Press release
Sterility Testing Market
Sterility testing in pharmaceutical manufacturing is a vital process during the sterilization and validation process along with release testing. It is an essential part of every sterilization validation process in several industries that makes it a vital procedure in manufacturing industry. It is an extremely difficult process that should be planned and executed with precision, which can be helpful in eliminating false positive results. Laboratory contamination or technician errors are the main causes of false positive results. Planned testing should be carried out in a clean room, where there is no or minimum chances of error are seen. This would also ensure compliance with U.S. Pharmacopeia (USP) requirements in terms of viable microbial air and surface counts. The unique requirements to employ sterility testing procedures as an official test and to determine the suitability are regulated under the good manufacturing practices guidelines.An understanding of sterility testing benefits in validation process, designing is an important quality assurance issue is the need of providing an appropriate and authentic sterility test data. Furthermore, sterility testing is a tedious and artful process, which requires to be carried out by trained professionals. Also, investigation during the failure of sterility test, is a process that needs close look in environmental data along with other factors such as sample collection and sample procurement procedures. The rise in demand for sterilized products and their exclusivity in various industries is expected to fuel growth of the sterility testing market in the near future. The growth media utilized during the sterility testing must be properly prepared and tested to make the microbial growth adequately efficient. Increasing advancements in terms of sterility testing technologies is expected to support the sterility testing market growth over the forecast period (2016-2024). Various organizations are laying efforts in making the testing process more time efficient. For instance, Milliflex Rapid System, launched in 2010, for making the sterility testing procedure more time efficient without hindering the test result quality. The following decade is expected to provide major growth traction to players involved in the sterility testing market.
Request a sample copy of this report@
https://www.coherentmarketinsights.com/insight/request-sample/174
Sterility Testing Market Taxonomy:
By Product Type
Kits and Reagents
Instruments
Service
By Test Type
Membrane Filtration
Direct Inoculation
Container closure integrity testing
Antimicrobial effectiveness testing
Rapid micro test
ATP bioluminescence
Fluorescent labeling
Electrical resistance
By Application
Pharmaceutical and Biological Manufacturing
Medical Devices Manufacturing
Bioburden Test
Others Applications
Novel Technology in the pharmaceutical testing market is expected to favor the growth in sterility testing market:
Conventionally used microbiological testing methods for detection, enumeration, sterilization and identification tend to use culture methods majorly and were so often time consuming and labor-intensive. The drawback of such procedures in practical considerations often limits the extent to which microbiological tests applied regularly in both formulation development stage and for microbiological quality assurance. Furthermore, there are various well latest and upcoming methods such as MF sterility test apparatus from Merk Millipore, which is aseptically filter liquid samples for sterility testing apparatus used in sterility testing and may find wide application in the pharmaceutical sector. This is expected to be one of the important factors supporting growth of the sterility testing market. Rapid testing methods such as ATP bioluminescence, fluorescent labeling, nucleic acid probes and electrical resistance are commonly used in pharmaceutical testing. Obtaining perfect results by incorporating single method for all requirements is challenging, as products vary in their specifications, which is need to be adapted by the specific demands of the pharmaceutical and medical device situation. However, it is satisfactory to have the developments in ATP bioluminescence offering with increased sensitivity under adenylate kinase amplification potential specificity, phage lysis and internal calibration against the effects of excipients, laser scanning cytometry can be benefited through more specificity in proposed development using fluorescently labeled antibodies.
Major players in the sterility testing market are engaged in research and development process. For instance, bioMérieux SA – a biopharmaceutical company engaged in world class products for in-vitro testing – launched The BacT/ALERT 3D Dual-T apparatus for rapid and easy product control with non-destructive, dual-temperature microbial detection system.
Presence of major players in parenteral product manufacturing is projected to favor growth of the sterility testing market
The global sterility testing market is highly competitive and fragmented. Major players in the market are developing novel products through major spending on research and development to get a competitive edge over their counterparts. For instance, due to the strong product pipeline under market segment, Merck Millipore and bioMérieux are considered to be the toughest competitor in the market. Key players operating in the sterility testing market include Merck Millipore, Thermo Fisher Scientific, Inc., bioMérieux SA, SGS S.A., Sartorius AG, WuXi AppTec, and Charles River Laboratories International, Inc.
Get More Details about this Report@
https://www.coherentmarketinsights.com/ongoing-insight/sterility-testing-market-174
About Coherent Market Insights:
Coherent Market Insights is a prominent market research and consulting firm offering action-ready syndicated research reports, custom market analysis, consulting services, and competitive analysis through various recommendations related to emerging market trends, technologies, and potential absolute dollar opportunity.
Contact Us:
Mr. Shah
Coherent Market Insights
1001 4th Ave,
#3200
Seattle, WA 98154
Tel: +1-206-701-6702
Email: sales@coherentmarketinsights.com
Visit Coherent Market Insights Blog @ https://blog.coherentmarketinsights.com
Connect with us on LinkedIn @ https://www.linkedin.com/company/coherent-market-insights
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Sterility Testing Market here
News-ID: 599651 • Views: …
More Releases from Biotechnology Industry Report By CMI

The New Research Trial Drives The Shigella Vaccines Market
Shigella is a gram-negative pathogenic enterobacteria that causes severe diarrhea and dysentery in humans. Symptoms associated with Shigella infection includes fever, stomach pain, tenesmus, watery diarrhea, vomiting, dehydration, and convulsions. Various strains of Shigella are encompassed, such as S. dysenteriae, S. flexneri, S. sonnei and S. boydii. Each species of Shigella has different serotypes classified on the basis of the structure of O-antigens repeats that are the polysaccharides moiety of the lipopolysaccharide, a virulence…
Flow Cytometry Market to Surpass US$ 7.64 Billion Threshold by 2025
Flow Cytometry – It’s not just for immunologist anymore
Flow cytometry is a widely used method for analyzing the expression of cell surface and intracellular molecules. It can also characterize and define different cell types in a heterogeneous cell population. Moreover, flow cytometry also allows simultaneous multi-parameter analysis of single cells. The growth in flow cytometry market is mainly driven by the combination of technological improvements, growing research activities in life…
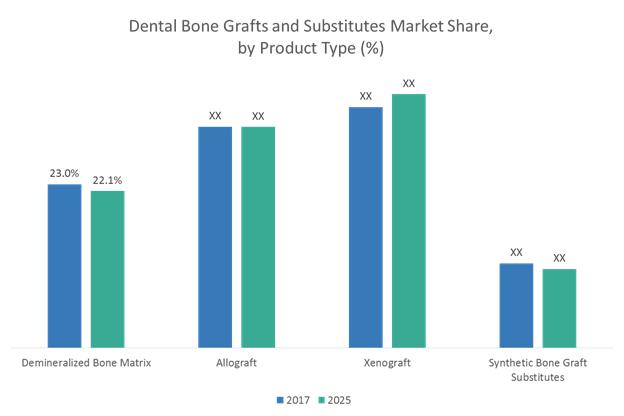
Dental Bone Graft and Substitutes Market to Surpass US$ 911.4 Million Threshold …
Dental Bone Graft and Substitutes Market Insights
Dental bone grafts are used as fillers or scaffolds that facilitate bone formation and helps in the wound healing and are suitable for a variety of clinical procedures such as filling of sockets, preservation of ridge volume (also referred to as ridge preservation), and osteogenesis i.e., the formation of new bones. Dental bone grafts are bioresorbable and are not reactive to antigen-antibody. Bone grafts…

Dental Bone Graft and Substitutes Market to Surpass US$ 911.4 Million Threshold …
Dental Bone Graft and Substitutes Market Insights
Dental bone grafts are used as fillers or scaffolds that facilitate bone formation and helps in the wound healing and are suitable for a variety of clinical procedures such as filling of sockets, preservation of ridge volume (also referred to as ridge preservation), and osteogenesis i.e., the formation of new bones. Dental bone grafts are bioresorbable and are not reactive to antigen-antibody. Bone grafts…
More Releases for Coherent
Comprehensive Soft Decision Forward Error Correction Coherent Digital Signal Pro …
Use code ONLINE20 to get 20% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
Soft Decision Forward Error Correction Coherent Digital Signal Processor (SD-FEC Coherent-DSP) Market Size Valuation Forecast: What Will the Market Be Worth by 2025?
The market size of the soft decision forward error correction coherent digital signal processor (SD-FEC coherent-DSP) has witnessed substantial growth recently. From 2024 to 2025,…
Surge In Air Traffic Boosts Demand For Advanced Coherent Radar Systems: Pivotal …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
Coherent Radar Market Size Growth Forecast: What to Expect by 2025?
In recent times, the coherent radar market has demonstrated potent growth. It is anticipated to expand from $8.18 billion in 2024 to $8.97 billion in 2025, boasting a compound annual growth rate (CAGR) of 9.8%. The historic growth…
Coherent Radar Market Analysis and Opportunities 2024
"The Business Research Company recently released a comprehensive report on the Global Coherent Radar Market Size and Trends Analysis with Forecast 2024-2033. This latest market research report offers a wealth of valuable insights and data, including global market size, regional shares, and competitor market share. Additionally, it covers current trends, future opportunities, and essential data for success in the industry.
According to The Business Research Company's, The coherent radar market size…
Immobilized Trypsin Market Report 2021 - Coherent Market Insights
Immobilized Trypsin Market is created in this intelligence report. This comprehensive research report is a well-thought-out compilation of detailed market development and growth factors optimizing the path of continued growth in terms of exact product data, strategies, and market share of the leading companies in this particular market. We follow an iterative model of research methodology to formulate the report that helps decision-makers make an informed investment appraisal. Literature research…
Bioinformatics Market Outlook Till, 2027 : Coherent Market Insights
The Bioinformatics Market report gives a weighty source to assess the market and other fundamental technicalities identifying with it. The examination unveils the total assessment and veritable parts of the Bioinformatics Market. The research analysts provide an elaborate description of the value chain and its distributor analysis. This Market study provides comprehensive data which enhances the understanding, scope and application of this report.The trends and expected prospects for the market…
AI App Development || Coherent Lab LLP
The IT industry is growing day by day and immense popularity. Nowadays, businesses are in search of better opportunities to make money in more innovative way. Considering the AI (artificial intelligence) in mobile and web app development, we at Coherent Lab LLP focuses on developing practical solutions which streamlines to generate more and more profits and smoothen the operations as well.
Situated in the heart of pinkcity, Jaipur, Coherent Lab…
