Press release
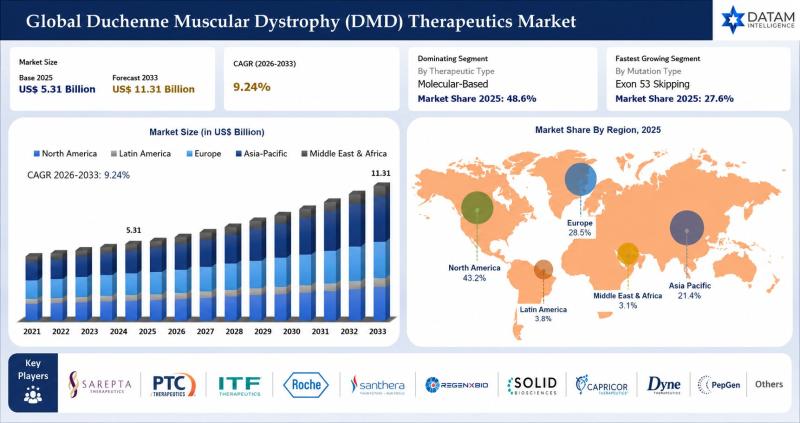
Duchenne Muscular Dystrophy Therapeutics Market to Reach USD 11.31 Billion by 2033 at 9.24% CAGR; North America Leads with 43.2% Share - Key Players: Sarepta Therapeutics, PTC Therapeutics, Roche
The global Duchenne Muscular Dystrophy (DMD) therapeutics market was valued at USD 5.31 billion in 2025 and is projected to reach nearly USD 11.31 billion by 2033, growing at a CAGR of 9.24% during the forecast period from 2026 to 2033. The market is witnessing strong growth driven by the increasing prevalence of Duchenne muscular dystrophy, rising awareness regarding rare genetic disorders, and growing demand for advanced disease modifying therapies. Expanding adoption of exon skipping therapies, corticosteroids, gene therapies, and mutation targeted treatments is significantly improving disease management and patient quality of life. In addition, increasing availability of early genetic screening programs and supportive healthcare infrastructure is further accelerating market expansion globally.The market is also benefiting from rapid advancements in gene editing technologies, increasing clinical trial activities, and rising investments in rare disease therapeutics research. Regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) continue to support the development of innovative DMD therapies through orphan drug designations, fast track approvals, and accelerated regulatory pathways. Leading companies including Sarepta Therapeutics, Pfizer Inc., Roche, and Solid Biosciences are actively focusing on gene therapies, exon skipping technologies, and strategic collaborations to strengthen their global presence in the Duchenne muscular dystrophy therapeutics market.
Get a Sample PDF Of This Report (Get Higher Priority for Corporate Email ID): https://www.datamintelligence.com/download-sample/global-dunche-muscular-dystrophy-therapeutics-market?sai-v
Key Developments
February 2026: Rising adoption of gene therapies, exon-skipping technologies, and RNA-based therapeutics accelerated innovation in the global Duchenne Muscular Dystrophy (DMD) Therapeutics market. Pharmaceutical companies expanded investments in advanced molecular therapies targeting disease progression and long-term muscle preservation.
January 2026: Increasing regulatory support for rare disease therapeutics and growing clinical trial activity strengthened development of next-generation Duchenne Muscular Dystrophy treatment solutions worldwide. Healthcare providers expanded utilization of precision medicine approaches and genetic diagnostics for early disease intervention.
December 2025: Growing advancements in cell therapies and exon-skipping therapeutics accelerated commercialization activities in the DMD therapeutics market globally. Capricor Therapeutics reported positive Phase 3 HOPE-3 trial results for deramiocel, marking a major advancement in cell therapy development for Duchenne Muscular Dystrophy treatment.
November 2025: Expansion of neuromuscular disease research programs and increasing investments in genetic medicine strengthened innovation in Duchenne Muscular Dystrophy therapeutics worldwide. Research institutions intensified focus on improving dystrophin restoration and long-term functional outcomes for DMD patients.
October 2025: Rising advancements in AI-enabled diagnostics, biomarker discovery, and wearable monitoring technologies improved disease tracking and personalized treatment management for Duchenne Muscular Dystrophy patients globally. Healthcare organizations increasingly adopted digital monitoring tools supporting real-time assessment of motor function and disease progression.
September 2025: Increasing collaborations between biotechnology companies, pharmaceutical firms, and research organizations accelerated development of innovative Duchenne Muscular Dystrophy therapies globally. Strategic partnerships focused on expanding gene therapy accessibility, improving manufacturing capabilities, and advancing late-stage clinical pipelines.
August 2025: Growing investments in rare disease healthcare infrastructure and pediatric neuromuscular treatment programs strengthened expansion of the DMD therapeutics market across North America, Europe, and Asia-Pacific. Governments and advocacy organizations increased focus on newborn screening initiatives and early diagnosis programs.
July 2025: Rising demand for disease-modifying therapies and long-term supportive care solutions accelerated innovation in Duchenne Muscular Dystrophy treatment pipelines worldwide. Companies expanded research activities involving CRISPR-based therapies, gene editing, and regenerative medicine platforms targeting improved patient mobility and survival outcomes.
June 2025: Increasing focus on improving quality of life, delaying disease progression, and expanding access to advanced therapeutics supported strong growth in the global Duchenne Muscular Dystrophy Therapeutics market. North America maintained market leadership due to strong rare disease research infrastructure, while Asia-Pacific emerged as a rapidly growing region driven by expanding healthcare investments and genetic testing adoption.
Key Players
Sarepta Therapeutics, Inc. | PTC Therapeutics, Inc. | ITF Therapeutics LLC | F. Hoffmann-La Roche Ltd | Santhera Pharmaceuticals Holding AG | REGENXBIO Inc. | Solid Biosciences Inc. | Capricor Therapeutics, Inc. | Dyne Therapeutics, Inc. | PepGen Inc. | Others
Key Highlights
Sarepta Therapeutics, Inc. - Holds a 21.3% share, driven by strong Duchenne muscular dystrophy gene therapy portfolio, advanced RNA-targeted therapeutics expertise, and expanding global rare neuromuscular disease commercialization capabilities.
PTC Therapeutics, Inc. - Holds a 16.4% share, supported by innovative nonsense mutation therapies, advanced rare disease drug development infrastructure, and strong neuromuscular disorder treatment focus.
ITF Therapeutics LLC - Holds a 11.8% share, fueled by corticosteroid-based Duchenne muscular dystrophy treatment commercialization, specialty rare disease expertise, and expanding patient support initiatives.
F. Hoffmann-La Roche Ltd - Holds a 10.2% share, driven by advanced biotechnology capabilities, precision medicine research infrastructure, and strategic investments in neuromuscular disease therapeutics.
Santhera Pharmaceuticals Holding AG - Holds a 8.4% share, supported by orphan drug development expertise, innovative respiratory and neuromuscular treatment programs, and expanding Duchenne muscular dystrophy therapy adoption.
REGENXBIO Inc. - Holds a 7.0% share, powered by proprietary NAV gene therapy platform technology, strong AAV vector development capabilities, and expanding rare genetic disease pipeline.
Solid Biosciences Inc. - Holds a 6.1% share, strengthened by precision genetic medicine research, advanced Duchenne muscular dystrophy gene therapy programs, and innovative neuromuscular treatment development.
Capricor Therapeutics, Inc. - Holds a 5.3% share, driven by cell and exosome-based therapeutic innovation, regenerative medicine expertise, and expanding cardiac complication treatment programs for Duchenne muscular dystrophy.
Dyne Therapeutics, Inc. - Holds a 4.1% share, supported by targeted exon-skipping platform development, advanced oligonucleotide delivery technologies, and expanding neuromuscular disease pipeline.
PepGen Inc. - Holds a 3.2% share, fueled by peptide-enhanced oligonucleotide delivery technologies, innovative RNA therapeutic development, and strong rare genetic disease research capabilities.
Others - Hold a combined 6.2% share, comprising emerging biotechnology firms, rare disease therapy developers, and next-generation gene editing companies advancing innovative Duchenne muscular dystrophy treatment solutions globally.
Purchase Corporate License | Market Intelligence: https://www.datamintelligence.com/buy-now-page?report=global-dunche-muscular-dystrophy-therapeutics-market?sai-v
Market Drivers
Increasing prevalence of Duchenne Muscular Dystrophy (DMD) and growing awareness regarding rare genetic disorders are significantly driving demand for advanced DMD therapeutics worldwide.
Rising adoption of exon-skipping therapies, corticosteroids, gene therapies, and mutation-specific treatments is accelerating expansion of the Duchenne Muscular Dystrophy therapeutics market globally.
Growing focus on early diagnosis, genetic screening programs, and personalized medicine approaches is strengthening demand for targeted DMD treatment solutions.
Continuous advancements in gene-editing technologies, viral vector delivery systems, and regenerative medicine are transforming the treatment landscape for Duchenne Muscular Dystrophy.
Increasing investments from pharmaceutical companies, biotechnology firms, and research organizations in rare disease drug development are contributing to rapid market growth and innovation.
Expansion of specialized neuromuscular care centers, rehabilitation services, and multidisciplinary treatment programs is improving patient access to advanced DMD therapies.
Rising regulatory approvals for novel gene therapies, exon-skipping drugs, and disease-modifying biologics are accelerating commercialization and expanding treatment options for patients.
Growing government support, orphan drug incentives, and patient advocacy initiatives are further propelling research and development activities in the DMD therapeutics market.
Industry Developments
Rapid advancement in gene therapy platforms and CRISPR-based technologies improving long-term treatment potential and disease management outcomes for DMD patients.
Increasing adoption of exon-skipping therapies and mutation-targeted medicines supporting personalized treatment approaches in Duchenne Muscular Dystrophy care.
Growing investments in next-generation biologics, stem cell therapies, and RNA-based therapeutics accelerating innovation across the DMD treatment landscape.
Expansion of clinical trials evaluating novel gene-editing therapies and advanced regenerative medicine solutions improving future treatment opportunities.
Rising collaborations among pharmaceutical companies, biotechnology firms, academic institutions, and rare disease organizations driving development of advanced DMD therapeutics.
Strategic product launches, orphan drug approvals, and regulatory clearances for innovative Duchenne Muscular Dystrophy therapies supporting global market commercialization.
Continuous innovation in wearable monitoring systems, digital rehabilitation technologies, and AI-assisted disease progression tracking improving long-term patient management and quality of life.
Regional Insights
North America 43.2% share: "Leads the market due to advanced healthcare infrastructure, strong presence of major biopharmaceutical companies, high adoption of gene therapies, and increasing investments in rare disease research and neuromuscular treatment programs."
Europe 28.5% share: "Growth supported by expanding rare disease treatment initiatives, favorable reimbursement policies, rising awareness regarding genetic disorders, and increasing adoption of advanced biologic therapies."
Asia Pacific 21.4% share: "Fastest-growing region driven by improving healthcare infrastructure, increasing genetic screening programs, rising awareness regarding neuromuscular disorders, and expanding investments in advanced treatment technologies across China, India, Japan, and South Korea."
Latin America 3.8% share: "Emerging growth supported by improving access to rare disease treatment services, increasing healthcare investments, and growing awareness regarding Duchenne Muscular Dystrophy management."
Middle East & Africa 3.1% share: "Gradual growth driven by expanding hospital infrastructure, rising awareness regarding genetic disorders, and improving access to specialized neuromuscular care services."
Speak to our analyst and get customization in the report as per your requirements:https://www.datamintelligence.com/customize/global-dunche-muscular-dystrophy-therapeutics-market?sai-v
Key Segments
➥ By Therapeutic Type
Molecular-Based: Represents the dominant segment, driven by increasing adoption of exon-skipping therapies, gene-targeted treatments, and ongoing advancements in precision medicine for Duchenne muscular dystrophy management.
Steroidal Therapy: Represents a significant segment, supported by widespread utilization of corticosteroids to slow muscle degeneration and improve mobility among DMD patients.
NSAIDs: Represents a moderate segment, fueled by growing use for pain management, inflammation reduction, and supportive symptomatic treatment in muscular dystrophy care.
Others: Represents an emerging segment, driven by ongoing development of regenerative therapies, combination treatment approaches, and supportive rehabilitation medications.
➥ By Mutation Type
Exon 51 Skipping: Represents the dominant segment, driven by higher prevalence among eligible DMD patients and increasing commercialization of approved exon-skipping therapies targeting exon 51 mutations.
Exon 53 Skipping: Represents a rapidly growing segment, supported by rising regulatory approvals and expanding research activities focused on advanced mutation-specific therapies.
Exon 45 Skipping: Represents a significant segment, fueled by increasing clinical trial investments and growing focus on personalized treatment approaches for exon 45 mutation patients.
Others: Represents a moderate segment, driven by ongoing advancements in genetic diagnostics and development of therapies targeting rare dystrophin gene mutations.
➥ By Route of Administration
Intravenous: Represents the dominant segment, driven by widespread administration of exon-skipping drugs and gene therapies requiring controlled intravenous delivery in clinical settings.
Subcutaneous: Represents a growing segment, supported by increasing development of patient-friendly biologic therapies and improved convenience in long-term disease management.
Others: Represents a moderate segment, fueled by expanding research into oral formulations and alternative drug delivery technologies for muscular dystrophy treatment.
➥ By Distribution Channel
Hospital Pharmacies: Represents the dominant segment, driven by high dependence on hospital-based specialty treatments, advanced infusion therapies, and multidisciplinary DMD care programs.
Specialty Pharmacies: Represents a significant segment, supported by increasing distribution of high-cost orphan drugs, personalized therapy management, and specialized patient support services.
Unlock 360° Market Intelligence with DataM Subscription Services: https://www.datamintelligence.com/reports-subscription
Power your decisions with real-time competitor tracking, strategic forecasts, and global investment insights all in one place.
✅ Competitive Landscape
✅ Sustainability Impact Analysis
✅ KOL / Stakeholder Insights
✅ Unmet Needs & Positioning, Pricing & Market Access Snapshots
✅ Market Volatility & Emerging Risks Analysis
✅ Quarterly Industry Report Updated
✅ Live Market & Pricing Trends
✅ Import-Export Data Monitoring
Have a look at our Subscription Dashboard: https://www.youtube.com/watch?v=x5oEiqEqTWg
Contact:
Fabian
DataM Intelligence 4market Research LLP
6th Floor, M2 Tech Hub, DataM Intelligence 4market Research LLP, Lalitha Nagar, Habsiguda, Secunderabad, Hyderabad, Telangana 500039
USA: +1 877-441-4866
UK: +44 161-870-5507
Email: fabian@datamintelligence.com
About Us -
DataM Intelligence is a Market Research and Consulting firm that provides end-to-end business solutions to organizations from Research to Consulting. We, at DataM Intelligence, leverage our top trademark trends, insights and developments to emancipate swift and astute solutions to clients like you. We encompass a multitude of syndicate reports and customized reports with a robust methodology.
Our research database features countless statistics and in-depth analyses across a wide range of 6300+ reports in 40+ domains creating business solutions for more than 200+ companies across 50+ countries; catering to the key business research needs that influence the growth trajectory of our vast clientele.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Duchenne Muscular Dystrophy Therapeutics Market to Reach USD 11.31 Billion by 2033 at 9.24% CAGR; North America Leads with 43.2% Share - Key Players: Sarepta Therapeutics, PTC Therapeutics, Roche here
News-ID: 4513246 • Views: …
More Releases from DataM intelligence 4 Market Research LLP

Medicinal Herbs Market (2026) | Herbal Medicine, Botanical Extracts, Medicinal P …
DataM Intelligence has unveiled its latest research study, "Medicinal Herbs Market Size 2026," delivering strategic intelligence designed to identify high-growth opportunities, evaluate competitive positioning, and stay ahead of rapidly evolving market trends. The report provides a comprehensive analysis of market size, revenue performance, CAGR projections, regional growth patterns, and detailed segmentation insights, along with an in-depth assessment of the key factors driving industry expansion. It also highlights emerging opportunities, investment…
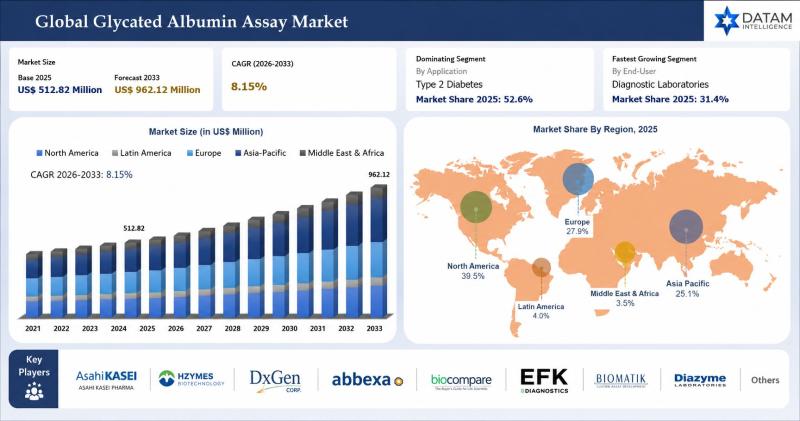
Glycated Albumin Assay Market to Reach USD 962.12 Million by 2033 at 8.15% CAGR; …
The global glycated albumin assay market was valued at USD 512.82 million in 2025 and is projected to reach nearly USD 962.12 million by 2033, growing at a CAGR of 8.15% during the forecast period from 2026 to 2033. The market is experiencing significant growth driven by the rising global prevalence of diabetes, increasing demand for advanced glycemic monitoring solutions, and growing awareness regarding early diabetes diagnosis and management. Glycated…

Photoinitiator Market Growth (2026) | UV Ink Expansion, Adhesives & Industrial C …
Market Growth & Size 2026-2033
Photoinitiator Market is estimated to reach US$ 160.70 million in 2025 to US$ 176.77 million in 2026 and projected to reach US$337.55 million by 2033 at a CAGR of 10% during the forecast period (2026-2033).
Connect with the right partners and unlock new growth opportunities today:- https://www.datamintelligence.com/partner-identification-enquiry/photoinitiator-market?kb
United States: Recent Industry Developments
✅ March 2026: Arkema S.A. expanded photoinitiator production capabilities in North America to support rising demand…
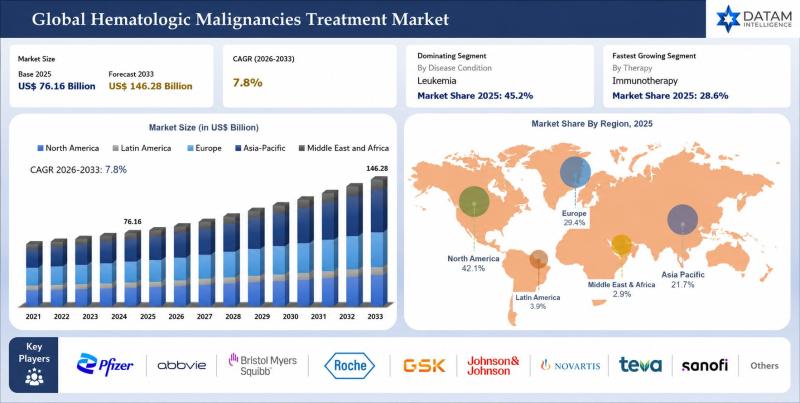
Hematologic Malignancies Treatment Market to Reach USD 146.28 Billion by 2033 at …
The global hematologic malignancies treatment market was valued at USD 76.16 billion in 2025 and is projected to reach nearly USD 146.28 billion by 2033, growing at a CAGR of 7.8% during the forecast period from 2026 to 2033. The market is witnessing substantial growth driven by the rising prevalence of blood cancers such as leukemia, lymphoma, and multiple myeloma, along with increasing demand for targeted therapies and personalized treatment…
More Releases for Duchenne
Duchenne Muscular Dystrophy (DMD) Market Growth in 2034
Market Overview
The Duchenne Muscular Dystrophy (DMD) Market is expanding rapidly as advances in genetic medicine, exon-skipping therapies, gene therapy platforms, and improved diagnostic capabilities reshape treatment options for this severe, progressive neuromuscular disorder.
DMD is caused by mutations in the dystrophin gene, leading to muscle degeneration beginning in early childhood. Growing awareness among clinicians and caregivers, widespread adoption of next-generation sequencing (NGS), and increasing availability of disease-modifying therapies have significantly strengthened…
Duchenne Muscular Dystrophy: Core Growth Enabler in the Rising Prevalence Of Chr …
Use code ONLINE30 to get 30% off on global market reports and stay ahead of tariff changes, macro trends, and global economic shifts.
What Will the Duchenne Muscular Dystrophy Industry Market Size Be by 2025?
The market size for Duchenne Muscular Dystrophy has shown significant expansion recently, burgeoning from $1.16 billion in 2024 to $1.25 billion in 2025 at a compound annual growth rate (CAGR) of 7.9%. The impressive growth in the…
Rising Prevalence Of Chronic Diseases Fuels Growth In The Duchenne Muscular Dyst …
What industry-specific factors are fueling the growth of the duchenne muscular dystrophy market?
The rise in chronic diseases is projected to stimulate the expansion of the Duchenne muscular dystrophy market in the coming years. Chronic diseases are health complications that endure for a minimum of a year. Duchenne muscular dystrophy is instrumental in the increase of chronic diseases as it addresses the unique therapeutic requirements of those suffering from this genetic…
Shaping the Duchenne Muscular Dystrophy (DMD) Therapeutics Market in 2025: Bit B …
How Big Is the Duchenne Muscular Dystrophy (DMD) Therapeutics Market Expected to Be, and What Will Its Growth Rate Be?
The Duchenne muscular dystrophy (DMD) therapeutics market will grow from $11.95 billion in 2024 to $16.45 billion in 2025, at a CAGR of 37.6%. The growth is attributed to the increasing prevalence of Duchenne muscular dystrophy, rising awareness of treatment options, healthcare spending, and government initiatives.
The Duchenne muscular dystrophy (DMD) therapeutics…
Duchenne Muscular Dystrophy Pipeline Outlook Report 2024
DelveInsight's, "Duchenne Muscular Dystrophy Pipeline Insight 2024" report provides comprehensive insights about 75+ companies and 75+ pipeline drugs in the Duchenne Muscular Dystrophy pipeline landscape. It covers the Duchenne Muscular Dystrophy pipeline drug profiles, including Duchenne Muscular Dystrophy clinical trials and nonclinical stage products. It also covers the therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
…
Duchenne Muscular Dystrophy Treatmcent Market
Global Duchenne Muscular Dystrophy Treatmcent Market Set for Robust Growth During Forecast Period
The global Duchenne Muscular Dystrophy Treatment Market is poised to witness significant growth at a high Compound Annual Growth Rate (CAGR) during the forecast period of 2023 to 2030. Duchenne muscular dystrophy (DMD) stands as a genetic disorder marked by progressive muscle degeneration and weakness, with therapeutics aimed at addressing the absence of dystrophin, a crucial protein in…