Press release
GM1 Gangliosidosis Treatment Market to Reach USD 626.86 Million by 2033 at 16.6% CAGR; North America Leads with 40.0% Share - Key Players: Passage Bio, Lysogene, Sanofi
The global GM1 gangliosidosis treatment market was valued at USD 186.26 million in 2025 and is projected to reach nearly USD 626.86 million by 2033, growing at a CAGR of 16.6% during the forecast period from 2026 to 2033. The market is witnessing rapid growth driven by increasing focus on rare genetic neurological disorders, rising investments in advanced gene therapy research, and growing adoption of precision medicine approaches for lysosomal storage diseases. GM1 gangliosidosis is a rare inherited disorder caused by mutations in the GLB1 gene, resulting in progressive neurodegeneration and severe neurological complications. Increasing clinical research activities focused on gene replacement therapies, substrate reduction therapies, and enzyme based treatment strategies are significantly accelerating market expansion globally. In addition, rising awareness regarding rare pediatric disorders, expanding newborn screening programs, and increasing support from rare disease advocacy organizations are improving early diagnosis and treatment accessibility.The market is also benefiting from strong regulatory support, orphan drug incentives, and accelerated approval pathways for ultra rare disease therapies. Regulatory authorities such as the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) continue to support innovative GM1 gangliosidosis therapies through orphan drug designation, fast track programs, and rare pediatric disease incentives. Companies including Passage Bio, Inc., Azafaros B.V., and Lysogene S.A. are actively focusing on advanced gene therapies, central nervous system targeted treatments, and expanded clinical development programs to strengthen their market presence. Furthermore, increasing collaborations between biotechnology companies, research institutions, and rare disease foundations are creating substantial growth opportunities for the GM1 gangliosidosis treatment market worldwide.
Get a Sample PDF Of This Report (Get Higher Priority for Corporate Email ID): https://www.datamintelligence.com/download-sample/gm1-gangliosidosis-treatment-market?sai-v
Key Developments
February 2026: Rising advancements in gene replacement therapies and adeno-associated viral vector technologies accelerated innovation in GM1 gangliosidosis treatment across North America and Europe. Major companies including Passage Bio, Sio Gene Therapies, and Axovant Gene Therapies strengthened research activities focused on neurodegenerative lysosomal storage disorders.
January 2026: Increasing investments in rare disease research and orphan drug development across the United States, Germany, Japan, and the United Kingdom improved clinical development pathways for GM1 gangliosidosis therapies. Regulatory agencies expanded support for fast-track and rare pediatric disease designation programs.
December 2025: Growing adoption of genetic screening and advanced diagnostic technologies improved early detection of GM1 gangliosidosis in pediatric populations globally. Hospitals and specialty genetic centers across Europe and North America strengthened integration of next generation sequencing and enzyme activity testing platforms.
November 2025: Expansion of clinical trials evaluating gene therapy and substrate reduction therapy approaches increased treatment development opportunities for GM1 gangliosidosis patients worldwide. Biotechnology companies collaborated with academic research institutions to improve long-term neurological outcomes and disease management strategies.
October 2025: Rising advancements in CNS-targeted delivery systems, biomarker monitoring, and neuroprotective therapeutic approaches improved precision treatment development for GM1 gangliosidosis globally. Research institutions accelerated innovation in blood-brain barrier penetration technologies supporting rare neurological disease therapies.
September 2025: Increasing collaborations between pharmaceutical companies, patient advocacy organizations, and rare disease foundations accelerated awareness programs and global clinical research initiatives. Asia-Pacific regions including Japan and South Korea strengthened investments in genetic disease treatment infrastructure and rare disease registries.
August 2025: Growing healthcare investments and orphan drug incentives across Middle East, Latin America, and Asia-Pacific regions supported expansion of rare disease treatment accessibility and specialized care services. Governments focused on improving reimbursement support and access to advanced genetic therapies for pediatric neurological disorders.
July 2025: Rising demand for personalized medicine, digital patient monitoring, and multidisciplinary neurological care improved long-term disease management for GM1 gangliosidosis patients globally. Healthcare providers increasingly integrated telehealth and remote monitoring solutions into rare disease management programs.
June 2025: Increasing focus on rare pediatric neurological disorders, precision medicine, and advanced genetic therapies supported strong growth in the GM1 gangliosidosis treatment market worldwide. North America maintained market leadership due to strong biotechnology research infrastructure, while Europe and Asia-Pacific emerged as significant growth regions driven by expanding rare disease research initiatives.
Key Players
Passage Bio | Lysogene | Sio Gene Therapies, Inc. | BioStrategies LC | Sanofi | AZAFAROS | Dorphan S.A. | Idorsia Pharmaceuticals Ltd | SphinCS GmbH | Viatris Inc. | Others
Key Highlights
Passage Bio - Holds a 16.2% share, driven by advanced gene therapy research programs, CNS-focused rare disease pipeline expansion, and strategic AAV-based therapeutic development capabilities.
Lysogene - Holds a 14.1% share, supported by innovative gene therapy platforms, rare neurodegenerative disorder treatment research, and expanding clinical-stage development initiatives.
Sio Gene Therapies, Inc. - Holds an 11.8% share, fueled by viral vector-based gene therapy technologies, orphan neurological disease treatment programs, and advanced precision medicine capabilities.
BioStrategies LC - Holds a 9.4% share, driven by specialty biotechnology consulting expertise, rare disease therapeutic strategy development, and expanding pharmaceutical commercialization support services.
Sanofi - Holds an 8.2% share, supported by strong rare disease treatment portfolio, extensive biologics manufacturing infrastructure, and growing investments in advanced genetic therapies.
AZAFAROS - Holds a 7.0% share, powered by novel rare metabolic disorder drug development, innovative small-molecule therapeutic research, and expanding orphan disease treatment programs.
Dorphan S.A. - Holds a 5.9% share, driven by orphan drug development expertise, specialty pharmaceutical commercialization capabilities, and strong focus on rare neurological disorders.
Idorsia Pharmaceuticals Ltd - Holds a 4.8% share, supported by innovative CNS-focused pharmaceutical R&D programs, advanced small molecule research technologies, and strategic rare disease collaborations.
SphinCS GmbH - Holds a 3.7% share, fueled by precision medicine technologies, specialized CNS therapeutic development, and expanding neuroscience research capabilities.
Viatris Inc. - Holds a 2.9% share, driven by global pharmaceutical manufacturing infrastructure, specialty medicine distribution capabilities, and expanding rare disease treatment accessibility initiatives.
Others - Hold a combined 16.0% share, comprising emerging biotechnology companies, orphan drug developers, and regional pharmaceutical innovators advancing gene therapies and rare neurological disorder treatment solutions globally.
Purchase Corporate License | Market Intelligence: https://www.datamintelligence.com/buy-now-page?report=gm1-gangliosidosis-treatment-market?sai-v
Market Drivers
Increasing prevalence of rare lysosomal storage disorders and growing awareness regarding GM1 gangliosidosis are significantly driving demand for advanced treatment solutions globally.
Growing adoption of gene therapies, enzyme replacement therapies, and substrate reduction therapies is accelerating expansion of the GM1 gangliosidosis treatment market worldwide.
Rising investments in rare disease research, orphan drug development, and precision medicine initiatives are supporting continuous innovation in GM1 gangliosidosis therapeutics.
Increasing implementation of newborn screening programs and advanced genetic diagnostics is improving early disease detection and enabling timely therapeutic intervention.
Expansion of regulatory incentives including orphan drug designation, fast-track approvals, and rare pediatric disease programs is strengthening commercialization opportunities for innovative GM1 therapies.
Growing healthcare expenditure, expanding access to specialty neurology care, and rising awareness regarding pediatric neurodegenerative disorders are contributing to market growth globally.
Continuous advancements in AAV-based gene therapies, CNS-targeted drug delivery technologies, and biomarker research are further propelling development of the GM1 gangliosidosis treatment landscape.
Industry Developments
Rapid advancement in gene replacement therapies targeting GLB1 mutations improving long-term treatment potential for GM1 gangliosidosis patients.
Increasing clinical development of therapies such as PBGM01, LYS-GM101, and nizubaglustat supporting expansion of the rare neurodegenerative disease treatment pipeline.
Growing investments in enzyme replacement therapies and substrate reduction therapies enhancing treatment strategies for neuropathic manifestations of GM1 gangliosidosis.
Expansion of clinical trials evaluating AAV9 and AAVhu68 gene therapy platforms accelerating innovation in CNS-targeted therapeutic approaches.
Rising collaborations among biotechnology companies, rare disease foundations, and research institutions accelerating development of advanced GM1 therapeutics globally.
Strategic orphan drug designations and supportive regulatory frameworks encouraging commercialization of breakthrough therapies for ultra-rare neurological disorders.
Continuous innovation in biomarker monitoring, intrathecal drug delivery systems, and personalized treatment approaches improving patient management and long-term clinical outcomes.
Regional Insights
North America 40.0% share: "Leads the market due to strong rare disease research infrastructure, advanced clinical trial capabilities, increasing orphan drug approvals, and high adoption of innovative gene therapies."
Europe 28.5% share: "Growth supported by expanding rare disease awareness programs, favorable regulatory incentives, increasing investments in gene therapy research, and strong healthcare reimbursement frameworks."
Asia Pacific 23.4% share: "Fastest-growing region driven by improving healthcare infrastructure, expanding rare disease registries, increasing genetic testing adoption, and rising pharmaceutical investments in countries such as China, Japan, South Korea, and India."
Latin America 4.5% share: "Emerging growth supported by improving access to rare disease diagnostics, increasing awareness regarding pediatric neurological disorders, and gradual expansion of specialty treatment centers."
Middle East & Africa 3.6% share: "Gradual growth driven by healthcare modernization initiatives, rising investments in rare disease management infrastructure, and improving access to advanced genetic diagnostic technologies."
Speak to our analyst and get customization in the report as per your requirements:https://www.datamintelligence.com/customize/gm1-gangliosidosis-treatment-market?sai-v
Key Segments
➥ By Type
GM1 Gangliosidosis Type 1 (Infantile): Represents the dominant segment, driven by high disease severity, early symptom onset, and increasing focus on early diagnosis and intensive therapeutic intervention.
GM1 Gangliosidosis Type 2 (Juvenile): Represents a significant segment, supported by growing awareness regarding rare lysosomal storage disorders and expanding access to supportive and targeted treatment options.
GM1 Gangliosidosis Type 3 (Adult): Represents a moderate segment, fueled by increasing diagnosis of late-onset neurological manifestations and ongoing advancements in long-term disease management approaches.
➥ By Treatment
Enzyme Replacement Therapy: Represents a significant segment, driven by increasing research into recombinant enzyme therapies aimed at reducing substrate accumulation and improving neurological outcomes.
Gene Therapy: Represents the rapidly growing segment, supported by rising investments in rare disease therapeutics and advancements in viral vector-based gene delivery technologies.
Stem Cell Transplants: Represents a moderate segment, fueled by ongoing exploration of regenerative treatment approaches for slowing disease progression and improving patient outcomes.
Substrate Reduction Therapy: Represents a growing segment, driven by increasing development of therapies designed to minimize ganglioside accumulation in affected cells.
Pharmacological Chaperones: Represents an emerging segment, supported by rising research into mutation-specific therapies that improve enzyme stability and functionality.
Miglustat: Represents a significant segment, fueled by increasing off-label and investigational use in managing lysosomal storage disorders and neurological symptoms.
N-Butyl-Deoxynojirimycin (NB-DNJ): Represents a developing segment, driven by ongoing clinical research focused on substrate reduction mechanisms for rare metabolic disorders.
Anticonvulsant Drugs: Represents the dominant supportive care segment, supported by widespread use in seizure management and neurological symptom control among GM1 gangliosidosis patients.
➥ By End User
Hospitals: Represents the dominant segment, driven by availability of specialized neurology departments, advanced diagnostic infrastructure, and multidisciplinary rare disease treatment services.
Ambulatory Surgical Centers: Represents a growing segment, supported by increasing preference for outpatient infusion therapies and minimally invasive supportive treatment procedures.
Others: Represents a moderate segment, fueled by expanding utilization in specialty clinics, rehabilitation centers, and research institutions focused on rare genetic disorders.
Unlock 360° Market Intelligence with DataM Subscription Services: https://www.datamintelligence.com/reports-subscription
Power your decisions with real-time competitor tracking, strategic forecasts, and global investment insights all in one place.
✅ Competitive Landscape
✅ Sustainability Impact Analysis
✅ KOL / Stakeholder Insights
✅ Unmet Needs & Positioning, Pricing & Market Access Snapshots
✅ Market Volatility & Emerging Risks Analysis
✅ Quarterly Industry Report Updated
✅ Live Market & Pricing Trends
✅ Import-Export Data Monitoring
Have a look at our Subscription Dashboard: https://www.youtube.com/watch?v=x5oEiqEqTWg
Contact:
Fabian
DataM Intelligence 4market Research LLP
6th Floor, M2 Tech Hub, DataM Intelligence 4market Research LLP, Lalitha Nagar, Habsiguda, Secunderabad, Hyderabad, Telangana 500039
USA: +1 877-441-4866
UK: +44 161-870-5507
Email: fabian@datamintelligence.com
About Us -
DataM Intelligence is a Market Research and Consulting firm that provides end-to-end business solutions to organizations from Research to Consulting. We, at DataM Intelligence, leverage our top trademark trends, insights and developments to emancipate swift and astute solutions to clients like you. We encompass a multitude of syndicate reports and customized reports with a robust methodology.
Our research database features countless statistics and in-depth analyses across a wide range of 6300+ reports in 40+ domains creating business solutions for more than 200+ companies across 50+ countries; catering to the key business research needs that influence the growth trajectory of our vast clientele.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release GM1 Gangliosidosis Treatment Market to Reach USD 626.86 Million by 2033 at 16.6% CAGR; North America Leads with 40.0% Share - Key Players: Passage Bio, Lysogene, Sanofi here
News-ID: 4511137 • Views: …
More Releases from DataM intelligence 4 Market Research LLP

Shale Gas Market (20260 | LNG Growth, Hydraulic Fracturing, US Production Domina …
DataM Intelligence has unveiled its latest research study, "Shale Gas Market Size 2026," delivering strategic intelligence designed to identify high-growth opportunities, evaluate competitive positioning, and stay ahead of rapidly evolving market trends. The report provides a comprehensive analysis of market size, revenue performance, CAGR projections, regional growth patterns, and detailed segmentation insights, along with an in-depth assessment of the key factors driving industry expansion. It also highlights emerging opportunities, investment…
Behavioral Health Market to Reach US$ 260.2 Billion by 2033 as Demand for Mental …
DataM Intelligence has released a new research report titled "Behavioral Health Market Size 2026". The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in terms…

Powered Wheelchair Market (2026-2033) | Smart Electric Chairs, Electric Mobility …
DataM Intelligence has unveiled its latest research study, "Powered Wheelchair Market Size 2026," delivering strategic intelligence designed to identify high-growth opportunities, evaluate competitive positioning, and stay ahead of rapidly evolving market trends. The report provides a comprehensive analysis of market size, revenue performance, CAGR projections, regional growth patterns, and detailed segmentation insights, along with an in-depth assessment of the key factors driving industry expansion. It also highlights emerging opportunities, investment…
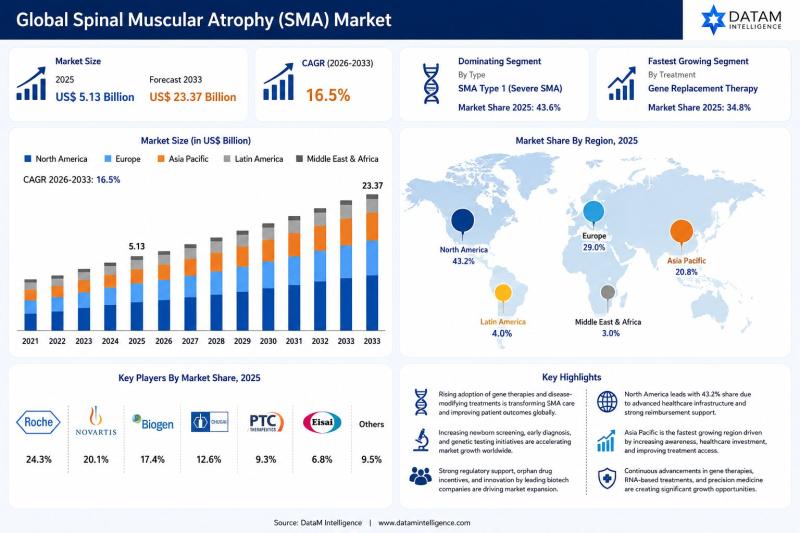
Spinal Muscular Atrophy Market to Reach US$ 23.37 Billion by 2033 at 16.5% CAGR; …
The global spinal muscular atrophy market reached US$ 5.13 billion in 2025 and is expected to reach US$ 23.37 billion by 2033, growing at a CAGR of 16.5% during the forecast period from 2026 to 2033. The market is witnessing rapid growth driven by increasing awareness of rare genetic neuromuscular disorders, rising adoption of advanced genetic testing technologies, and growing availability of disease modifying therapies for spinal muscular atrophy (SMA).…
More Releases for GM1
GM1 Gangliosidosis Treatment Market (2026) | GM1 Gangliosidosis Type 2 (Juvenile …
DataM Intelligence has released a new research report titled "GM1 Gangliosidosis Treatment Market Size 2026" The report delivers in-depth insights into key market dynamics, including regional growth trends, market segmentation, CAGR projections, and the revenue performance of leading industry players. It also highlights major growth drivers shaping the market landscape. Designed to provide a clear and comprehensive perspective, the report offers a detailed view of the current market size in…
Leading Companies Fueling Innovation and Growth in the GM1 Gangliosidosis Market
Exploring the Future Growth of the GM1 Gangliosidosis Market
The GM1 gangliosidosis market is poised for significant expansion over the coming years. Forecasts predict the market will reach $0.96 billion by 2030, growing at a compound annual growth rate (CAGR) of 10.8%. This upward trend is supported by advancements in the gene therapy pipeline, incentives for orphan drug development, enhanced diagnostic screening methods, stronger patient advocacy efforts, and increased…
GM1 Gangliosidosis Market Expected to Reach USD 1.6 Billion by 2034
GM1 gangliosidosis is a rare, inherited lysosomal storage disorder caused by mutations in the GLB1 gene, leading to a deficiency of the β-galactosidase enzyme. This results in the accumulation of GM1 gangliosides in cells, primarily affecting the central nervous system. The disease is classified into infantile, juvenile, and adult-onset forms, with infantile GM1 being the most severe and often fatal in early childhood.
Download Full PDF Sample Copy of Market Report…
GM1 Gangliosidosis Market Massive Growth opportunity Ahead
GM1 Gangliosidosis Market Outlook 2024-2034: Expanding Research, Orphan Drug Approvals, and Regional Growth
Introduction
GM1 Gangliosidosis is an ultra-rare, inherited lysosomal storage disorder caused by mutations in the GLB1 gene, leading to progressive neurodegeneration. Patients experience a range of symptoms-from developmental delays and skeletal abnormalities to severe motor dysfunction and premature mortality. With no approved curative therapy to date, treatment options remain largely supportive.
However, the last decade has witnessed a surge in…
GM1 Gangliosidosis Market Size And Global Industry Forecast 2034
Introduction
The GM1 Gangliosidosis Market is gaining attention as advancements in genetic research and rare disease drug development open new possibilities for this ultra-rare lysosomal storage disorder. GM1 gangliosidosis is caused by mutations in the GLB1 gene, leading to a deficiency of the β-galactosidase enzyme and progressive neurodegeneration. With no approved curative treatments currently available, ongoing innovation in gene therapy, enzyme replacement therapy (ERT), and small molecule chaperones is fueling significant…
GM1 Gangliosidosis Treatment Market
The GM1 Gangliosidosis Treatment Market is expected to witness substantial growth during the forecast period of 2023-2030. Here's an overview of the dynamics, segment analysis, geographical share, and impact of COVID-19 and the Russia-Ukraine war:
Market Overview:
The global GM1 gangliosidosis treatment market is anticipated to experience significant growth due to factors such as rising lysosomal storage disorders, increased research and development activities, and government investments in pharmaceuticals and biotechnology.
Market Dynamics:
Drivers:
Government Investments…
