Press release
Bispecific Antibodies Market to Reach $16.81 Billion by 2035, Growing at 7.5% CAGR | Roche, Amgen, Genmab Lead the Race
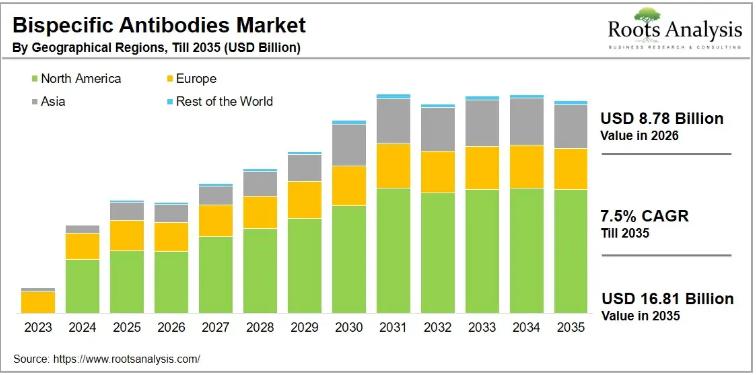
The global bispecific antibodies market, valued at USD 8.93 billion in 2025, will reach USD 16.81 billion by 2035, advancing at a compound annual growth rate of 7.5% over the forecast period from 2026 to 2035. Fifteen bispecific antibodies have already received regulatory approval worldwide, and a clinical pipeline of more than 220 candidates signals that the market's growth arc is far from exhausted. For biopharmaceutical executives, investors, and payers tracking the next wave of oncology and immunology innovation, this market represents one of the most active and consequential segments in drug development today.To explore the complete findings, request a free sample of the report at https://www.rootsanalysis.com/reports/bispecific-antibodies/request-sample.html
Market Overview
Bispecific antibodies (BsAbs) are a distinct class of therapeutic proteins engineered to simultaneously bind two separate antigens or two different epitopes on the same antigen. Unlike conventional monoclonal antibodies, which hit a single target, BsAbs redirect immune effector cells directly toward diseased tissue, deploying mechanisms that include antibody-dependent cell-mediated cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP), and complement-dependent cytotoxicity (CDC). That dual-targeting ability translates into sharper efficacy and greater disease pathway coverage, particularly in oncology, where tumor cells routinely exploit multiple escape routes.
The regulatory environment has shifted decisively in favor of BsAbs over the last four years. Six bispecific antibodies earned approval in 2022 alone, setting a single-year record. In May 2024, the U.S. FDA granted accelerated approval to Amgen's Imdelltra (tarlatamab-dlle) for extensive-stage small cell lung cancer, marking the first bispecific antibody to target a solid tumor and opening an entirely new frontier for the modality. The European Commission has kept pace; in August 2024 it approved Regeneron's odronextamab (Ordspono) for two types of lymphoma, further broadening the addressable patient population.
Corporate investment is reinforcing clinical momentum. In November 2024, BioNTech agreed to acquire Biotheus for an upfront payment of USD 800 million, plus milestone payments of up to USD 150 million, gaining a phase I/II stage PD-L1 x VEGF-A bispecific antibody (BNT327/PM8002) in the process. That same month, Merck secured a global license from LaNova for LM-299, an investigational PD-1/VEGF bispecific antibody. More recently, in April 2026, Sidewinder Therapeutics closed a USD 137 million Series B round led by OrbiMed and the Novartis Venture Fund to advance bispecific antibody-drug conjugates (ADCs) into clinical trials.
Key Growth Drivers
Expanding Regulatory Approvals Building Market Confidence The approval of 15 bispecific antibodies to date, including seven in the last year or so, has substantially raised stakeholder confidence in the modality. Each new approval validates a novel mechanism or target antigen, which in turn attracts additional R&D investment and partnership capital. The accelerated-approval pathway in the US and priority review designations in Europe are shortening development timelines and pulling more candidates toward commercialization.
Superior Efficacy Compared to Conventional Monoclonal Antibodies BsAbs engage two disease-relevant targets simultaneously, which reduces the likelihood that tumors or immune-mediated diseases will develop resistance through single-pathway escape. Clinical data across approved products, from Hemlibra in hemophilia A to Tecvayli in multiple myeloma, have demonstrated response rates and durability that single-agent monoclonals frequently cannot match. That clinical differentiation is driving formulary adoption and fueling premium pricing.
Surging Clinical Pipeline Across Oncology and Beyond More than 220 bispecific antibodies are currently approved or in clinical development, while a further 180-plus candidates remain in preclinical evaluation. Gastric cancer alone has more than 15 BsAbs in active development. In March 2023, Akeso Biopharma completed enrollment for a phase III trial of Cadonilimab in combination with chemotherapy as a first-line treatment for advanced gastric or gastroesophageal junction cancer, a milestone that illustrates just how rapidly the pipeline is maturing.
High-Value Partnership and Licensing Activity Big pharma players are actively expanding their BsAb portfolios through external deals rather than waiting for internal programs to advance. Product development and commercialization agreements and R&D licensing deals are the most common transaction types. Notable partnerships include GSK's licensing agreement with Wuxi Biologics (January 2023), Genentech's commercialization agreement with Biogen (December 2022), and Summit Therapeutics' January 2026 clinical collaboration with GSK to evaluate ivonescimab in non-small cell lung cancer.
Rising Prevalence of Chronic and Oncological Disorders Growing global incidence of hematological malignancies, solid tumors, autoimmune disorders, and ophthalmic conditions is broadening the addressable patient pool. Bispecific antibodies are particularly well-positioned in oncology because most advanced cancers are driven by multiple concurrent pathways. As treatment guidelines increasingly recognize combination or multi-pathway blockade as standard of care, BsAb products are moving earlier into treatment sequences, expanding their commercial reach.
Market Segmentation
The bispecific antibodies market segments across therapeutic area, mechanism of action, target antigen, antibody format, and geography. By therapeutic area, oncology is the clear frontrunner. Hematological malignancies, solid tumors, and skin cancers collectively account for the largest share of both approved products and late-stage pipeline candidates, reflecting the biology-driven fit between BsAb mechanisms and cancer immunotherapy. Non-oncology applications, including autoimmune disorders and ophthalmic diseases (where VABYSMO addresses neovascular AMD and diabetic macular edema), represent a growing but comparatively smaller portion of the market today.
On the mechanism-of-action dimension, T-cell retargeting and activation remains the most commercially productive approach, underpinning blockbuster products such as Blincyto and Tecvayli. Target antigens including CD20 x CD3, PD-1 x CTLA-4, and PD-L1 x TGF-beta are commanding the largest development pipelines. By antibody format, asymmetric formats represent the dominant structural class, while fragment-based formats are gaining ground in indications where smaller molecule size and tissue penetration are priorities. Asia is projected to be the fastest-growing sub-segment by region across the forecast period.
To request quote of this report, please visit:
https://www.rootsanalysis.com/reports/bispecific-antibodies/request-quote.html
Regional Insights
North America and Europe together capture approximately 64% of current bispecific antibodies market revenues, with the United States generating the majority of that combined share. The US market benefits from the FDA's willingness to grant accelerated approval for BsAbs in unmet oncology indications, the density of leading academic medical centers conducting investigator-initiated trials, and the commercial infrastructure that large biopharma companies have built over decades to support complex biologic launches. The EU's active approval cadence, exemplified by the August 2024 authorization of Regeneron's odronextamab, ensures that European patients access these therapies close to the US timeline, sustaining European commercial revenues.
Asia is the fastest-growing regional market over the 2026 to 2035 forecast period. China, in particular, has become a significant source of both clinical innovation and early-stage investment. Akeso Biopharma, which originated the PD-1/VEGF bispecific ivonescimab (now subject to multiple global partnerships), exemplifies the strength of Chinese BsAb developers. South Korea and Japan are also contributing through domestic trials and manufacturing investments. Regulatory agencies across the region are shortening review timelines for novel biologics, a structural change that will accelerate the commercialization of Asian-developed BsAbs within local markets and globally.
Competitive Landscape
The bispecific antibodies market features over 120 drug developers spanning large multinationals, mid-sized biotechs, and venture-backed startups. Key players include Akeso Biopharma, Amgen, AstraZeneca, Genmab, Immunocore, Janssen Research & Development, Merck, Pfizer, Roche, and Zymeworks.
Roche holds the broadest commercial footprint, with three approved BsAbs: VABYSMO (February 2022), Tecvayli (October 2022), and Columvi (full FDA approval June 2023). Amgen's BLINCYTO, one of the earliest approvals in the class (2014), continues to generate substantial revenues, while Imdelltra's 2024 approval in small cell lung cancer has opened a new revenue stream. Technology platform differentiation is a primary competitive axis; platforms such as Amgen's BiTE, Genmab's DuoBody, Roche's CrossMab, and Xencor's XmAb are each associated with distinct intellectual property positions that underpin licensing and co-development deals. The market is not yet consolidating around a small group of winners; instead, it is expanding as new mechanisms and indications open up, giving well-capitalized mid-sized players genuine opportunities to compete with established leaders through pipeline strength and partnership access.
Browse Full Report Description + Research Methodology + Table of Content + Infographics here:
https://www.rootsanalysis.com/reports/bispecific-antibodies/286.html
Contact Details
Gaurav Chaudhary
Email: Gaurav.chaudhary@rootsanalysis.com or sales@rootsanalysis.com
Website: https://www.rootsanalysis.com
About Roots Analysis
Roots Analysis is a global leader in the pharma / biotech market research. Having worked with over 750 clients worldwide, including Fortune 500 companies, start-ups, academia, venture capitalists and strategic investors for more than a decade, we offer a highly analytical / data-driven perspective to a network of over 450,000 senior industry stakeholders looking for credible market insights. All reports provided by us are structured in a way that enables the reader to develop a thorough perspective on the given subject. Apart from writing reports on identified areas, we provide bespoke research / consulting services dedicated to serve our clients in the best possible way.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Bispecific Antibodies Market to Reach $16.81 Billion by 2035, Growing at 7.5% CAGR | Roche, Amgen, Genmab Lead the Race here
News-ID: 4511281 • Views: …
More Releases from Roots Analysis
Metabolomics Market to Reach $1.06 Billion by 2035 at 14.8% CAGR | Metabolon, Cr …
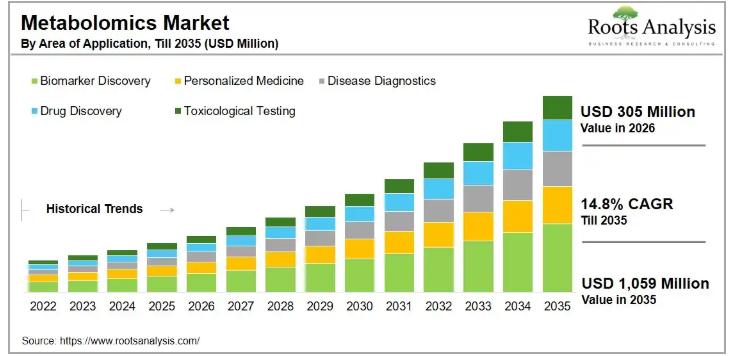
The global metabolomics market, valued at USD 265 million in 2025 and USD 305 million in 2026, will grow to USD 1,059 million by 2035, compounding at 14.8% annually across the 2026-2035 forecast period. That trajectory reflects surging demand from pharmaceutical companies, academic research institutions, and contract research organizations that need precise metabolite profiling but lack the infrastructure to perform it in-house. The numbers make a clear business case: this…
Manufacturing Execution System Market to Reach $20.46 Billion by 2035 at 16.0% C …
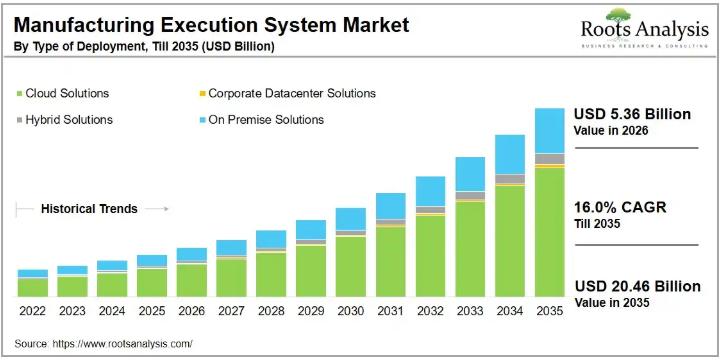
The global manufacturing execution system market, valued at USD 4.62 billion in 2025, is projected to reach USD 5.36 billion in 2026 and USD 20.46 billion by 2035, representing a compound annual growth rate of 16.0% over the forecast period. Sustained demand from pharmaceutical, life science, and medical device manufacturers, combined with accelerating Industry 4.0 adoption, is pushing the market into one of its highest growth cycles on record.
To explore…
Biopreservation Market to Reach USD 15.5 Billion by 2035 at 23.6% CAGR | BioLife …
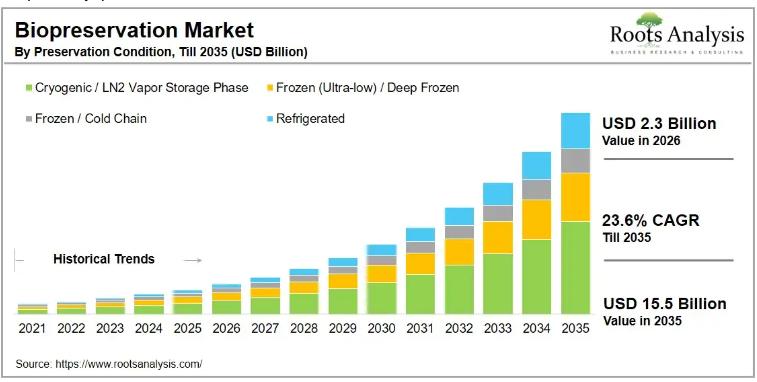
The global biopreservation market for media, valued at USD 1.9 billion in 2025, is on course to reach USD 2.3 billion in 2026 and USD 15.5 billion by 2035, expanding at a compound annual growth rate (CAGR) of 23.6% across the 2026 to 2035 forecast period. This growth trajectory reflects a structural shift in how the life sciences industry stores and transports temperature-sensitive biological materials, with cell therapies, biobanking, and…
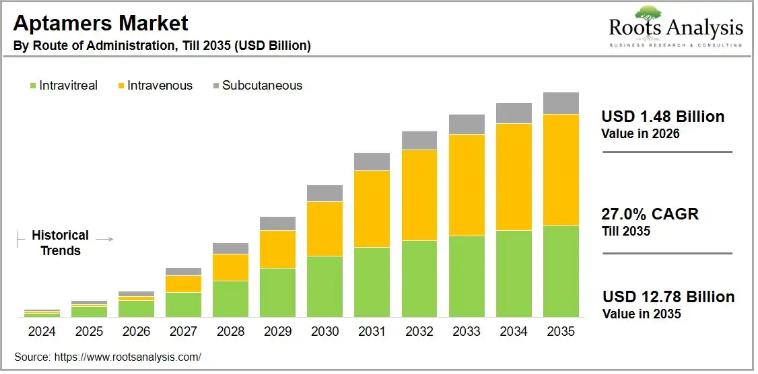
Aptamers Market to Reach $12.78 Billion by 2035 at 27.0% CAGR | SomaLogic, NOXXO …
The global aptamers market, valued at USD 0.92 billion in 2025, will reach USD 1.48 billion in 2026 and expand to USD 12.78 billion by 2035, advancing at a compound annual growth rate (CAGR) of 27.0% over the forecast period. This growth reflects accelerating investment across aptamer therapeutics, technologies, and services, as pharmaceutical developers recognize the clinical and commercial potential of these highly specific, single-stranded DNA, RNA, and peptide molecules.…
More Releases for BsAb
Creative Biolabs Takes the Spotlight in Bispecific Innovation: The Partner for " …
New York, USA - March 3, 2025 - A revolution in bispecifics is brewing in labs worldwide-and Creative Biolabs is leading the charge to turn bsAbs from "gambles" into precision tools.
Image: https://www.getnews.info/uploads/0c59d26f78bf6dd58aafee5bc68d7d8c.jpg
BsAb Bottleneck: Why Most Stumble Out of the Gate
"BsAbs aren't just fancy science-they're patient game-changers," says a lead bispecific engineer at Creative Biolabs. "Imagine an antibody that drags a cancer cell straight to immune system hitmen and blocks its…
Creative Biolabs Returns to Exhibit at Antibody Engineering & Therapeutics (US) …
Creative Biolabs will present their bispecific antibody expertise at Antibody Engineering & Therapeutics (US) 2024 next week.
New York, USA - December 10, 2024 - The Antibody Engineering & Therapeutics (US) conference for 2024 is over the horizon, from December 15 to 18 in San Diego, hosted by Informa, which, well honored as a prime event in antibody engineering, will reunite leaders all around the world to bring up the brightest…
Bispecific Antibodies: The Future of Therapeutics and Creative Biolabs' Comprehe …
Creative Biolabs stands as a trusted partner for researchers and pharmaceutical companies navigating the complexities of bsAb research and development.
New York, USA - December 2, 2024 - "Bispecific antibodies [https://www.creative-biolabs.com/bsab/] are 'stealing' the spotlight in the therapeutic antibody world," a scientist at Creative Biolabs says, "with their unique ability to simultaneously bind two antigens or epitopes, thus reshaping approaches to disease treatment with unparalleled versatility."
Image: https://www.getnews.info/uploads/0c59d26f78bf6dd58aafee5bc68d7d8c.jpg
As these next-gen antibodies have…
Dual-Target Power: Creative Biolabs Launches a One-Stop BsAb Platform for Cancer …
According to Creative Biolabs, they begin their custom development of bispecific antibodies by working together with the researchers to enhance the development of cancer immunotherapy.
New York, USA - November 7, 2024 - "Bispecific antibodies [https://www.creative-biolabs.com/bsab/bispecific-antibody-bsab-development-service.htm] can bind and link two separate antigens simultaneously, bypassing the functional limitations of conventional mAbs," said a Creative Biolabs' representative. "This way, tumor cells can be precisely targeted and immune cells can be effectively turned…
Bispecific Drug Innovation - Creative Biolabs Concludes Its Journey at the 15th …
On September 5, Creative Biolabs successfully concluded its participation in the 15th Annual World Bispecific Summit.
New York, USA - September 10, 2024 - The summit brought together leading experts in the bispecific antibody [https://www.creative-biolabs.com/bsab/bispecific-antibody-bsab-development-service.htm] (BsAb) field from around the globe, offering attendees a rich platform to discuss cutting-edge developments and future directions.
Image: https://www.getnews.info/uploads/b7ea13d648fcb7917ba9b55571b2ba35.png
In recent years, this field has grown rapidly, becoming a focal point in biopharmaceutical research and development. A…
Bispecific Antibodies Market Outlook to 2026 - Pfizer, Novartis , Amgen, Bayer A …
Bispecific Antibodies Market Overview
Bispecific antibody (BsAb) is an artificial protein that is composed of fragments of two different monoclonal antibodies and has ability to bind to two different types of antigen. Cancer immunotherapy is the most widely explored application of bispecific antibody. Lung, breast and colon cancer are the wider applications of BsAb. Bispecific antibody simultaneously binds to a cytotoxic cell and target tumor cell and destroys it. Bispecific antibodies…