Press release
Novel Drug Delivery Systems to Augment Presbyopia Therapeutics Pipeline
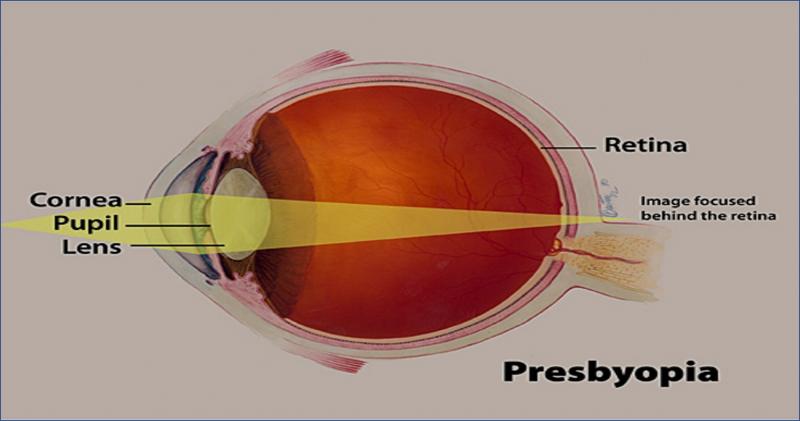
Presbyopia is a common vision disorder that manifests with increasing age. With age, the ciliary muscles of the eye start losing their flexibility that leads to the eye lens losing its flexibility as well. The eye gradually loses its ability to focus light directly on to the retina, and the image ends up forming behind it. This results in poor visibility of objects that are close to the eye. The normal aging process that causes presbyopia cannot be stopped or reversed. However, it can be rectified by wearing contact lenses and eyeglasses, or via surgeries.Download the report sample at: https://www.pharmaproff.com/request-sample/1233
Signs and symptoms associated with presbyopia are a difficulty in reading small prints, headaches, eye muscle fatigue, and eyestrain. One of its early signs is blurred vision while holding things at the normal reading distance. Working in dimly lit environments could exacerbate the condition. Its diagnosis is done based on the patient’s age and medical history. In case of premature presbyopia, medical history becomes more important as it may correlate with a systemic disease. Conditions, such as neurological and vascular diseases, trauma, and diabetes mellitus and certain drugs, may lead to premature presbyopia.
The treatment for presbyopia is based on ophthalmologists and optometrists’ recommendations. The initial treatment involves prescribing of near-vision optical aids, such as reading glasses, magnifying lenses, and monocles. Vison-correction surgeries are also used for the treatment for long-sightedness (another term for the condition), as is the experimental pharmaceutical approach. One of the investigational drugs in the presbyopia therapeutics pipeline is naphazoline. Other drugs, such as polyethylene glycol, pheniramine, and nepafenac, are being tried with alpha-1 agonists, as they reduce ciliary muscle spasm, vascular congestion, and pilocarpine-induced hyperemia, thereby treating excess pupil constriction.
Get the detailed analysis at: https://www.pharmaproff.com/report/presbyopia-therapeutics-pipeline-analysis
One of the key driving factors for the presbyopia therapeutics pipeline is the invention of novel drug delivery systems. For example, AcrySof IQ PanOptix Toric intraocular lens for presbyopia was launched by the eye care division of Novartis, which is seen as a significant development in the presbyopia treatment scene. This is indicated for adults who have undergone cataract surgery and wish to achieve independence from spectacles. Similarly, the U.S. Food and Drug Administration approved the Raindrop Near Vision Inlay to be implanted in the cornea for presbyopia correction, in 2016.
The presbyopia therapeutics pipeline is segmented by development phase, route of administration, and molecule type. One of the candidates in the pipeline is carbachol and brimonidine drops, being developed by Principal Investigator Almamoun Abdelkader of the Department of Ophthalmology, Faculty of Medicine, Al-Azhar University, Cairo. Carbachol is a positively charged quaternary ammonium compound and a synthetic choline ester. The effect of acetylcholine on nicotinic and muscarinic receptors is mimicked by carbachol, which is a parasympathomimetic compound. The intraocular pressure is ultimately reduced due to the induction of miosis in the eye.
Brimonidine is a selective alpha-2 adrenergic receptor agonist and an imidazole derivative. It binds to these receptors present on the presynaptic nerve endings of the dilator muscle, which inhibits the neurotransmitter release into the synaptic cleft. This reduces the activity of the dilator muscle, thereby leading to a more-mitotic pupil. The production of aqueous humor is decreased due to the action of this compound on blood vessels, which causes them to constrict.
Therefore, as more such combination therapies are devised, people suffering from presbyopia might not have such a hard time in the coming years.
Contact Us:
Pharma Proff
Toll-free: +1-888-778-7886 (USA/Canada)
International: +1-347-960-6455
Email: enquiry@pharmaproff.com
Web: www.pharmaproff.com
About Us:
Pharma Proff, is a frontline market intelligence and consulting solutions provider catering to the information and data needs of burgeoning sectors across the world, including pharmaceutical and healthcare. With meticulously curated research reports on the industry landscape, we empower companies in the healthcare and pharmaceutical sectors to make informed business decisions and base their marketing strategies with astuteness.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Novel Drug Delivery Systems to Augment Presbyopia Therapeutics Pipeline here
News-ID: 1806123 • Views: …
More Releases from Pharma Proff
Interleukin-6 (IL-6) Inhibitors Analysis, Clinical Trails and Developments
Interleukin-6 (IL-6) inhibitors currently exhibit a proliferating pipeline with 25+ therapeutic candidates.
IL-6 is a cytokine encoded by IL-6 gene, which helps in the maturation of B cells and is actively involved in the inflammatory response. The IL-6 protein is primarily produced at the sites of acute and chronic inflammation, where it is secreted into the serum and brings a transcriptional inflammatory response through IL-6? receptor. This protein is assumed to…
Retinitis Pigmentosa Therapeutics Analysis, Clinical Trials and Other Developmen …
Retinitis pigmentosa therapeutics currently exhibits a proliferating pipeline with 25+ therapeutic candidates.
Retinitis pigmentosa (RP) is a group of hereditary disorders that lead to the breakdown and loss of cells in the retina. Mutation in genes encoding the proteins required by retinal cells for their proper functioning, results in occurrence of RP. The disease results in partial vision loss during the earlier stage of the disease, which may lead to complete…
Probiotics Therapeutics -Pipeline Analysis 2018, Clinical Trials & Results, Pate …
Probiotics are live microorganisms that produces beneficial effects in human gut. Probiotics can be taken as a dietary supplement. They prevent diarrhoea and help to reduce the symptoms of irritable bowel syndrome.
Download the sample report at: https://www.pharmaproff.com/request-sample/1189
Probiotics comprise of a variety of microorganisms belonging to the Lactobacillus and Bifidobacterium groups. The only yeast probiotic that has proven to be effective is Saccharomyces boulardii. According to researchers, probiotics are effective in…

Niemann-Pick Disease Type C (NPC) Therapeutics Pipeline to Witness Significant G …
Niemann-Pick disease is divided into four main types: type A, type B, type C1, and type C2. These types are classified on the basis of genetic cause and signs and symptoms of the disease. NPC is identified as a rare genetic disorder in which the body is unable to transport cholesterol and lipids inside the cells. This causes accumulation of these substances within various tissues of the body. These substances…
More Releases for Presbyopia
Presbyopia Pipeline Assessment Report 2025 | DelveInsight
DelveInsight's, "Presbyopia Pipeline Insight, 2025" report provides comprehensive insights about 10+ companies and 10+ pipeline drugs in Presbyopia pipeline landscape. It covers the Presbyopia pipeline drug profiles, including clinical and nonclinical stage products. It also covers the Presbyopia pipeline therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Discover the latest drugs and treatment options in the Presbyopia…
Rising Geriatric Population Fuels Growth Of Myopia And Presbyopia Treatment Mark …
The Myopia and Presbyopia Treatment Market Report by The Business Research Company delivers a detailed market assessment, covering size projections from 2025 to 2034. This report explores crucial market trends, major drivers and market segmentation by [key segment categories].
What Is the Myopia and Presbyopia Treatment Market Size and Projected Growth Rate?
The market size for myopia and presbyopia treatment has witnessed substantial growth over several recent years. The market is projected…
Myopia and Presbyopia Treatment Market: Addressing Vision Impairment Globally
The global market for myopia and presbyopia treatment has been witnessing remarkable growth, driven by the rising prevalence of vision impairment worldwide. As per recent findings from Transparency Market Research, the market was valued at approximately US$16.7 billion in 2021 and is projected to reach nearly US$45 billion by 2032, with a promising CAGR of 9.5% over the next decade. Corrective lenses emerge as the leading treatment type, with revenue…
Presbyopia Treatment Market Growth Analysis 2023-2030
Global Presbyopia Treatment Market Gains Momentum Amidst Rising Aging Population and Innovations in Therapeutics:
The global presbyopia treatment market has witnessed substantial growth in recent years, fueled by the increasing prevalence of presbyopia and the expanding aging population. Presbyopia, a common vision loss associated with aging, has prompted a surge in demand for effective treatments, leading to advancements in eyeglasses, contact lenses, surgeries, eye drops, and nutrient-based interventions.
Refractive surgeries, including Conductive…
Presbyopia Pipeline: Insights into Novel Pipeline Therapies, Key Pharma Companie …
DelveInsight's, "Presbyopia Pipeline Insight, 2022," report provides comprehensive insights about 12+ companies and 12+ pipeline drugs in Presbyopia pipeline landscape. It covers the pipeline drug profiles, including clinical and nonclinical stage products. It also covers the therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Key takeaways from the Presbyopia Pipeline Insight Report
• Presbyopia Pipeline report offers a comprehensive…
Presbyopia Market 2020-2027 Sets the Table for Continued Growth || Top Growing C …
A transparent research method has been accomplished with the right tools and techniques to make this Presbyopia Market research report world-class. Two of the most widely used techniques namely SWOT analysis and Porter's Five Forces Analysis have been used while generating this report. Competitive analysis conducted in this report puts light on the moves of the key players in the Healthcare industry such as new product launches, expansions, agreements, joint…