Press release
New Detailed Review of Oral Ketamine for Depression Published in JCP
Memphis, TN (April 16, 2019) – A review article published today in The Journal of Clinical Psychiatry indicates that orally administered ketamine is efficacious for depression but may take longer to yield results. The review, authored by Joshua D. Rosenblat and colleagues at the University of Toronto, analyzed 13 articles, including 2 randomized controlled trials (RCTs).The RCTs demonstrated antidepressant efficacy and good tolerability for oral ketamine, but significant changes in depressive symptom severity were observed only after 2–6 weeks of treatment. This lag time is in contrast to the rapid effects of intravenous ketamine, which has been investigated for its antidepressant and antisuicidal effects over the past 2 decades.
Ketamine is an anesthetic agent that works differently from currently available antidepressants. Intranasal esketamine, approved by the FDA in March for treatment-resistant depression, and oral ketamine represent alternatives to intravenous ketamine, which can present challenges in terms of accessibility.
Due to risks of sedation and dissociative effects, as well as possible misuse, the FDA approval of intranasal esketamine requires that the drug be administered in a medical office where the patient can be monitored for 2 hours afterward. Rosenblat and colleagues note that, intriguingly, dissociative effects, often reported with IV ketamine, were rarely observed in the studies of oral ketamine. However, they caution that oral ketamine might lend itself to take-home doses that could be diverted or misused without adequate precautions. Given these concerns, along with insufficient current evidence, they emphasize that prescribing oral ketamine for depression cannot be recommended until further evidence is available.
Access the entire article, “Oral Ketamine for Depression: A Systematic Review,” by Joshua D. Rosenblat, MD; Andre F. Carvalho, MD, PhD; Madeline Li, MD, PhD; Yena Lee, BSc; Mehala Subramanieapillai, MSc; and Roger S. McIntyre, MD, at https://doi.org/10.4088/JCP.18r12475.
The Journal of Clinical Psychiatry, a peer-reviewed publication that is indexed and abstracted by the National Library of Medicine’s PubMed, is the official publication of the American Society of Clinical Psychopharmacology and is among the top 10 most cited psychiatry journals worldwide.
Physicians Postgraduate Press, Inc.
Post Office Box 752870
Memphis, Tennessee 38175-2870
901-273-2711
JClinPsych@Psychiatrist.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release New Detailed Review of Oral Ketamine for Depression Published in JCP here
News-ID: 1705762 • Views: …
More Releases from Physicians Postgraduate Press
Antioxidants May Aid in the Treatment of Tardive Dyskinesia
Memphis, TN (May 9, 2019) – Today, The Primary Care Companion for CNS Disorders published a case report on the use of antioxidants in the treatment of tardive dyskinesia. While new pharmacologic discoveries have occurred in the area of movement disorders, their use comes with a large price tag. Thus, for mild cases, clinicians may want to seek out more cost-effective options. Antioxidants may serve a role in the prevention…
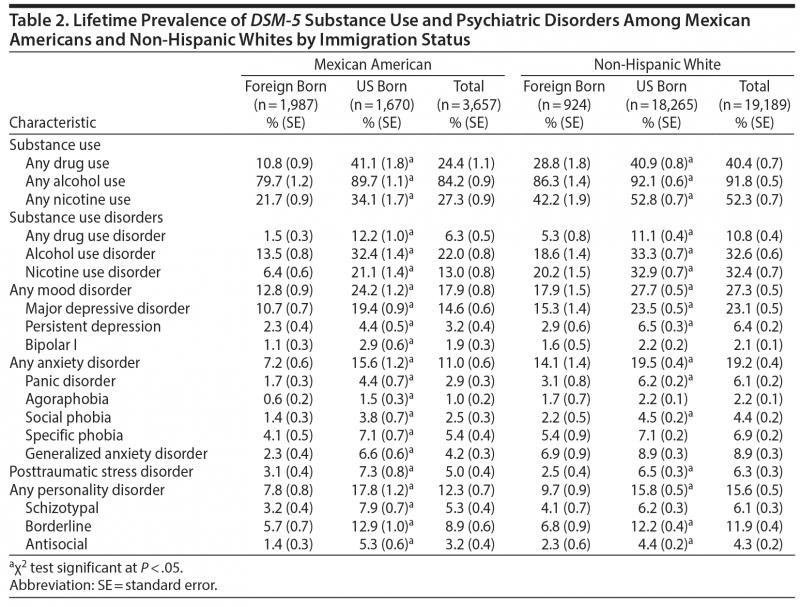
Immigrants at Lower Risk of Substance Use, Psychiatric Disorders Than US-Born Co …
Memphis, TN (February 28, 2019) – Today The Primary Care Companion for CNS Disorders published a study comparing prevalence rates of alcohol, nicotine, and other drug use and major psychiatric disorders between US-born and foreign-born Mexican Americans and non-Hispanic whites.
Hispanics are the leading ethnic majority in the US, and Mexican Americans constitute 60% of the US Hispanic population. Therefore, focus on this group is timely and important, especially since…
More Releases for Clinical
Miami Clinical Research Sets the Standard for Clinical Trials
Miami Clinical Research, a frontrunner in the world of clinical trials and medical research, has emerged as the prime choice for global corporate pharmaceutical giants. With a deep understanding of the complexities of medical studies, the organization champions the crucial role of research in the evolution of transformative therapeutic interventions.
Miami, FL - Renowned as a first-rate center for professional medical exploration, Miami Clinical Research [https://miamiclinicalresearch.com] boasts state-of-the-art facilities, advanced technologies,…
E-Clinical Solutions Market: Revolutionizing Healthcare and Clinical Trials
Introduction
The e-Clinical solutions market has become a pivotal component of the healthcare and pharmaceutical industries. E-Clinical solutions refer to a set of software, tools, and platforms designed to streamline clinical trials and healthcare management. These solutions include electronic data capture (EDC), clinical trial management systems (CTMS), laboratory information management systems (LIMS), and other integrated tools that improve the efficiency, accuracy, and speed of clinical trials and healthcare services. The primary…
E-Clinical Solutions Market: Revolutionizing Clinical Trials
The e-clinical solutions market has experienced significant growth in recent years, driven by the increasing complexity of clinical trials and the need for efficient, accurate, and compliant data management. E-clinical solutions provide a comprehensive suite of tools and technologies to streamline clinical trial processes, accelerate drug development, and improve patient outcomes.
Market Size and Growth
The global e-clinical solutions market is estimated to be worth billions of dollars, with a significant portion…
Clinical Trials Management System Market Optimizing Clinical Trials: The Crucial …
Clinical Trials Management System Market to reach over USD 5.06 billion by the year 2031- Exclusive Report by InsightAce Analytic
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Clinical Trials Management System Market Size, Share & Trends Analysis Report By Solution Type (Enterprise and Site based), By Delivery Mode (Web & Cloud-based, On-premise), By Component (Software, Services), By End-user (Pharmaceutical and Biotechnology Firms, Medical…
Clinical Research and Clinical Trials Summit
Clinical Research 2019 has been designed in an interdisciplinary manner with a multitude of tracks to choose from every segment and provides you with a unique opportunity to meet up with peers from both industry and academia and establish a scientific network between them. We cordially invite all concerned people to come join us at our event and make it successful by your participation.
This is the premier interdisciplinary forum for…
E-Clinical Trial Solutions Market To Accelerating Clinical Development Technolog …
The study of the "Global e-Clinical Trial Solutions Market" provides the market size information and market trends along with the factors and parameters impacting it in both short and long term. The study ensures a 360° view, bringing out the complete key insights of the industry.
The Global e-Clinical Trial Solutions Market Research Report Forecast 2017-2021 is a valuable source of insightful data for business strategists. It provides the e-Clinical…