Press release
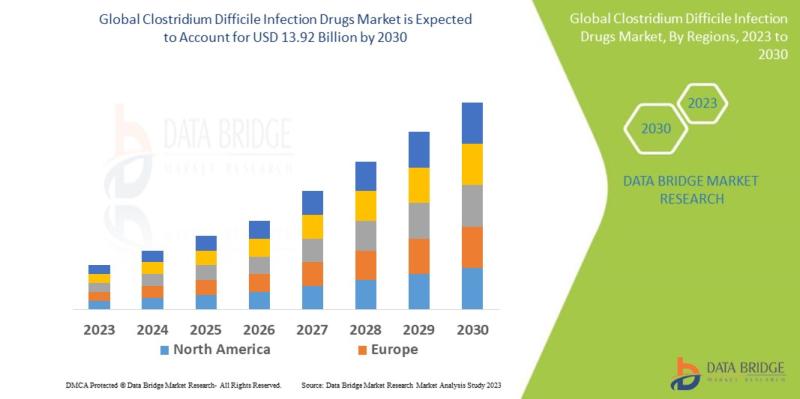
Clostridium Difficile Infection Drugs Market Size, Share & Forecast 2026-2030 | Expected to Reach USD 13.92 Billion by 2030 at a CAGR of 7.0%
As per Data Bridge Market Research analysis, the Clostridium difficile infection drugs market was estimated at USD 9.93 billion in 2025. The market is expected to grow from USD 10.62 billion in 2026 to USD 13.92 billion in 2030, at a CAGR of 7.0% during the forecast period with driven by the rising demand for advanced anti-infective therapies, increasing incidence of hospital-acquired infections, growing adoption of microbiome-based therapeutics, and expanding healthcare investments in infectious disease management.Get the full PDF sample copy of the report: (Includes full table of contents, list of tables and figures, and graphs): https://www.databridgemarketresearch.com/request-a-sample/?dbmr=global-necrobiosis-lipoidica-market
Market Size & Forecast
2025 Market Size: USD 9.93 Billion
2026 Projected Market Size: USD 10.62 Billion
2030 Projected Market Size: USD 13.92 Billion
CAGR (2026-2030): 7.0%
Largest Region: North America
Fastest Growing Region: Asia-Pacific
Key Market Report Takeaways
North America accounted for the largest market share, contributing approximately 39% of global revenue due to advanced healthcare infrastructure and high diagnosis rates.
Asia-Pacific is projected to register the fastest CAGR through 2030 owing to expanding healthcare expenditure and rising infection awareness.
Antibiotic therapeutics segment holds the highest market share due to widespread use of fidaxomicin and vancomycin-based therapies.
Hospital-acquired infection treatment remains the dominant application segment driven by increasing inpatient CDI incidence.
Hospitals and specialty clinics represent the leading end-use segment due to higher patient admissions and antimicrobial treatment volumes.
Microbiome restoration therapies are gaining commercial traction in recurrent infection management.
Increasing regulatory approvals for live biotherapeutic products are reshaping treatment standards globally.
Key Market Trends & Highlights
North America continues to dominate the global market due to strong reimbursement systems, higher healthcare-associated infection prevalence, and rapid adoption of advanced CDI therapeutics.
Asia-Pacific is the fastest-growing regional market, supported by healthcare modernization, improving diagnostic capabilities, and increasing infectious disease surveillance programs.
Antibiotics remain the dominant therapeutic segment; however, microbiome-based therapeutics are witnessing accelerated adoption in recurrent infection treatment.
Rising elderly population, growing antibiotic resistance concerns, and increasing hospitalization rates are major long-term growth drivers.
Emerging microbiome therapeutics, precision biologics, and fecal microbiota-based therapies are transforming recurrent CDI management.
Strategic collaborations, FDA approvals, pipeline expansion, and investments in infectious disease therapeutics are intensifying global competition.
Details about the report and current availability can be viewed: https://www.databridgemarketresearch.com/reports/global-necrobiosis-lipoidica-market
Market Dynamics
Market Drivers
Rising Incidence of Hospital-Acquired Infections
The increasing prevalence of hospital-acquired and antibiotic-associated infections is significantly driving demand for CDI therapeutics globally. Aging populations, prolonged hospitalization, and higher antibiotic consumption continue to elevate infection risks. North America and Europe report high CDI burden due to extensive inpatient care utilization. Healthcare systems are prioritizing infection prevention and recurrence reduction strategies, supporting market expansion.
Growing Adoption of Microbiome-Based Therapeutics
The emergence of microbiome restoration therapies is reshaping recurrent CDI treatment approaches. Regulatory approvals for advanced microbiota-based therapeutics have improved treatment outcomes and reduced recurrence rates. Pharmaceutical companies are increasing investments in microbiome research and live biotherapeutic products. The U.S. market is witnessing particularly strong adoption due to favorable regulatory pathways and clinical validation.
Increasing Antimicrobial Resistance Concerns
Escalating antimicrobial resistance is driving demand for next-generation CDI therapeutics with targeted mechanisms of action. Conventional broad-spectrum antibiotics have increased recurrence risks, encouraging the adoption of specialized therapies. Healthcare providers are increasingly shifting toward narrow-spectrum and recurrence-prevention treatments. This trend is accelerating R&D investment across North America, Europe, and Asia-Pacific.
Expansion of Healthcare Infrastructure in Emerging Economies
Rapid healthcare infrastructure development in Asia-Pacific and Latin America is improving infectious disease diagnosis and treatment access. Governments are increasing healthcare spending, hospital capacity, and diagnostic laboratory capabilities. Growing awareness regarding gastrointestinal infections is further supporting early diagnosis rates. Expanding healthcare coverage is expected to strengthen long-term market penetration.
Regulatory Support and Clinical Advancements
Regulatory agencies are increasingly supporting innovative anti-infective and microbiome therapeutics through expedited approvals and orphan drug pathways. Clinical advancements in biologics and fecal microbiota therapies are improving treatment efficacy for recurrent CDI cases. Strong pipeline activity and late-stage clinical trials are encouraging market competition. Regulatory encouragement is particularly strong in the U.S. and Europe.
Market Restraints
High Cost of Advanced Therapies
Advanced microbiome-based therapeutics and biologic treatments involve significantly higher costs compared to conventional antibiotics. Treatment affordability remains a major concern in low- and middle-income countries. Reimbursement limitations and pricing pressures can restrict patient access to premium therapies. Cost-sensitive healthcare systems in emerging economies continue to rely heavily on generic antibiotics.
Complex Regulatory Approval Processes
Strict regulatory requirements for biologics, microbiome therapeutics, and live biotherapeutic products increase development timelines and commercialization costs. Compliance with manufacturing, safety, and clinical validation standards remains highly demanding. Regulatory uncertainty surrounding fecal microbiota therapies may slow market expansion in certain countries. Smaller biotechnology firms face greater barriers to entry.
Limited Awareness and Underdiagnosis
Underdiagnosis of CDI remains a significant issue in developing healthcare systems due to limited diagnostic infrastructure and low awareness. Misdiagnosis often delays treatment initiation and affects market penetration. In several emerging economies, gastrointestinal infections are frequently underreported. Lack of standardized testing protocols also limits therapeutic adoption rates.
Pricing Pressure from Generic Antibiotics
Generic vancomycin and metronidazole therapies continue to dominate cost-sensitive healthcare markets. Intense pricing competition restricts premium pricing opportunities for innovative products. Healthcare providers often prioritize lower-cost therapies despite recurrence concerns. This dynamic creates commercialization challenges for novel microbiome-based products.
Supply Chain and Manufacturing Constraints
Biopharmaceutical and microbiome-based therapies require highly controlled manufacturing and cold-chain logistics. Supply chain disruptions can affect product availability and treatment continuity. Dependence on specialized biological processing infrastructure increases operational complexity. Global logistics challenges and raw material shortages continue to impact pharmaceutical manufacturing efficiency.
Market Opportunities
Expansion of Microbiome Therapeutics
The growing clinical acceptance of microbiome restoration therapies presents substantial revenue opportunities for pharmaceutical and biotechnology companies. Increasing recurrence rates of CDI are encouraging adoption of advanced microbiota-based products. Ongoing clinical trials and pipeline innovations are expected to broaden therapeutic applications. North America remains the leading commercialization hub for microbiome therapeutics.
Untapped Growth Potential in Asia-Pacific
Asia-Pacific offers significant expansion opportunities due to rising healthcare expenditure, expanding hospital infrastructure, and increasing infectious disease awareness. Countries such as China, India, and Japan are strengthening diagnostic and pharmaceutical capabilities. Improving reimbursement systems and healthcare accessibility are expected to support long-term growth. International companies are increasingly targeting the region for expansion.
Strategic Partnerships and Licensing Agreements
Pharmaceutical companies are actively pursuing collaborations, licensing deals, and acquisitions to strengthen infectious disease portfolios. Partnerships between biotechnology firms and large pharmaceutical companies are accelerating commercialization timelines. Strategic alliances support R&D expansion, geographic penetration, and manufacturing scalability. Such collaborations are expected to intensify over the forecast period.
Increasing Investment in Precision Anti-Infectives
Growing focus on precision medicine and targeted anti-infective therapies is opening new innovation avenues. Advanced diagnostics and personalized treatment approaches are improving clinical outcomes. Investment in targeted biologics and recurrence-prevention therapies is increasing globally. This trend supports development of differentiated products with premium pricing potential.
Government Focus on Infection Prevention Programs
Governments and healthcare organizations are increasing investment in infection prevention, antimicrobial stewardship, and hospital safety programs. Rising policy emphasis on reducing healthcare-associated infections is strengthening therapeutic demand. Public health initiatives are improving disease surveillance and diagnostic rates. Regulatory support for innovative therapies further enhances market opportunities.
Market Challenges
Recurring Infection and Treatment Complexity
High recurrence rates remain a major challenge in CDI management despite therapeutic advancements. Patients often require multiple treatment cycles, increasing healthcare costs and clinical complexity. Recurrent infections reduce treatment effectiveness and patient adherence. Healthcare providers continue to seek more durable therapeutic solutions.
Fragmented Competitive Environment
The market includes established pharmaceutical companies, specialty biotech firms, and emerging microbiome therapy developers. Intense competition increases pricing pressure and commercialization challenges. Smaller players often face resource limitations in global expansion and regulatory compliance. Competitive fragmentation may impact profitability and market consolidation.
Limited Accessibility in Developing Regions
Healthcare inequality and inadequate diagnostic infrastructure limit treatment accessibility across several low-income markets. Rural healthcare systems frequently lack advanced laboratory testing and specialized treatment availability. Delayed diagnosis contributes to higher complication rates. These limitations restrict broader global market penetration.
Clinical and Regulatory Uncertainty
Microbiome-based therapeutics continue to face evolving regulatory frameworks and long-term safety evaluation requirements. Variability in clinical outcomes and standardization challenges can slow physician adoption. Regulatory inconsistencies across regions create commercialization barriers. Manufacturers must invest heavily in post-market surveillance and compliance activities.
Economic and Reimbursement Pressures
Healthcare cost containment measures and reimbursement limitations continue to impact market profitability. Public healthcare systems increasingly evaluate cost-effectiveness before approving premium therapies. Economic instability in developing regions may reduce pharmaceutical spending. Pricing negotiations and reimbursement delays can slow market adoption.
Market Segmentation & Analysis
Key Insights
Largest Region: North America
Fastest Growing Region: Asia-Pacific
Competitive Landscape
Market Structure Overview
The global CDI drugs market is moderately consolidated with the presence of multinational pharmaceutical companies, biotechnology firms, and emerging microbiome therapy developers. Competition is driven by innovation in anti-infective therapeutics, biologics, and microbiome restoration therapies. Companies are focusing on product differentiation, clinical efficacy, and recurrence prevention capabilities. Competitive landscape analysis helps evaluate strategic positioning, innovation strength, and geographic expansion strategies of major participants.
Key Industry Players
Leading market participants focus on expanding infectious disease portfolios, strengthening R&D pipelines, and commercializing advanced microbiome therapeutics. Global pharmaceutical companies maintain strong distribution networks and regulatory expertise, while biotechnology firms emphasize innovation in microbiome-based therapies. Market leaders continue to invest in clinical trials, strategic collaborations, and regional expansion initiatives.
List of Key Industry Players
Merck & Co.
Ferring Pharmaceuticals
Seres Therapeutics
Pfizer Inc.
ANI Pharmaceuticals
Vedanta Biosciences
Finch Therapeutics
MGB Biopharma
Competitive Strategies
Companies are increasingly focusing on microbiome therapeutics, targeted anti-infective drugs, and recurrence prevention technologies. Strategic partnerships and licensing agreements are accelerating commercialization and geographic expansion. Product launches, clinical trial advancements, and M&A activity continue to intensify market competition. Firms are leveraging innovation, data analytics, and advanced biologic platforms to improve treatment efficacy and patient outcomes.
Emerging Players & Market Dynamics
Emerging biotechnology firms are disrupting traditional treatment approaches through microbiome engineering and live biotherapeutic development. Startups are introducing specialized and cost-effective therapeutic alternatives targeting recurrent CDI. Increasing venture capital investment and pharmaceutical collaborations are supporting innovation pipelines. Growing digital health integration and precision medicine approaches are further reshaping market dynamics.
Latest Developments
January 2025 - Merck & Co. : The company announced the discontinuation of Zinplava for recurrent CDI prevention, potentially reshaping competitive dynamics in recurrence management therapies.
August 2024 - Nestlé Health Science: The company acquired global rights to VOWST from Seres Therapeutics to strengthen its microbiome therapeutics portfolio and expand commercialization capabilities globally.
May 2024 - Ferring Pharmaceuticals: New real-world clinical outcomes data for REBYOTA demonstrated effectiveness in recurrent CDI management, supporting broader physician confidence and market adoption.
May 2024 - Vedanta Biosciences: The company initiated Phase III RESTORATiVE303 clinical trials for VE303 targeting recurrent CDI, expanding competition within microbiome-based therapeutics.
May 2024 - Nestlé Health Science and Seres Therapeutics: The companies presented integrated efficacy and safety data for VOWST at DDW 2024, strengthening evidence supporting microbiota-based recurrence prevention therapies.
January 2024 - Seres Therapeutics: The company reported increasing commercial adoption of VOWST following FDA approval, highlighting strong enrollment and patient uptake trends in the U.S. market.
2024 - Global Regulatory Authorities: Continued regulatory support for microbiome therapeutics and live biotherapeutic products accelerated clinical research and commercialization activity within infectious disease therapeutics.
Check out more related studies published by Data Bridge Market Research:
https://www.databridgemarketresearch.com/reports/global-cephalosporins-market
https://www.databridgemarketresearch.com/reports/global-head-lice-treatment-market
https://www.databridgemarketresearch.com/reports/global-ulcerative-colitis-market
https://www.databridgemarketresearch.com/reports/global-urinary-incontinence-market
https://www.databridgemarketresearch.com/reports/global-avian-influenza-vaccines-market
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email:- corporatesales@databridgemarketresearch.com
Data Bridge Market Research set forth itself as an unconventional and neoteric market research and consulting firm with an unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market. Data Bridge endeavors to provide appropriate solutions to the complex business challenges and initiates an effortless decision-making process. Data Bridge is an aftermath of sheer wisdom and experience which was formulated and framed in the year 2015 in Pune.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Clostridium Difficile Infection Drugs Market Size, Share & Forecast 2026-2030 | Expected to Reach USD 13.92 Billion by 2030 at a CAGR of 7.0% here
News-ID: 4513768 • Views: …
More Releases from Data Bridge Market Research
Clostridium Difficile Infection Drugs Market to Reach USD 13.92 Billion by 2030, …
As per Data Bridge Market Research analysis, the Clostridium Difficile Infection Drugs Market was estimated at USD 9.93 billion in 2025. The market is expected to grow from USD 10.62 billion in 2026 to USD 13.92 billion in 2030, at a CAGR of 7.0% during the forecast period with driven by the rising demand for advanced anti-infective therapies, increasing incidence of hospital-acquired infections, growing elderly population, and expanding investments in…
Smart Implantable Pumps Market Growth Analysis
As per Data Bridge Market Research analysis, the Smart Implantable Pumps Market was estimated at USD 3.62 billion in 2025. The market is expected to grow from USD 3.62 billion in 2025 to USD 6.59 billion by 2033, at a CAGR of 7.79% during the forecast period with driven by the rising demand for advanced drug delivery systems, increasing prevalence of chronic diseases, and growing adoption of minimally invasive treatment…
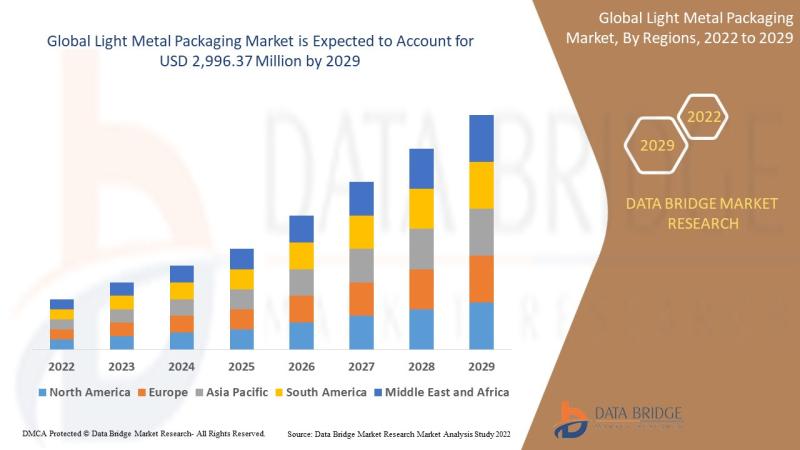
Light Metal Packaging Market Growth Analysis
As per Data Bridge Market Research analysis, the Light Metal Packaging Market was estimated at USD 2.35 billion in 2025. The market is expected to grow from USD 1,810.50 million in 2021 to USD 2,996.37 million by 2029, at a CAGR of 6.50% during the forecast period with driven by the rising demand for sustainable packaging solutions, increasing consumption of packaged food and beverages, growing emphasis on recyclability, and expanding…
Erythema Nodosum Market Size, Share, Growth Analysis, and Forecast Report, 2026- …
Market Summary
As per Data Bridge Market Research analysis, the erythema nodosum market was estimated at USD 45.01 billion in 2025. The market is expected to grow from USD 47.66 billion in 2026 to USD 59.93 billion in 2030, at a CAGR of 5.90% during the forecast period with driven by the rising demand for advanced inflammatory disease treatment solutions, increasing autoimmune disease prevalence, growing healthcare expenditure, and expanding investments in…
More Releases for CDI
1,1'-Carbonyldiimidazole (CDI) Market Size, Demand, Share & Future Growth Opport …
According to QYResearch New Study Report '1,1'-Carbonyldiimidazole (CDI) Market 2026' provides a comprehensive analysis of the industry with market insights will definitely facilitate to increase the knowledge and decision-making skills of the business, thus providing an immense opportunity for growth. Finally, this will increase the return rate and strengthen the competitive advantage within. Since it's a personalised market report, the services are catered to the particular difficulty. The correct methodology…
Clostridioides Difficile Infections (CDI) Market is expected to reach USD 4.1 bi …
Clostridioides difficile (C. difficile) infection (CDI) is one of the most prevalent healthcare-associated bacterial infections, responsible for significant morbidity and mortality worldwide. It occurs when the gut microbiota balance is disrupted-often after antibiotic exposure-allowing the toxin-producing C. difficile bacteria to proliferate and cause severe diarrhea and colitis.
Download Full PDF Sample Copy of Market Report @ https://exactitudeconsultancy.com/request-sample/73639
The rising incidence of antibiotic-associated diarrhea, coupled with hospital-acquired infections and recurrence rates of CDI,…
Clostridioides difficile Infection (CDI) Market 2025-2034 Business Outlook, Crit …
Introduction
Clostridioides difficile infection (CDI) is a leading cause of healthcare-associated infections worldwide, primarily affecting patients undergoing broad-spectrum antibiotic therapy. CDI leads to severe diarrhea, colitis, and potentially life-threatening complications, with high recurrence rates posing a persistent challenge.
The global CDI market is driven by the increasing incidence of hospital-acquired infections, rising antimicrobial resistance, and the urgent need for therapies that reduce recurrence rates. With advances in antibiotics, microbiome therapies, fecal microbiota…
CDI Of Monroe: Leading the Way in Comprehensive Wastewater Solutions for the Flo …
CDI Of Monroe combines decades of experience, a commitment to environmental stewardship, and cutting-edge technology to deliver expert service and reliability to clients in the Florida Keys.
Image: https://www.getnews.info/uploads/4b4127f92fb2ed399746ec83ca6a39ef.jpg
Providing essential services like wastewater management often presents significant challenges for small towns. Limited resources, aging infrastructure, and geographic constraints can all contribute to the complexities faced by these communities. Fortunately, CDI of Monroe [https://cdiofmonroe.com/] is tackling these challenges head-on and offering tailored…
Clinical Documentation Improvement (CDI) Market: Position Your Product To Outper …
Clinical Documentation Improvement (CDI) Market report will provide one with overall market analysis, statistics, various trends, drivers, opportunities, restraints, and every minute data relating the Clinical Documentation Improvement (CDI) market necessary for forecasting its revenue, factors propelling & growth. The Clinical Documentation Improvement (CDI) market study provides unique guidance in thoughtful details regarding the development factors and has used a top-down and bottom-up approach to keep it error-free and accurate.…
Carbodiimides (CDI)Market to Witness Robust Expansion by 2025
LP INFORMATION recently released a research report on the Carbodiimides (CDI) analysis, which studies the Carbodiimides (CDI)industry coverage, current market competitive status, and market outlook and forecast by 2025.
Global “Carbodiimides (CDI)Market 2020-2025” Research Report categorizes the global Carbodiimides (CDI)by key players, product type, applications and regions,etc. The report also covers the latest industry data, key players analysis, market share, growth rate, opportunities and trends, investment…