Press release
Global Antithrombin III Deficiency Testing Market
The Antithrombin III Deficiency Testing Market report provides in-depth analysis of parent market trends, macro-economic indicators and governing factors along with market attractiveness as per segments. The report also maps the qualitative impact of various market factors on market segments and geographies.Antithrombin deficiencies are mainly divided into two types: type I and type II. The type I antithrombin deficiency is characterized by an inadequate amount of normal antithrombin present in the bloodstream. In this condition, there is simply not enough antithrombin present to inactivate the coagulation factors. The type II antithrombin deficiency is characterized by adequate amount of antithrombin present in the bloodstream. However, it does not function properly and is unable to carry out its normal functions.
A sample of this report is available upon request @ www.persistencemarketresearch.com/samples/4272
Antithrombin III is a non-vitamin, K-dependent protease that naturally blocks abnormal blood clot formation in the bloodstream. Deficiency of antithrombin III causes various blood clot disorders in the human body. For instance, congenital antithrombin III deficiency is a common antithrombin deficiency, which usually occurs when individuals receive the abnormal antithrombin III gene from their parents. This abnormal gene causes abnormal blood clots in the human body which block blood flow and damage organs.
The major symptoms of blood clotting include fainting, coughing up blood, shortness of breath, pain when taking deep breaths, and swelling of legs. In addition, some physical symptoms of blood clots are abnormal lung sounds, fast breathing, swollen foot or leg, and fast heart rate.
The global antithrombin III deficiency testing market is categorized on the basis of various types of deficiencies, diagnostic procedures, and end users. Based on the type of deficiencies, the report covers type I antithrombin deficiency and type II antithrombin deficiency. Based on the type of technology, the report covers monoclonal antibodies, molecular diagnostics, differential light scattering, immunoassays, flow cytometry, chromatography, gel micro droplets, diagnostic imaging, and others. Based on end users, the report covers commercial or private labs, physician offices, hospitals, public health labs, and academic research institutes.
In terms of geography, North America dominates the global antithrombin III deficiency testing market. This is due to increased government funding on healthcare infrastructure in the region. In addition, technological advancement in antithrombin III diagnostic procedures has also propelled the market in North America. The U.S. represents the largest market for antithrombin deficiency testing in North America, followed by Canada. In Europe, Germany, France, and the U.K. hold the major shares of the antithrombin III deficiency testing market. The antithrombin III deficiency testing market in Asia is also expected to show high growth rates in the next five years. This is due to the various initiatives undertaken by governments to increase awareness about blood clotting disorders in the region. In addition, increased investments by many foreign diagnostic instrument companies have also propelled the growth of the antithrombin III deficiency testing market in the region. India, China, and Japan are expected to be the fastest-growing markets for antithrombin III deficiency testing in Asia.
Advanced applications of antithrombin deficiency diagnostic instruments and increasing prevalence of blood clot disorders are the key drivers for the global antithrombin III deficiency testing market. Also, increased government support in the form of funding and rapid technological advancements have fueled the growth of this market.
An increasing number of mergers and acquisitions involving diagnostic consumables manufacturing companies and rapid product launches are the key trends in the global antithrombin III deficiency testing market.
To view TOC of this report is available upon request @ www.persistencemarketresearch.com/toc/4272
The major companies operating in this market are Abbott Laboratories, Avocet Medical Inc., Axis-Shield plc, Beckman Coulter Inc., Becton, Dickinson and Company, Trinity Biotech PLC, Thermo Fisher Scientific, Inc., Siemens AG, Roche, Chrono-log Corporation, Sysmex Corporation, Hyphen Biomed and American Diagnostica GmbH.
About Us
Persistence Market Research (PMR) is a third-platform research firm. Our research model is a unique collaboration of data analytics and market research methodology to help businesses achieve optimal performance.
To support companies in overcoming complex business challenges, we follow a multi-disciplinary approach. At PMR, we unite various data streams from multi-dimensional sources. By deploying real-time data collection, big data, and customer experience analytics, we deliver business intelligence for organizations of all sizes.
Contact Us
Persistence Market Research
305 Broadway
7th Floor, New York City,
NY 10007, United States,
USA - Canada Toll Free: 800-961-0353
Email: sales@persistencemarketresearch.com
Web: http://www.persistencemarketresearch.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Global Antithrombin III Deficiency Testing Market here
News-ID: 484096 • Views: …
More Releases from Persistence Market Research Pvt. Ltd

Innovative Rehabilitation Technologies Drive Growth in Stroke Rehabilitation Mar …
Stroke Rehabilitation Market Size
The global stroke rehabilitation market size was valued at US$ 266.4 million in 2021 and is expected to grow at a CAGR of 9% from 2022 to 2032. Revenue generated from the sales of stroke rehabilitation solutions will surge to US$ 679.6 Million by the end of 2032.
The growth of the market is being driven by the increasing incidence of strokes, the growing demand for innovative rehabilitation…
Launch of New Laser-Based Devices by Key Players Fueling Growth of Global Aesthe …
Market Study on Aesthetic Lasers
The aesthetic lasers market reached US$2.8 billion in global revenue in 2021, and it will continue to expand quickly, with a value CAGR of 6.4%, to reach US$5.5 billion by the end of 2032. The growth of the market is being driven by the increasing demand for minimally invasive cosmetic procedures, rising disposable income, technological advancements, and growing medical tourism.
Stay Ahead with Market Intelligence Get Free…

Predictive Insights for Precision Medicine: The Global Biosimulation Market is S …
Biosimulation Market Research
The Global Biosimulation Market Size was valued at USD 2.9 billion in 2022 and is expected to reach USD 10.0 billion by 2030, registering a CAGR of 16.9% during the forecast period. The growth of the biosimulation market is being driven by the increasing use of biosimulation in a variety of applications, such as drug discovery, clinical trials, and toxicity prediction.
Market Overview
The biosimulation market has experienced rapid growth…
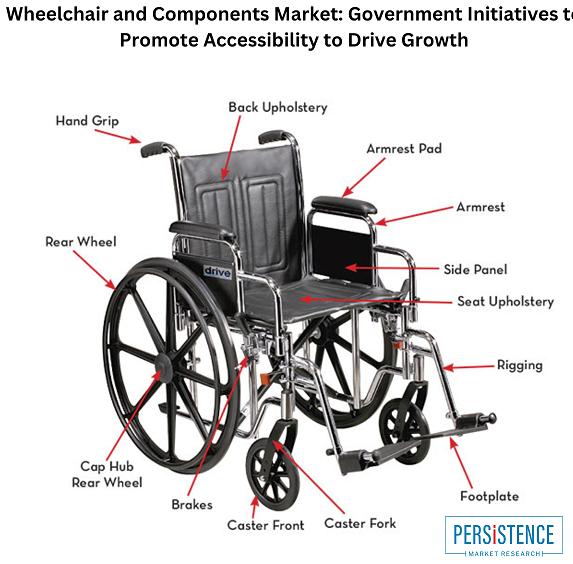
Wheelchair and Components Market: Growing Demand for Lightweight and Smart Wheel …
Wheelchair and Components Market
The global wheelchair and components market is projected to reach USD 17.9 billion by 2033, at a CAGR of 3.7% from 2022-2033. The growth of the market is driven by the increasing prevalence of disabilities, the rising geriatric population, and technological advancements in wheelchair design and manufacturing.
Click Here to Get Free Sample Copy of this Report@ https://www.persistencemarketresearch.com/samples/12961
The global wheelchair and components market has witnessed significant growth in…
More Releases for III
Rubber Knowledge III: Styrene-Butadiene Rubber (SBR)
Styrene-Butadiene Rubber (SBR for short) is a synthetic rubber formed by the copolymerization of styrene and butadiene. It is one of the most widely used synthetic rubbers globally, accounting for over 50% of the total synthetic rubber production. Its properties lie between those of natural rubber (NR) and butadiene rubber (BR), and it is renowned for its excellent wear resistance, low cost, and high stability.
Nature
Feature
Comparison with natural rubber (NR)
Abrasion resistance
Better…
Ytterbium(III) Sulfate Market Size and Forecast
𝐔𝐒𝐀, 𝐍𝐞𝐰 𝐉𝐞𝐫𝐬𝐞𝐲- The global Ytterbium(III) Sulfate Market is expected to record a CAGR of XX.X% from 2024 to 2031 In 2024, the market size is projected to reach a valuation of USD XX.X Billion. By 2031 the valuation is anticipated to reach USD XX.X Billion.
Ytterbium(III) Sulfate Market Size And Scope
The Ytterbium(III) Sulfate market is projected to grow significantly, with a market size valued at approximately USD 110.3 million in…
What is Ruthenium III Chloride used for?
Ruthenium(III) chloride hydrate, also known as ruthenium trichloride hydrate, is a compound of great significance in various fields. This compound consists of ruthenium, chlorine and water molecules. With its unique properties, ruthenium(III) chloride hydrate has a wide range of applications in different industries. In this article, we explore the uses of ruthenium(III) chloride and emphasize its importance.
Ruthenium(III) chloride hydrate is widely used as a catalyst in organic synthesis. It can…
Mucopolysaccharidosis III (MPS III) (Sanfilippo Syndrome) Therapeutics - Pipelin …
Mucopolysaccharidosis type III (MPS III), also known as Sanfilippo syndrome, is a progressive disorder that primarily affects brain and spinal cord (central nervous system). People with MPS III generally do not display features at birth, but they begin to show signs and symptoms of this disorder during early childhood. Affected children often initially have delayed speech and behaviour problems.
Download the sample report @ https://www.pharmaproff.com/request-sample/1146
In later stages of this disorder, people…
EXCEEMO Industrial SATA III SSD
Extremely impressive performance, the highest reliability and guaranteed fixed BOM - all in one service package for industrial customers
EXCEEMO, the memory and storage specialist from Landsberg am Lech, presents the industrial 2.5“ SATA III Solid-State-Drives with ultra-fast read/write transfer speed of up to 555MB/515MB per second and proven long-term reliability. The noiseless, energy-efficient and particularly robust solid state drives are available both in standard grade (0°/+70°C-MLC/SLC) and in industrial…
NewBlueFX Introduces “Video Essentials III” Effects Plugins
NewBlueFX Introduces “Video Essentials III” Effects Plugins
The Video Editor’s Wish List of Tools & Techniques in One Package
Video, Multimedia and Technology Editors
LA JOLLA, CA (November 04, 2009)—
NewBlue, Inc. today launched Video Essentials III, the third and latest offering in its best-selling Video Essentials series of video effects and transitions plugins. NewBlue Video Essentials III provides highly-practical video tools designed to solve everyday production issues. The collection includes Rolling Shutter,…
