Press release
Impact of Existing and Emerging Insulin Biosimilars Market Trends 2016-2026
A new race has been initiated between biosimilar and branded drug manufacturers to gain maximum revenue share from insulin market. The upcoming three years are very crucial for insulin manufacturers, as most of the branded insulin are going to be off patent which would results into the loss of around US$53 Bn revenue for branded manufacturers in 2016. Generic manufacturers are expected to take smaller chunk of this amount owing to their local presence which would restrain them take share from other markets. In such dynamic situation, the original drug manufacturers are seeking for plan B to sustain their hold in the market. As a result, these companies are now integrated with biosimilar manufacturers through agreement, alliance. Moreover, legal troubles pertaining patent infringement is hampering the development insulin Biosimilars. In order to overcome such infringement issues, the companies are now collaborating with each other to develop biosimilars versions of insulin. Recent example of global collaboration agreement between Mylan N.V. and Momenta Pharmaceuticals, Inc. has provided a competitive advantage to Mylan by increasing the number of insulin biosimlars under development. This collaboration will capitalized Mylan’s existing portfolio of biologics and insulin analog development.Insulin Biosimilars Market: Drivers and Restraints
Increasing prevalence of Type I diabetes, higher cost of existing insulin drugs are expected to drive growth of insulin biosimilars market. Government authorities are also focusing on the approval of insulin biosimilars owing to substantial financial burden in terms of reimbursements. Recently, the U.S. FDA has approved new insulin glargine Basaglar, for type 1 and type 2 diabetes which is Biosimilar version of Sanofi’s basal insulin Lantus (insulin glargine). Additionally, Lilly and Boehringer Ingelheim's biosimilar insulin glargine has got approval through European Medicines Agency's (EMA's) Biosimilar pathway. Such ongoing approvals by the respective authorities are expected to drive the growth of insulin Biosimilar market. However, Insulin patent protection rights and strong retaliation from the branded manufactures has restricted the growth of insulin biosimilar development.
Request For Report Sample@ http://www.futuremarketinsights.com/reports/sample/rep-gb-1883
Insulin Biosimilars Market: Segmentation
The global market for insulin Biosimilar is segmented on the basis of disease indication and geography.
On the basis of disease indication, the global insulin Biosimilar market is segmented as,
Type I Diabetes
Type II Diabetes
Insulin Biosimilars Market: Overview
Development of clinically proven insulin Biosimilar will pave the road for new entrants. The upcoming patent expiration of blockbuster insulin drugs will open a new avenue and unprecedented opportunities for biosimilar development and manufacturing companies. The cost of insulin biosimilar is currently high owing to stringent adherence requirements in terms of complex regulations and manufacturing and operations. Prices of biosimilar drugs are expected decreased by an average of 20% as compared to branded drugs.
Insulin Biosimilars Market: Region-wise Outlook
Geographically, the global insulin biosimilar market is segmented into regions namely, North America, Latin America, Western Europe, Eastern Europe, Asia-Pacific Excluding Japan, Japan, Middle East and Africa. Among all the regions, North America will continue to lead the global market for insulin biosimilars market due to high demand for insulin. Europe insulin market is expected to account for second largest share in global market primarily due to early approval for biosimilar which is expected to fuel the market growth. Asian companies are competing on the basis of price and has patient pool suffering diabetes. Hence, the Asia Pacific insulin biosimilar market is expected to witness fastest growth in overall market over the forecast period.
Visit For TOC@ http://www.futuremarketinsights.com/toc/rep-gb-1883
Insulin Biosimilars Market: Key Players
Key players operating in the global insulin biosimilar market are Eli Lilly & Co., Boehringer Ingelheim, Merck & Co., Pfizer Inc., Biocon, Mylan N.V. and others
ABOUT US:
Future Market Insights (FMI) is a leading market intelligence and consulting firm. We deliver syndicated research reports, custom research reports and consulting services, which are personalized in nature. FMI delivers a complete packaged solution, which combines current market intelligence, statistical anecdotes, technology inputs, valuable growth insights, an aerial view of the competitive framework, and future market trends.
CONTACT:
Future Market Insights
616 Corporate Way, Suite 2-9018,
Valley Cottage, NY 10989,
United States
T: +1-347-918-3531
F: +1-845-579-5705
Email: sales@futuremarketinsights.com
Website: www.futuremarketinsights.com
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Impact of Existing and Emerging Insulin Biosimilars Market Trends 2016-2026 here
News-ID: 475357 • Views: …
More Releases from Future Market Insights
Global Pallet Pooling Market Outlook 2025-2035: Growth Accelerates on Sustainabl …
Global Pallet Pooling Market: Growth Outlook and Strategic Context
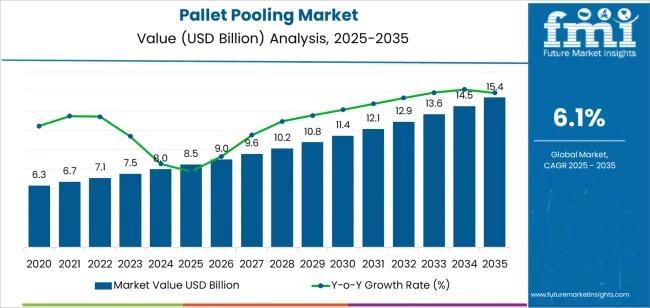
The global Pallet Pooling Market is witnessing sustained expansion, supported by rising global trade volumes, growing emphasis on supply chain efficiency, and increasing adoption of sustainable logistics solutions. Valued at USD 8.5 billion in 2025, the market is forecast to reach USD 15.4 billion by 2035, registering a CAGR of 6.1% over the forecast period.
The transition from pallet ownership to rental-based pooling…
Global Industrial Safety Products Market to Reach USD 36.9 Billion by 2036 with …
Industrial Safety Products Market Growth to 2036
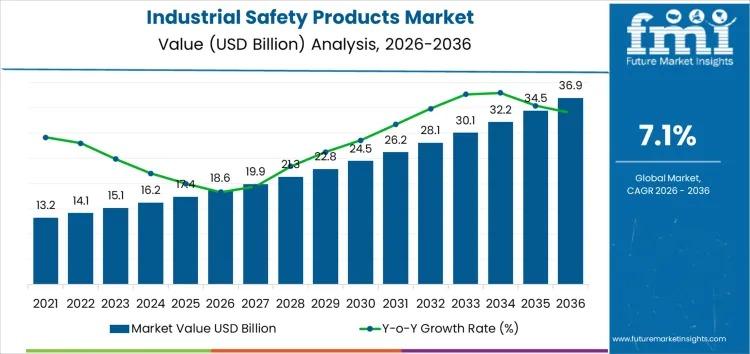
The Industrial Safety Products Market is poised for strong growth, expanding from USD 18.6 billion in 2026 to USD 36.9 billion by 2036, reflecting a CAGR of 7.1%. This growth is driven by increasing regulatory requirements across manufacturing, oil & gas, construction, healthcare, and logistics industries, emphasizing worker protection, compliance, and risk mitigation.
Subscribe for Year-Round Insights → Stay ahead with quarterly and annual data…
Spectrum Monitoring and Interference Analysis Systems Market Growth Outlook 2026 …
Market Overview: Growth Fueled by Wireless Expansion
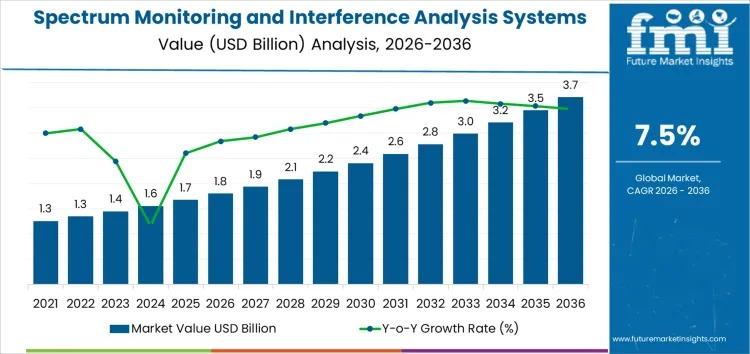
The Spectrum Monitoring and Interference Analysis Systems Market is poised for significant growth, projected to expand from USD 1.8 billion in 2026 to USD 3.7 billion by 2036 at a CAGR of 7.5%. This surge is driven by rising 5G adoption, proliferation of IoT devices, and increasingly complex radio frequency (RF) environments, which demand robust interference detection and analysis capabilities.
Subscribe for Year-Round Insights →…
Dynamic Measurement System Market Growth Outlook 2026-2036 Driven by Real-Time D …
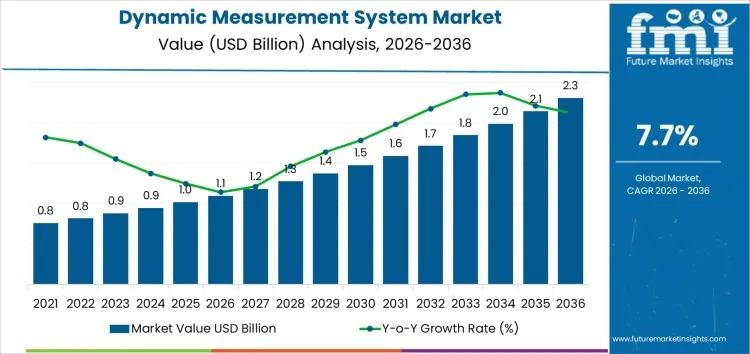
The global Dynamic Measurement System Market is witnessing steady expansion as industries intensify their reliance on real-time, high-precision data for performance monitoring and safety assurance. Valued at USD 1.1 billion in 2026, the market is projected to reach USD 2.3 billion by 2036, growing at a CAGR of 7.70%. This growth is strongly linked to rising adoption across automotive, aerospace, energy, manufacturing, and research sectors, where time-sensitive data plays a…
More Releases for Biosimilar
Interchangeable Biosimilar Humira Market Share Driven by Biologic Therapy Adopti …
Interchangeable Biosimilar Humira Market
The global market for Interchangeable Biosimilar Humira was valued at US$ million in the year 2024 and is projected to reach a revised size of US$ million by 2031, growing at a CAGR of %during the forecast period
View sample report
https://reports.valuates.com/request/sample/QYRE-Auto-33I15005/Global_Interchangeable_Biosimilar_Humira_Market_Research_Report_2023
The Interchangeable Biosimilar Humira Market is experiencing significant market growth as healthcare providers and patients increasingly adopt biosimilar therapies for autoimmune and inflammatory conditions. Market trends indicate rising…
Key Trend Reshaping the Biosimilar Monoclonal Antibodies Market in 2025: Advance …
What Are the Projections for the Size and Growth Rate of the Biosimilar Monoclonal Antibodies Market?
In recent times, the biosimilar monoclonal antibodies sector has experienced a swift expansion. The market size, which stands at $8.04 billion in 2024, is projected to climb to $9.25 billion in 2025, marking a compound annual growth rate (CAGR) of 15.1%. Factors such as expired patents, an increased understanding of biosimilars, governmental strategies, heightened financial…
Key Trend Reshaping the Biosimilar Monoclonal Antibodies Market in 2025: Advance …
What Are the Projections for the Size and Growth Rate of the Biosimilar Monoclonal Antibodies Market?
In recent times, the biosimilar monoclonal antibodies sector has experienced a swift expansion. The market size, which stands at $8.04 billion in 2024, is projected to climb to $9.25 billion in 2025, marking a compound annual growth rate (CAGR) of 15.1%. Factors such as expired patents, an increased understanding of biosimilars, governmental strategies, heightened financial…
Biosimilar Market Treating More for Less: The Booming Infliximab Biosimilar Mark …
Infliximab Biosimilar Market worth $ XX Million by 2030 - Exclusive Report by InsightAce Analytic
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the "Global Infliximab Biosimilar Market- by Application (Crohn's Disease, Psoriatic Arthritis, Rheumatoid Arthritis, Ulcerative Colitis, Ankylosing Spondylitis, Plaque Psoriasis and Others), End User (Hospital Pharmacy, Retail Pharmacy, Online Pharmacy and Other Direct Distribution Channels), Trends, Industry Competition Analysis, Revenue and Forecast To 2030."
Get…
Biosimilar Monoclonal Antibodies Market
InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the " "Global Biosimilar Monoclonal Antibodies Market by Product (infliximab, trastuzumab, rituximab, adalimumab, bevacizumab, cetuximab, ranibizumab, denosumab, eculizumab, and other pipeline products), Indication (oncology, inflammatory & autoimmune disorders, chronic diseases, blood disorders, and other indications), Clinical Trial/Pipeline Analysis, Future Trends, Industry Competition Analysis, Revenue and Forecast To 2031."
The Biosimilar Monoclonal Antibodies Market Size is valued at 5.02…
Infliximab Biosimilar Insight, 2022 | DelveInsight
DelveInsight's, "Infliximab Biosimilar Insight, 2022" report provides comprehensive insights about 35+ companies and 45+ marketed and pipeline drugs in Infliximab Biosimilars landscape. It covers the marketed and pipeline drug profiles, including clinical and nonclinical stage products. It also covers the therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Interested to know more about the functioning of…
