Press release
Document Management System (DMS) in Pharma | A Complete Guide
The pharmaceutical industry is one of the most highly regulated sectors in the world. Every process, from drug development to manufacturing, must follow strict rules and maintain proper documentation. This is where a Document Management System (DMS) plays a very important role.A DMS helps pharma companies store, manage, track, and control documents digitally. Instead of using paper-based systems, organisations can use a centralised platform to handle all their documents efficiently.
Request a free Demo:- https://www.amplelogic.com/schedule-demo?from=%2Fgamp-solutions%2Fdocument-management-system
What is a Document Management System (DMS)?
A Document Management System (DMS) is a software solution that helps organisations store, organise, and manage documents electronically. It also tracks document history, versions, and approvals.
In the pharmaceutical industry, a DMS is designed to meet regulatory requirements such as FDA 21 CFR Part 11 and EU Annex 11 compliance. It ensures that all documents are accurate, secure, and easily accessible.
According to AmpleLogic, a modern DMS provides complete document lifecycle management, from creation to approval, distribution, and archival.
Why is DMS Important in Pharma?
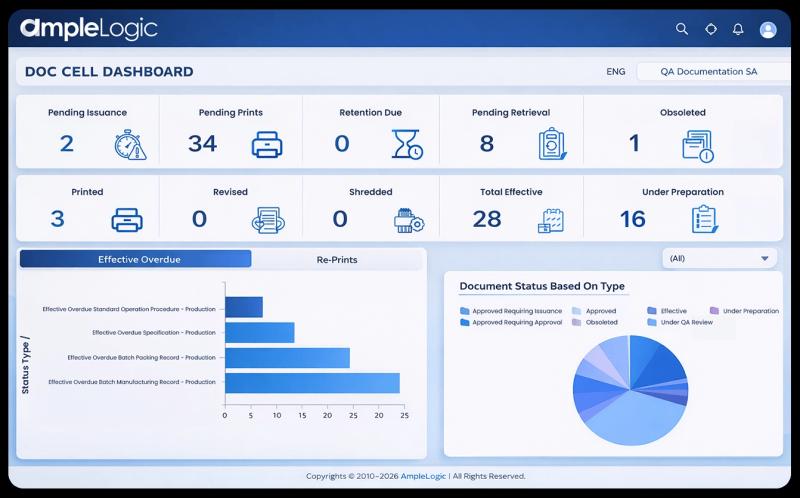
Pharma companies deal with thousands of documents such as SOPs, batch records, quality reports, and regulatory files. Managing these manually can lead to errors, delays, and compliance risks.
Here are some key reasons why DMS is essential:
1. Regulatory Compliance
Pharma companies must follow strict guidelines. A DMS ensures compliance with global regulations by maintaining audit trails, electronic signatures, and controlled workflows.
2. Version Control
Using outdated documents can lead to serious issues. A DMS ensures that only the latest version of a document is used and tracks all changes.
3. Improved Efficiency
Manual document handling is time-consuming. A DMS automates workflows such as approvals, reviews, and document retrieval.
4. Data Security
Sensitive pharmaceutical data must be protected. A DMS provides role-based access, encryption, and secure storage.
5. Audit Readiness
During audits, companies need to provide documents quickly. A DMS ensures that all records are easily searchable and available.
Key Features of a Pharma DMS
A modern DMS offers several advanced features designed specifically for the pharmaceutical industry.
1. Document Lifecycle Management
A DMS manages documents from creation to archival. This includes drafting, reviewing, approving, and storing documents in a structured way.
2. Electronic Signatures
Electronic signatures help ensure compliance and eliminate the need for paper-based approvals.
3. Audit Trails
Every change made to a document is recorded. This helps maintain transparency and accountability.
4. Advanced Search and Retrieval
Users can quickly find documents using keywords, metadata, or filters, saving time and effort.
5. Integration with Other Systems
A DMS can integrate with systems like eQMS, LIMS, and ERP to streamline operations.
6. Controlled Document Distribution
It ensures that only authorized users can access specific documents and prevents the use of obsolete versions.
Types of Documents Managed in Pharma DMS
A pharmaceutical DMS manages a wide range of documents, including:
- Standard Operating Procedures (SOPs)
- Quality documents and reports
- Manufacturing records
- Laboratory and QC documents
- Regulatory and compliance documents
- Training records
These documents are critical for maintaining product quality and compliance.
Find out more about on:-
https://www.amplelogic.com/products/document-management-system
Benefits of Using DMS in Pharma
Implementing a DMS brings multiple benefits to pharmaceutical organisations:
1. Reduced Paperwork
A DMS eliminates the need for physical documents, making the organization eco-friendlier and more efficient.
2. Better Collaboration
Teams can access and work on documents from anywhere, improving collaboration.
3. Faster Decision-Making
Quick access to accurate information helps in faster decision-making.
4. Error Reduction
Automation reduces human errors in document handling.
5. Improved Productivity
Employees spend less time searching for documents and more time on important tasks.
Challenges Without a DMS
Without a proper DMS, pharma companies may face:
- Use of outdated or incorrect documents
- Lack of version control
- Difficulty in tracking approvals
- Compliance risks and audit failures
- Data loss or security issues
For example, weak version control can lead to outdated SOPs being used, which may result in compliance violations.
How AmpleLogic DMS Helps Pharma Companies
The AmpleLogic DMS is designed specifically for regulated industries like pharmaceuticals. It offers:
- AI-powered document management
- Automated workflows for approvals and reviews
- Strong version control and audit trails
- Compliance with FDA and global standards
- Easy integration with other pharma systems
It also uses advanced technologies like OCR and smart search to make document retrieval faster and easier.
Additionally, the platform ensures complete traceability and security, which are critical for maintaining data integrity in pharma operations.
Schedule Free demo today:- https://www.amplelogic.com/contact
Future of DMS in Pharma
The future of DMS in pharma is moving towards automation and artificial intelligence. Modern systems are becoming smarter, with features like:
- AI-based document classification
- Predictive analytics
- Automated compliance checks
- Cloud-based access
These advancements will help pharma companies become more efficient, compliant, and competitive.
A Document Management System (DMS) is no longer optional for pharmaceutical companies. It is a critical tool for ensuring compliance, improving efficiency, and managing complex documentation.
By implementing a robust DMS like the one offered by AmpleLogic, organisations can streamline their processes, reduce risks, and stay audit-ready at all times.
In a highly regulated industry like pharma, having the right document management system can make a significant difference in operational success and regulatory compliance.
Read more details: https://www.amplelogic.com/gamp-solutions/document-management-system
Melange Tower, 2nd Floor, Wing-C, Patrika Nagar, HITEC City, Madhapur - 500081, Hyderabad, India
9:00 AM - 7:00 PM IST
Get in Touch:
Email Us: info@amplelogic.com
Call Us:
1800 2023 269 (Global)
+91-7396660171 (India)
Since 2010, AmpleLogic has been the trusted pharmaceutical software partner for 200,000+ users across 30+ countries - delivering a unified, AI-powered, 21 CFR Part 11 compliant platform that accelerates digital transformation across quality, laboratory, manufacturing, and regulatory operations in pharma, biotech, and life sciences.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Document Management System (DMS) in Pharma | A Complete Guide here
News-ID: 4512222 • Views: …
More Releases from Amplelogic

Annual Product Quality Review (APQR) Software - Transforming Pharma Quality with …
In the pharmaceutical industry, maintaining product quality is not a static task; it is a rigorous, continuous commitment. The Annual Product Quality Review (APQR) stands as a cornerstone of this commitment, ensuring that products remain consistent, safe, and fully compliant with evolving global regulatory standards.
Traditionally, APQR has been a reactive, labor-intensive hurdle. However, the emergence of AI-powered APQR compliance software is shifting the paradigm-moving the industry from "once-a-year" reporting…
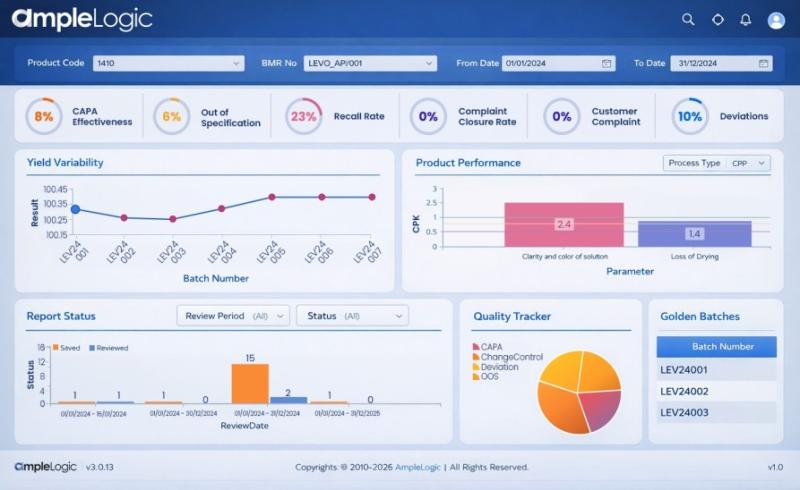
APQR Software in Pharma- A Simple Guide to Smarter Quality Reviews
In the pharmaceutical industry, maintaining product quality is not optional. It is a strict requirement set by global regulatory bodies. One of the most important processes that ensures this is the Annual Product Quality Review (APQR). Traditionally, APQR has been a manual, time-consuming, and error-prone activity. Today, with digital transformation, APQR software is changing the way pharma companies manage quality.
This explains what APQR software is, why it matters, and…
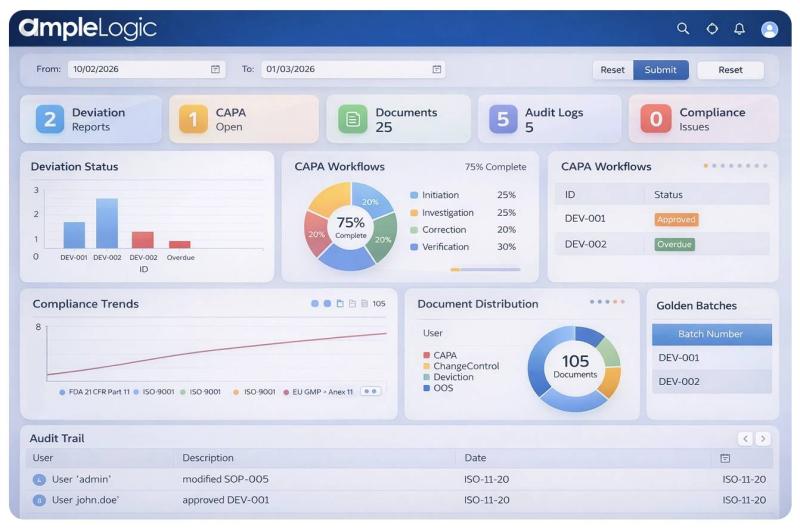
eQMS in Pharma 2026 | How AI is Transforming Electronic Quality Management Syste …
In the pharmaceutical industry, quality is everything. Every process must follow strict regulations to ensure patient safety and product effectiveness. Traditionally, many companies relied on manual systems to manage quality. However, with digital transformation, Electronic Quality Management Systems (eQMS) have become essential.
Now, the next big step is AI-powered eQMS-a smarter, faster, and more efficient way to manage quality.
We will explore what eQMS is, how AI enhances it, and…
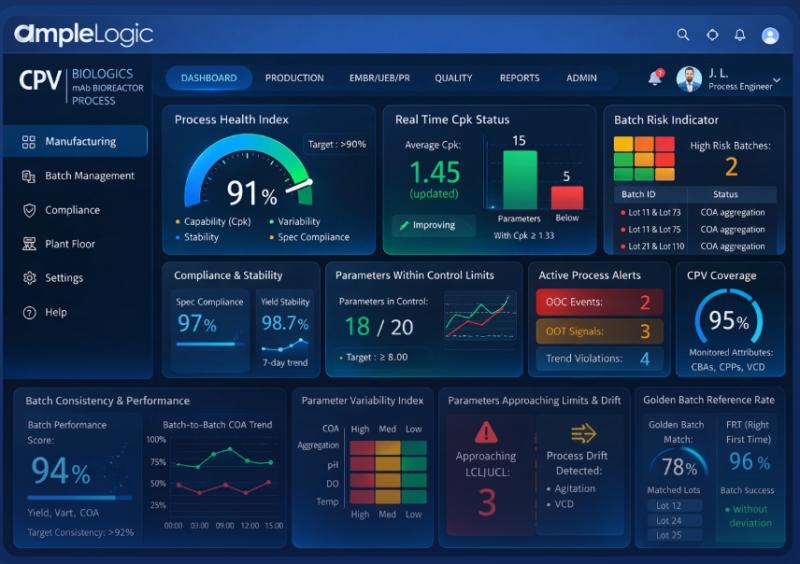
AI-Powered Continued Process Verification (CPV) in Pharma | AmpleLogic
In the pharmaceutical industry, maintaining product quality is critical. Every medicine must be safe, effective, and consistent. This is where Continued Process Verification (CPV) plays an important role.
Today, with the rise of Artificial Intelligence (AI), CPV is becoming smarter, faster, and more reliable. AI is helping pharma companies move from manual monitoring to intelligent, automated decision-making.
Continued Process Verification (CPV) is the third stage of the process validation lifecycle. It focuses…
More Releases for DMS
HBS Systems Earns Elite Status Kubota DMS Certification
HBS Systems, a leading provider of dealership management solutions for agricultural and heavy equipment dealerships, proudly announces its attainment of Elite Status in the prestigious Kubota Dealership Management System Certification (DMS) for 2024. This significant achievement follows a rigorous two-year certification process spanning 2022 to 2023, during which HBS Systems demonstrated exceptional performance in project participation, timely deliverables, and dealer usage statistics.
Certified with an impressive score of 484 points out…
Massage Gun Market is Going to Boom : DMS, Hyperice, Pleno
HTF MI introduces new research on Massage Gun covering the micro level of analysis by competitors and key business segments (2023-2029). The Massage Gun explores a comprehensive study of various segments like opportunities, size, development, innovation, sales, and overall growth of major players. The research is carried out on primary and secondary statistics sources and it consists of both qualitative and quantitative detailing.
Some of the major key players profiled…
Digital Marketing Software (DMS) Market Set for More Growth
The Digital Marketing Software (DMS) market has witnessed growth from USD XX million to USD XX million from 2017 to 2022. With the CAGR of X.X%, this market is estimated to reach USD XX million in 2029.
The report focuses on the Digital Marketing Software (DMS) market size, segment size (mainly covering product type, application, and geography), competitor landscape, recent status, and development trends. Furthermore, the report provides detailed cost analysis,…
Automotive DMS Market (Driver Monitoring System) Size, Industry Chain, Developme …
Automotive DMS Market research: the installations of DMS soared by 244% on a like-on-like basis in the first nine months of 2021.
Our data show that in the first nine months of 2021, China sold 251,511 sets of DMS for new passenger cars, 244% more than in the same period of the previous year, of which 15,201 sets, or 6% of the total were for joint venture brand cars, and 236,310…
Devacon presents ECM solution eMODAT at DMS Expo in Stuttgart
Devacon GmbH is participating as one of the exhibitors of the DMS Expo taking place in Stuttgart from 24th to 26th September. The leading trade fair for business IT is the perfect venue for the software developers to display their mobile data capturing software eMODAT which allows users to manage workflows via smartphone and tablet PC. During those three days Devacon is supported by the IT consulting company milanconsult, a…
Newgen launches its DMS OmniDocsTM version 7.0
Newgen Software Technologies Limited, a market leader in Business Process Management (BPM) and Enterprise Content Management (ECM) has announced the launch of its latest version of Document Management System (DMS) solution OmniDocsTM version 7.0. OmniDocs™ provides a highly scalable, unified repository for securely storing and managing enterprise content.
OmniDocsTM 7.0 is an ideal solution for Enterprise archival and retrieval of documents in a high-performance environment. It provides an Enterprise Content Management…