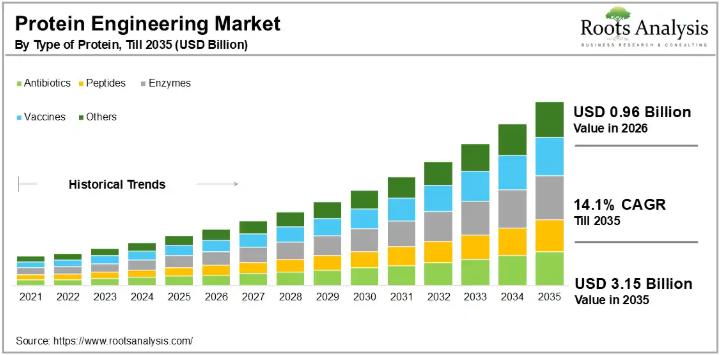
Protein Engineering Market to Reach $3.15 Billion by 2035 at 14.1% CAGR | ATUM, …
The global protein engineering market, valued at USD 0.85 billion in 2025, will grow to USD 3.15 billion by 2035, advancing at a compound annual growth rate of 14.1% over the 2026 to 2035 forecast period. The market is already crossing the USD 0.96 billion mark in 2026, signaling strong near-term momentum. With more than 85 specialized companies active in the space and over 550 patents filed or granted in…
More Releases for ADME
ADME Toxicology Testing Market Trends That Will Shape the Next Decade: Insights …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
How Large Will the ADME Toxicology Testing Market Size By 2025?
There has been a swift expansion in the market size of ADME toxicology testing in the past few years. The market, which is projected to surge from $10.4 billion in 2024 to $11.53 billion in 2025, is expected…
Radiolabelled ADME Studies Market Outlook and Future Projections for 2030
The radiolabelled adme studies market represents a dynamic and continually evolving landscape, shaped by changing consumer demands and technological advancements. In this comprehensive report, we provide an in-depth exploration of the market, designed for a wide range of stakeholders including manufacturers, suppliers, distributors, and investors. Our goal is to equip industry participants with essential insights that enable informed decision-making in an ever-changing market environment. This analysis not only examines the…
ADME Toxicology Testing Market Know the Scope and Trends
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "ADME Toxicology Testing Market"- By Type of Services (Absorption, Distribution, Metabolism, Excretion), Type of Assays(Batch / Fed-Batch, Continuous), Type of Molecule (Biologics, Small Molecules), End User(Pharmaceutical and Biotechnology Companies, Academic / Research Institutes), Therapeutic Areas(Blood Disorder, Cardiovascular Disorder, Gastrointestinal and Vascular Disorder, Hormonal Disorder, Infectious Diseases, Immunological Disorders, Mental Disorders, Metabolic Disorders, Neurological Disorders, Oncology…
Leading Element Driving Change in the ADME Toxicology Testing Market in 2025: Su …
How Are the key drivers contributing to the expansion of the adme toxicology testing market?
The ADME toxicology testing market's expansion is predicted to be fueled by an increasing need for new medicines. These are pharmaceutical substances that haven't been previously sanctioned or marketed for therapeutic purposes. ADME toxicity testing is a critical phase in drug development, ensuring that the new medicine is safe and doesn't generate any harmful toxins in…
Global ADME Toxicology Testing Market Insights and Forecast 2024
"The Business Research Company recently released a comprehensive report on the Global ADME Toxicology Testing Market Size and Trends Analysis with Forecast 2024-2033. This latest market research report offers a wealth of valuable insights and data, including global market size, regional shares, and competitor market share. Additionally, it covers current trends, future opportunities, and essential data for success in the industry.
Ready to Dive into Something Exciting? Get Your Free Exclusive…
Radiolabelled ADME Studies Market Outlook and Future Projections for 2030
The radiolabelled adme studies market represents a dynamic and continually evolving landscape, shaped by changing consumer demands and technological advancements. In this comprehensive report, we provide an in-depth exploration of the market, designed for a wide range of stakeholders including manufacturers, suppliers, distributors, and investors. Our goal is to equip industry participants with essential insights that enable informed decision-making in an ever-changing market environment. This analysis not only examines the…