Press release
Latest Guidebook for Review and Approval Procedures of Overseas Imported New Drugs for Chinese Clinical Urgent Demand (2019 Edition)
China is one of the fastest growing global economies with one fifth population in the world. Nowadays, China has become the world's second largest healthcare market after the United States.The Chinese “National Medical Products Administration (NMPA)” and the “National Health Commission (NHC)” jointly issued the latest “Review and Approval Procedures of Overseas Imported New Drugs for Clinical Urgent Demand” on October 23, 2018, which provided a dedicated pathway for priority review and approval of overseas drugs importing to Chinese healthcare market and clarified the specific review and approval procedures.
View Full Report: https://reports.valuates.com/sreport/ACCE-Book-32/Guidebook
Key Highlights:
—A latest description for the China’s changing healthcare market landscape and rapidly changing regulatory framework that let the overseas and multinational pharmaceutical manufacturers clearly understand the present-day realities of the Chinese healthcare market landscape and rapidly changing regulatory framework, and tell the overseas and multinational pharmaceutical manufacturers the opportunities and challenges.
—The latest regulations on priority review and approval procedures of overseas imported new drugs of Chinese clinical urgent demand to smoothly navigate regulatory requirements step by step.
—The scope and selection of drug variety for the priority review and approval procedures of overseas imported new drugs for Chinese clinical urgent demand.
—The detailed Chinese drug regulatory authority's requirements for application materials that the overseas applicants apply for the overseas imported new drugs of Chinese clinical urgent demand. The overseas applicants should prepare the application materials in strict accordance with these requirements.
—The duties and obligations of the overseas applicants on the drugs on the post-marketing in China that are stipulated by the Chinese drug regulatory authority. The overseas applicants must undertake their duties and obligations of their drugs on the post-marketing in China. Otherwise, when the drug on post-marketing has been confirmed to have serious adverse reactions, the Chinese drug regulatory authority will take the emergency control measures to stop sale and use of the drug.
—The detailed Chinese drug regulatory authority's latest “Administrative Measures for Communication and Exchange on Drug Research and Development and Technical Review and Approval” to guide the overseas applicants to take the key first step entry into the dedicated pathway of priority review and approval procedures and to smoothly pass the review and approval procedures.
—A full set of the English and Chinese bilingual application form, materials and minutes template for Communication and Exchange Meeting, which cover the “Application Form of Communication and Exchange Meeting” (annex 1), “The Materials for Communication and Exchange Meeting” (annex 2) and the "Communication and Exchange Meeting Minutes Template" (annex 3) to guide the overseas applicants how to communicate with the Chinese drug regulatory authority.
—The first batch list of overseas imported new drugs for Chinese clinical urgent demand that has been selected by the Chinese drug regulatory authority, which is calling the overseas applicants to submit the application for marketing in China to the Chinese drug regulatory authority, also let the overseas pharmaceutical manufacturers understand the Chinese drug regulatory authority’s distinctive regulatory current status. The list covers 40 drug varieties that have been approved to market in the United States, EU or Japan but that have not been yet marketed in China, and involves with 33 overseas pharmaceutical manufacturers.
Inquire for Sample: https://reports.valuates.com/request/sample/ACCE-Book-32/Guidebook
The audiences of this guidebook are the overseas pharmaceutical manufacturers wishing to enter into the Chinese healthcare market, and the multinational pharmaceutical manufacturers have penetrated into the Chinese healthcare market, as well as their senior executive officers engaging in regulatory affairs expecting to understand the latest regulations on review and approval procedures of overseas imported new drugs for Chinese clinical urgent demand. After having skimmed through this guidebook, audiences can clearly acquire a comprehensive and thorough knowledge of the latest regulations on priority review and approval procedures of overseas imported new drugs for Chinese clinical urgent demand. Access China Management Consulting Ltd hopes this guidebook, based on the full and accurate regulations, can guide the overseas and multinational pharmaceutical manufacturers to achieve a successful entry into the Chinese healthcare market, and smoothly operate their companies in China.
Reasons to Buy:
The Chinese “National Medical Products Administration (NMPA)” and the “National Health Commission (NHC)” jointly issued the latest “Review and Approval Procedures of Overseas Imported New Drugs for Clinical Urgent Demand” on October 23, 2018, which provided a dedicated pathway for priority review and approval of overseas drugs importing to Chinese healthcare market and clarified the specific review and approval procedures. This is a huge business opportunities for the overseas pharmaceutical manufacturers. To capture the huge business opportunities of the Chinese healthcare market and seize a larger part of Chinese healthcare market, how do the foreign pharmaceutical manufacturers in compliance with the latest “Review and Approval Procedures of Overseas Imported New Drugs for Clinical Urgent Demand”? How do the overseas pharmaceutical manufacturers operate business smoothly in China? The overseas and multinational pharmaceutical manufacturers and their senior executive officers engaging in regulatory affairs need a thorough knowledge of the latest regulations for review and approval procedures of overseas imported new drugs for Chinese clinical urgent demand. The Chinese regulatory approach is unique.
The Latest Guidebook for Review and Approval Procedures of Overseas Imported New Drugs for Chinese Clinical Urgent Demand (2019 Edition) is an essential resource for overseas and multinational pharmaceutical manufacturers to successfully acquire the marketing authorization in China, which provided a detailed guidance for comprehensive knowledge of the latest regulations on review and approval procedures of overseas imported new drugs for Chinese clinical urgent demand to navigate regulatory requirements step by step.
Inquire for Discount: https://reports.valuates.com/request/discount/ACCE-Book-32/Guidebook
Our aim is to collate unparalleled Market insights and notify our customers as and when it happens. Valuates is curating premium Market Research Reports from the leading publishers around the globe. We will help you map your information needs to our report repository of Market research reports and guide you through your purchasing decision. We are based out of silicon valley of India (Bengaluru) and provide 24/7 online and offline support to all our customers and just a phone call away.
For Similar Reports:
Access China: https://reports.valuates.com/search?qccode=All&qstr=acce&qcname=All&category=
Valuates Reports
sales@valuates.com
For U.S. Toll Free Call +1 (425) 388-2538
For IST Call +91-8040957137
To know more visit us on:
Website: https://reports.valuates.com/
Linkedin: www.linkedin.com/company/valuatesreports
Facebook: www.facebook.com/valuatesreports
Twitter: www.twitter.com/valuatesreports
Instagram: www.instagram.com/reportsvaluates
Youtube: www.youtube.com/channel/UCH4wNXynaTZbiD5m92WQI4A
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Latest Guidebook for Review and Approval Procedures of Overseas Imported New Drugs for Chinese Clinical Urgent Demand (2019 Edition) here
News-ID: 1755445 • Views: …
More Releases from Valuates Reports
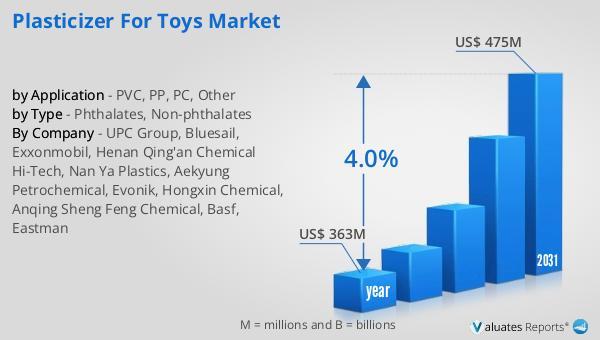
Plasticizer for Toys Market Share Driven by Stringent Safety Regulations and Shi …
Plasticizer for Toys Market Size
The global market for Plasticizer for Toys was valued at US$ 363 million in the year 2024 and is projected to reach a revised size of US$ 475 million by 2031, growing at a CAGR of 4.0% during the forecast period.
View sample report
https://reports.valuates.com/request/sample/QYRE-Auto-20U18169/Global_Plasticizer_for_Toys_Market_Research_Report_2024
The Plasticizer for Toys Market is witnessing steady market growth driven by increasing safety regulations and rising demand for non-toxic, child-safe materials. Plasticizers are…

Global Memory Interface Chip Market Intelligence: US$ 3.6 Billion Projection by …
Memory Interface Chip Market Size
The global Memory Interface Chip revenue was US$ 641.4 million in 2022 and is forecast to a readjusted size of US$ 3,612.7 million by 2029, growing at an impressive CAGR of 28.5% during the forecast period (2023-2029).
Get Free Sample: https://reports.valuates.com/request/sample/QYRE-Auto-19X16265/Global_and_India_Memory_Interface_Chip_Market_Report_Forecast_2023_2029
Major Trends
• Transition to 6400+ MT/s Speeds: The industry is rapidly standardizing higher speed tiers, pushing the limits of traditional RCD and Data Buffer designs.
• AI-Optimized Data Buffering: Manufacturers…

Global High Speed Memory Interface Chip Market Set for Resilient Growth, Project …
High Speed Memory Interface Chip Market Size
The global market for semiconductor was estimated at US$ 579 billion in the year 2022 and is projected to reach US$ 790 billion by 2029, growing at a CAGR of 6% during the forecast period.
Get Free Sample: https://reports.valuates.com/request/sample/QYRE-Auto-2X14355/Global_High_Speed_Memory_Interface_Chip_Market_Research_Report_2023
Major Trends
• Transition to DDR5-6400+: The industry is rapidly standardizing higher speed tiers, pushing the physical limits of traditional RCD and DB designs.
• AI-Centric Memory Architecture: Hyperscalers are adopting…
Global EV Memory Market Acceleration: US$ 4.45 Billion Projection by 2030, Fuele …
EV Memory Market Size
The global EV Memory market was valued at US$ 603 million in 2023 and is anticipated to reach US$ 4,453 million by 2030, witnessing an unprecedented CAGR of 31.7% during the forecast period 2024-2030.
• Growth Drivers by Segment: The DRAM segment is emerging as the largest contributor to market value, driven by the massive throughput required for on-vehicle AI inference in ADAS. In terms of application, the Vehicle…
More Releases for Chinese
Chinese New Year: Celebrate with Chinese Spirits in Exquisite Glass Bottle Packa …
Image: https://ecdn6.globalso.com/upload/p/3535/image_other/2026-02/you-are-welcome.png
As the most grand traditional festival in China, Chinese New Year is a time for family reunion, blessing exchange and warm gatherings. When the Spring Festival couplets are pasted and the red lanterns are lit, a bottle of mellow Chinese spirits housed in elegant Glass Bottle [https://www.jpglassbottle.com/glass-bottle-1/] packaging becomes an indispensable part of the celebration. Among numerous Chinese spirits brands, Moutai and Wuliangye stand out as iconic representatives, using…
Download Telegram & Telegram Chinese or Telegram in Chinese - Complete Guide
In today's connected world, messaging apps are essential for communication, business, and content sharing. One of the most popular platforms globally is Telegram. If you are searching for how to Download Telegram or want to understand Telegram Chinese or Telegram in Chinese, this detailed guide will help you get started easily. You can also visit https://www.telegram-ios.com/ for more information and updates related to Telegram.
________________________________________
Download Telegram: Step-by-Step Guide
If you want to…
Gao Yuan at UN Chinese Language Day: Bridging Cultures Through Traditional Chine …
On April 20, 2025, CCTV host Gao Yuan participated in the UN Chinese Language Day celebrations at the United Nations Headquarters in New York, showcasing the profound heritage and innovative vitality of traditional Chinese arts to a global audience.
Image: https://www.globalnewslines.com/uploads/2025/04/556563a6bf5cb3de8982c8ef94cb6380.jpg
During the thematic forum "Civilizational Dialogue and Artistic Symbiosis," Gao Yuan moderated a cultural salon titled "Digital Technology Empowers Traditional Arts." She highlighted China's recent efforts to reinterpret calligraphy and ink…
The TV program "Chinese Practice with Chinese Wisdom" Officially Airs: The Endur …
Chinese Practice with Chinese Wisdom is a 10-episode TV program produced by Jiangsu Broadcasting Corporation, one of the most influential media groups in China. It gives a vivid presentation of traditional Chinese cultural elements such as poetry, calligraphy, seal-cutting, traditional Chinese operas, dance, and martial arts, aiming to enrich audiences with cultural enlightenment and aesthetic enjoyment. Through questions, disucssions, elaborations, feelings, and stories shared by guests from around the world,…
Chinese Fenjiu Shines at SCWC Conference, Bridging Chinese Liquor and Western Cu …
Fenjiu Debuts at the International Stage, Blending Tradition, Innovation, and Cultural Fusion, while Captivating Global Food and Beverage Experts.
Barcelona, Spain - November 22, 2024 - Fenjiu, a premier Chinese liquor brand, made a landmark appearance at the Science and Cooking World Congress (SCWC) in Barcelona, Spain. Renowned for its light-fragrance style and rich cultural heritage, Fenjiu captivated global food and beverage professionals as it showcased its unique blend of tradition,…
Introducing A New Chinese Reader to Enable Chinese Language Students to Master C …
Capturing Chinese presents a new advanced Chinese reader helping students master the literature of China's best authors. Allowing students to put down their dictionaries and instead curl up with a book of Chinese literature, Capturing Chinese is a must have for intermediate and advanced Chinese language students.
The new book, titled Capturing Chinese Stories: Prose and Poems by Revolutionary Authors, features five of the most influential authors in recent Chinese history…
