Press release
Clinical Trial Supply and Logistics Market to Reach $7.93 Billion by 2034 as Decentralized Trials and Cold Chain Demands Reshape Drug Development Infrastructure
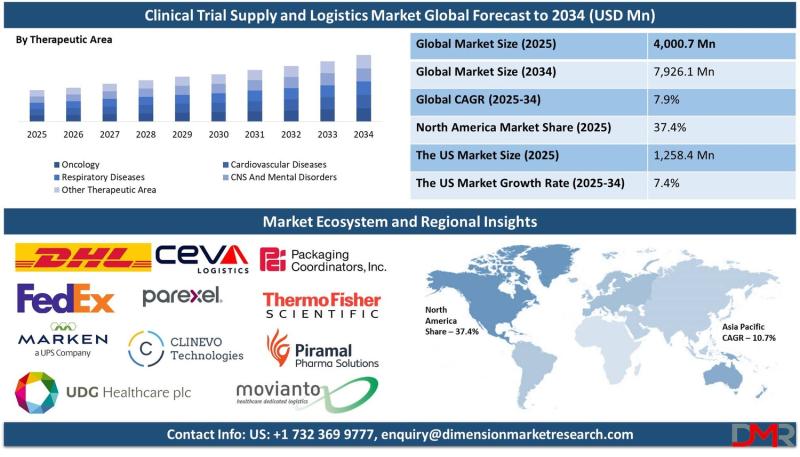
The global Clinical Trial Supply and Logistics Market is on a robust growth trajectory, with market valuation projected to rise from an estimated USD 4,000.7 million in 2025 to USD 7,926.1 million by 2034, registering a strong compound annual growth rate (CAGR) of 7.9%. According to Dimension Market Research, this expansion is being driven by four converging forces: the escalating number of global clinical trials across pharmaceutical and biotech sectors, the rising complexity of temperature-sensitive biologics and cell and gene therapies, the rapid adoption of decentralized and direct-to-patient trial models, and the integration of digital technologies including AI, blockchain, and IoT into supply chain management.Clinical trial supply and logistics-encompassing logistics and distribution, storage and retention, packaging, labeling, manufacturing, and comparator sourcing-has become a critical success factor as pharmaceutical companies conduct increasingly complex, multi-country trials. According to Dimension Market Research, the U.S. market alone is projected to reach USD 1,258.4 million in 2025 and grow to USD 2,396.5 million by 2034 at a CAGR of 7.4% , driven by the U.S. conducting nearly 40% of all global clinical trials with the FDA's well-structured regulatory framework. With North America commanding 37.4% of global market revenue and the total number of registered clinical studies reaching 453,803 by 2023, the sector is witnessing unprecedented demand for specialized, technology-enabled supply chain solutions.
📄 Get Your Sample Report Today → https://dimensionmarketresearch.com/request-sample/clinical-trial-supply-and-logistics-market/
🔷 The News Angle: From Paper-Based Tracking to AI-Powered Orchestration-The Clinical Supply Chain Revolution
The dominant narrative reshaping the clinical trial supply and logistics market is the fundamental transition from manual, paper-based supply chain management to digitally orchestrated, AI-powered logistics platforms that enable real-time tracking, predictive analytics, and end-to-end visibility across global clinical trial networks.
The rise of decentralized and direct-to-patient trials is the most powerful catalyst. Decentralized clinical trials (DCTs) are revolutionizing the supply chain with direct-to-patient (DTP) delivery of investigational drugs. Patient-centric trials and studies of rare diseases have fueled demand for flexible distribution frameworks from logistics providers. DCTs reduce patient burden through reduced site visits, with drugs and medical consumables delivered directly to subjects. This momentum accelerated post-pandemic, with companies searching for efficient, effective trials in compliance with Good Distribution Practices (GDP). Home trials have prompted strong supply chain capabilities enabling real-time tracking and remote monitoring.
Cold chain logistics as a critical imperative is equally transformative. Cold chain logistics is imperative for gene therapies, vaccines, and biologics, driving demand for temperature-controlled shipping and storage. The expansion of biologic and gene therapy studies demands high-tech supply options for temperature-sensitive shipping and storage of drugs. Ultra-low temperature freezers, real-time tracking tools, and specialized packaging represent significant infrastructure investments. According to the report, cold chain logistics is particularly critical for oncology trials, which involve monoclonal antibodies and CAR-T cell therapy requiring ultra-cold temperatures.
Digital technology integration represents the third pillar. The logistics and supply base in clinical trials is changing with integration of new technologies including AI, blockchain, and IoT. AI-powered predictive analysis is improving efficiency in pharmaceutical companies' supply chains, minimizing drug wastage, and improving inventory forecasting. Blockchain technology is offering enhanced traceability, authenticating drug genuineness, and improving compliance with regulators through secure, unalterable transaction records. IoT-facilitated temperature monitoring technology is becoming increasingly significant, with real-time tracking offering assurance for temperature-sensitive drug integrity. These technological enhancements minimize operational inefficiencies and improve transparency in the supply base.
🔷 Key Insights: Data Points Defining the Clinical Trial Supply and Logistics Market
North America Leads (37.4% Share in 2025): High density of pharmaceutical and biotechnology industries, strong infrastructure, advanced technological capabilities, and the FDA's well-structured regulatory framework drive regional dominance.
Logistics & Distribution Dominates Service Segment (27.1% Share): Timely and secure delivery of investigational drugs, medical devices, and bio-samples across multi-country trials with temperature-sensitive shipments and real-time tracking.
Phase I Trials Lead Phase Segment: High demand for complex supply chain offerings; investigational drugs require careful handling, secure shipping, and stringent protocol adherence; approximately 30-40% failure rate creates constant resupply demand.
Oncology Leads Therapeutic Area: High disease burden, constant development of new anti-cancer drugs, temperature-sensitive biologics, and personalized therapies drive logistical complexity.
Pharmaceuticals Lead End-User Segment: Significant investment in drug development, multi-country trials, and high demand for specialist logistics providers; presence in biologics and precision medicine drives cold chain demand.
U.S. Clinical Trial Leadership: The U.S. conducts nearly 40% of all global clinical trials; accounts for 35% of global clinical trial participants.
Global Trial Growth: Registered clinical studies grew from 362,481 in 2020 to 453,803 in 2023, reflecting consistent expansion in global clinical trial activities.
Recruitment Challenges: Approximately 80% of clinical trials experience delays or premature termination due to participant recruitment challenges; 37% of research sites struggle to enroll enough volunteers.
Venture Capital Investment: VC investments in clinical trial startups reached USD 4.2 billion in 2023, focusing on disruptive technologies to streamline clinical research.
Data Dissemination Gap: 94% of interventional clinical studies have posted results, whereas only 6% of observational studies have made results publicly available.
Regulatory Complexity: GMP and GDP compliance requires significant infrastructure investment; automated compliance software, electronic labels, and real-time shipment tracking are increasingly essential.
📄 Get the Insights You Need to Drive Real Impact → https://dimensionmarketresearch.com/request-sample/clinical-trial-supply-and-logistics-market/
🔷 Market Dynamics: Drivers, Restraints, and Strategic Opportunities
Drivers: Increasing Clinical Trials & Regulatory Requirements
The primary driver is the increasing number of clinical trials globally. The escalating burden of chronic diseases-cancer, cardiovascular disease, neurological disease-is creating demand for new drugs and trials. Pharmaceutical and government investments in R&D are increasing the intensity of clinical trials. India, Brazil, and China have become key regions for trial conduct at lower cost, with rich diversity in patient pools and rapid drug approval pathways. Biologic and gene therapy studies expansion is generating demand for high-tech supply options, including temperature-sensitive shipping and storage of drugs.
Simultaneously, stringent regulatory requirements are driving compliance-based logistics solutions. The FDA, European Medicines Agency (EMA), and China's National Medical Products Administration (NMPA) have placed strong requirements for logistics in clinical trials, including cold chain maintenance, serialization, and drug tracking. GMP and GDP compliance necessities have led pharmaceutical companies to partner with experienced logistics providers. Regulatory requirements make drugs safer and more efficient but require high infrastructure and technology investments. Automated compliance software, electronic labels, and real-time shipment tracking have become increasingly adopted.
Restraints: High Cold Chain Costs & Supply Chain Disruptions
Despite momentum, significant barriers remain. High costs associated with cold chain logistics represent a major restraint. Cold chain logistics is imperative for gene therapies, vaccines, and biologics but costly in terms of temperature controls. Strong demand for temperature tracking tools, secure stores, and specialized packaging carries high operational costs. Temperature deviation risks cause degradation and financial loss. Significant infrastructure investment in ultra-low temperature freezers and real-time tracking tools is required. Cold chain logistics is particularly challenging for CROs and small pharmaceutical companies with lesser financial capacities.
Additionally, supply chain disruptions and geopolitical risks challenge clinical trial logistics. Global supply chain disruptions-including during the COVID-19 pandemic, political unrest, and trade restrictions-have delayed drug deliveries, caused raw material shortages, and created compliance barriers impacting timelines. Varying transportation costs and manpower shortages make logistics planning even more challenging. Pharmaceutical companies and logistics providers must apply contingency planning, including diversifying providers and leveraging regional distribution platforms, to counter such barriers.
Opportunities: Emerging Markets & Personalized Medicine Expansion
Pharmaceutical companies have increasingly conducted trials in emerging economies across Asia-Pacific, Latin America, and the Middle East for cost savings and access to treatment-untreated patient populations. China and India have effective regulatory processes and high recruitment rates, with governments investing in infrastructure and reforming legislation to attract more clinical studies. Logistics providers with strong regional presence can benefit from increased demand for clinical trial supply chain management, including import/export compliance, cold storage, and last-mile delivery.
The transition toward personalized therapy, including cell and gene therapies, creates new avenues for clinical trial supply and logistics. Unlike conventional drugs, these therapies require custom-made supply capabilities, including ultra-cold storage and rapid delivery to clinic settings. Logistics providers are investing in capabilities including cryogenic shipping and just-in-time delivery platforms to serve complex therapeutics. As precision therapy continues to expand, companies with flexible, adaptable supply capabilities will gain competitive market advantage.
🔷 Selective Segmentation: Where the Growth is Concentrated
By Service (Logistics & Distribution-27.1% Share): Logistics and distribution dominate due to their critical role in timely and secure delivery of investigational drugs, medical devices, and bio-samples across multi-country trials. Cold chain logistics is a strong growth catalyst, with biologics, gene and cell therapies, and vaccines requiring temperature-controlled environments. GPS tracking, IoT sensors, and blockchain investments have become necessary for transparency and supply chain disruption prevention. Adherence to Good Distribution Practice (GDP) directives governs careful handling protocols. The growing use of decentralized clinical trials (DCTs) with direct-to-patient (DTP) distribution channels further strengthens this segment.
By Phase (Phase I-Leading Share): Phase I trials have a significant presence due to high demand for complex supply chain offerings and their critical role in drug development. Phase I studies involve small patient groups but require high-control environments. The difficulty lies in providing rapid drug development cycles, just-in-time production, and delivering investigational drugs under stringent protocols. Cold chain logistics and ultra-low temperature storage become dominating issues for biologics and gene therapies. Approximately 30-40% of drugs in Phase I fail due to efficacy or safety concerns, creating constant resupply demand. Phase II and Phase III represent significant secondary segments with larger patient populations and longer durations.
By Therapeutic Area (Oncology-Dominant Share): Oncology is the dominant player due to high disease burden and constant development of new anti-cancer drugs. The multi-faceted development of oncology drugs-chemotherapy, immunotherapy, targeted therapies, gene therapies-creates complex, specialized supply infrastructure. Temperature-sensitive biologics and personalized therapies require ultra-cold temperatures for shipping and storage. Decentralized clinical trials (DCTs) in oncology have increased direct-to-patient (DTP) delivery models, reducing patient burden. Regulatory complexity, with critical approval mechanisms for anti-cancer drugs, requires adherence to both GMP and GDP protocols. Cardiovascular Diseases, CNS Disorders, and Respiratory Diseases represent significant secondary therapeutic areas.
By End-User (Pharmaceuticals-Dominant Share): Pharmaceutical companies represent the largest end-user industry with significant drug development investment, multi-country trials, and high demand for specialist logistics providers. The pharmaceutical sector carries out most global clinical trials, creating demand for efficient supply chains, regulatory compliance, and cold chain logistics. Pharmaceutical companies require full-service logistics including production, packaging, labeling, and distribution. Presence in biologics, biosimilars, and precision medicine drives cold chain demand. Emerging trends in DTP and decentralized trials are driving pharmaceutical supply chains toward innovation. Biologicals represent the fastest-growing end-user segment, driven by cell and gene therapy expansion. Medical Devices represent a smaller but steady segment.
📄 Get the Full Premium Report Now- https://dimensionmarketresearch.com/checkout/clinical-trial-supply-and-logistics-market/
🔷 Regional Analysis: North America Leads, Asia-Pacific Emerges as Fastest-Growing
North America (37.4% Revenue Share in 2025): North America is in a commanding position in the clinical trial supply and logistics sector, with a combination of several strategic factors. North America has a high density of pharmaceutical and biotechnology industries, creating a strong environment for clinical trials to flourish. Strong infrastructure and state-of-the-art technological capabilities maximize supply chain efficiency and ensure timely delivery of clinical materials. The U.S., through the FDA, puts in place definite guidelines, streamlines trial processes, and opens doors for increased in-country trials. North America has high R&D investments, backed by supportive government programs and drug development funding. Established healthcare infrastructure and a high patient pool make recruitment and patient maintenance easier. Alliances between key industry leaders, CROs, and logistics providers maximize capabilities for managing complex regulatory environments and optimizing supply chains.
The U.S. Market (USD 1,258.4 million in 2025, 7.4% CAGR): The U.S. market is the largest globally, driven by a strong pharmaceutical sector, leading contract research organizations (CROs), and high R&D investments. The U.S. conducts nearly 40% of all global clinical trials, supported by the FDA's well-structured regulatory framework. With rising trials and emerging demand for personalized therapy, the market will expand steadily. Increased use of biologics, cell and gene therapies has grown demand for high-tech supply chain offerings, namely cold chain logistics. A significant advantage of the U.S. market is high demographic diversity, enabling effective subject recruitment across ethnic groups for improved drug integrity and efficacy testing. The market is also benefiting from technological innovation, including AI-facilitated logistics, blockchain-facilitated drug tracking, and computerized inventory management software.
Europe: Europe's market is driven by strong regulatory frameworks from the European Medicines Agency (EMA), the EU Clinical Trials Regulation, and established pharmaceutical and biotech hubs in Germany, the UK, France, and Switzerland. The region's focus on supply chain transparency and patient-centric trial models supports adoption of advanced logistics solutions.
Asia-Pacific (Fastest-Growing Region): Asia-Pacific is emerging as the fastest-growing region, driven by pharmaceutical companies conducting trials in emerging economies for cost savings and access to large, treatment-untreated patient populations. China and India have effective regulatory processes and high recruitment rates, with governments investing in infrastructure and reforming legislation to attract more clinical studies. The region's well-established manufacturing capacity for pharmaceuticals and biologicals, increasing CRO presence, and government initiatives supporting clinical research accelerate market growth.
🔷 Competitive Landscape: Global Leaders, Specialized Logistics Providers, and Technology Innovators
The clinical trial supply and logistics sector features prominent global logistics providers, specialized CROs, and technology-enabled supply chain innovators.
Global Logistics and CRO Leaders: Thermo Fisher Scientific, Catalent, Inc., Parexel International, Almac Group, Marken (a UPS company), Piramal Pharma Solutions, UDG Healthcare, DHL, FedEx, Movianto, and Ceva Logistics dominate with extensive global networks, cold chain capabilities, and regulatory expertise. Thermo Fisher Scientific partnered with Pfizer (January 2025) to enhance supply chain transparency through blockchain integration, improving drug traceability and minimizing counterfeiting risks. Catalent, Inc. announced a USD 150 million investment in cold chain infrastructure expansion (December 2024), focusing on temperature-controlled storage and distribution for biologics, cell and gene therapies, and mRNA-based treatments. Parexel International acquired a minority stake in a leading Indian logistics provider (October 2024) to strengthen its presence in Asia-Pacific.
Specialized Service Providers: Packaging Coordinators Inc. and Clinevo Technologies focus on packaging, labeling, blinding, and comparator sourcing services essential for clinical trial supply integrity.
Recent Developments Highlighting Market Momentum:
January 2025: Thermo Fisher Scientific partnered with Pfizer to enhance supply chain transparency through blockchain integration.
December 2024: Catalent, Inc. announced a USD 150 million investment in cold chain infrastructure expansion for biologics and cell and gene therapies.
November 2024: DHL International GmbH showcased IoT-enabled tracking solutions at the Global Clinical Trials Expo in Berlin.
October 2024: Parexel International acquired a minority stake in a leading Indian logistics provider to strengthen Asia-Pacific presence.
September 2024: The World Clinical Trials Conference in London emphasized AI's role in clinical supply chain optimization.
August 2024: Almac Group collaborated with Moderna to accelerate vaccine trial logistics for temperature-sensitive materials.
🔷 The Road Ahead: What Decision-Makers Need to Know
For B2B decision-makers-pharmaceutical supply chain executives, CRO operations directors, logistics providers, and clinical trial sponsors-the strategic imperative is clear: clinical trial supply and logistics has moved from a back-office function to a strategic differentiator. The 7.9% CAGR reflects sustained demand driven by trial complexity, biologic expansion, and decentralized trial adoption.
Key strategic imperatives include:
Invest in cold chain logistics capabilities. With biologics, cell and gene therapies, and mRNA treatments expanding, ultra-low temperature storage, cryogenic shipping, and real-time temperature monitoring are essential for drug integrity.
Adopt AI-powered predictive analytics and IoT-enabled tracking. AI improves inventory forecasting, reduces drug wastage, and prevents shortages. IoT sensors enable real-time temperature and location tracking for regulatory compliance.
Expand into decentralized and direct-to-patient trial logistics. DCTs and DTP delivery models are growing rapidly, requiring flexible, patient-centric distribution frameworks with real-time tracking and remote monitoring.
Leverage blockchain for supply chain transparency and compliance. Blockchain offers enhanced traceability, authenticates drug genuineness, and provides secure, unalterable transaction records to satisfy regulatory requirements.
Expand into emerging markets strategically. Asia-Pacific offers the highest growth potential, with cost savings, high patient pools, and rapid approval processes in China and India. Local partnerships and regulatory expertise are essential.
The full report from Dimension Market Research provides granular segmentation by service (logistics & distribution, storage & retention, packaging/labeling/blinding, manufacturing, comparator sourcing), phase (Phase I, II, III, IV), therapeutic area (oncology, cardiovascular diseases, respiratory diseases, CNS and mental disorders), end-user (pharmaceuticals, biologicals, medical devices), and 20+ regional markets, offering actionable intelligence for strategic planning.
📄 Explore the Report with TOC → https://dimensionmarketresearch.com/report/clinical-trial-supply-and-logistics-market/
For Sales or Inquiries, Contact
Robert John
957 Route 33, Suite 12 #308 Hamilton Square, NJ-08690 USA
Email: enquiry@dimensionmarketresearch.com
United States: (+1 732 369 9777)
Tel No: +91 88267 74855
Dimension Market Research (DMR) is a market research and consulting firm based in India & US, with its headquarters located in the USA. The company believes in providing the best and most valuable data to its customers using the best resources and analysts to work on, to create unmatchable insights into the industries and markets while offering in-depth results of over 30 industries, and all major regions across the world. We also believe that our clients don't always want what they see, so we provide customized reports as well, as per their specific requirements, to create the best possible outcomes for them and enhance their business through our data and insights in every possible way.
This release was published on openPR.
Permanent link to this press release:
Copy
Please set a link in the press area of your homepage to this press release on openPR. openPR disclaims liability for any content contained in this release.
You can edit or delete your press release Clinical Trial Supply and Logistics Market to Reach $7.93 Billion by 2034 as Decentralized Trials and Cold Chain Demands Reshape Drug Development Infrastructure here
News-ID: 4510994 • Views: …
More Releases from Dimension Market Research
Cellulose Insulation Market to Reach $8.7 Billion by 2034 as Energy-Efficient Bu …
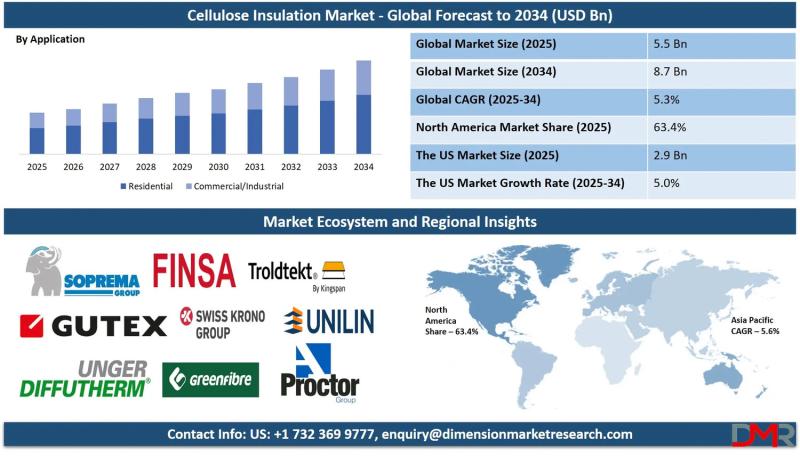
The global Cellulose Insulation Market is on a steady growth trajectory, with market valuation projected to rise from an estimated USD 5.5 billion in 2025 to USD 8.7 billion by 2034, registering a compound annual growth rate (CAGR) of 5.3%. According to Dimension Market Research, this expansion is being driven by three converging forces: stringent energy efficiency regulations and building codes across developed economies, rising consumer demand for sustainable and…
Healthy Snacks Market to Reach $212.2 Billion by 2035 as Clean-Label Demand and …
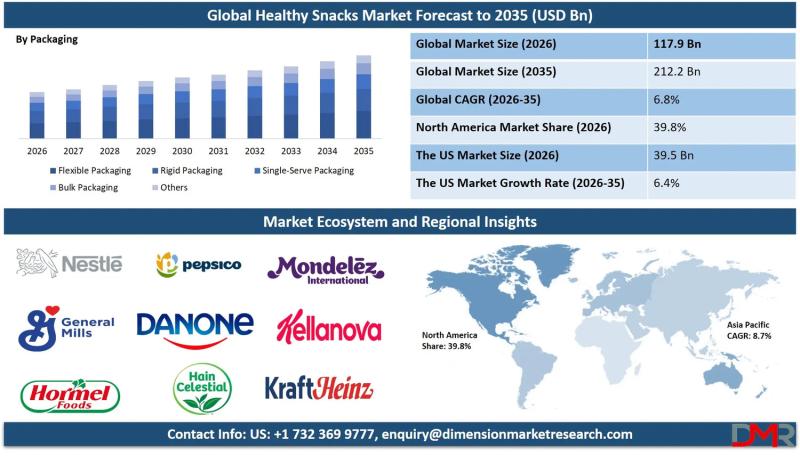
The global Healthy Snacks Market is on a steady growth trajectory, with market valuation projected to rise from an estimated USD 117.9 billion in 2026 to USD 212.2 billion by 2035, registering a compound annual growth rate (CAGR) of 6.8%. According to Dimension Market Research, this expansion is being driven by three converging forces: surging consumer demand for clean-label and functional ingredients, government regulations mandating transparent front-of-pack nutrition labeling, and…
Varicose Vein Treatment Market to Reach $3.8 Billion by 2034 as Minimally Invasi …
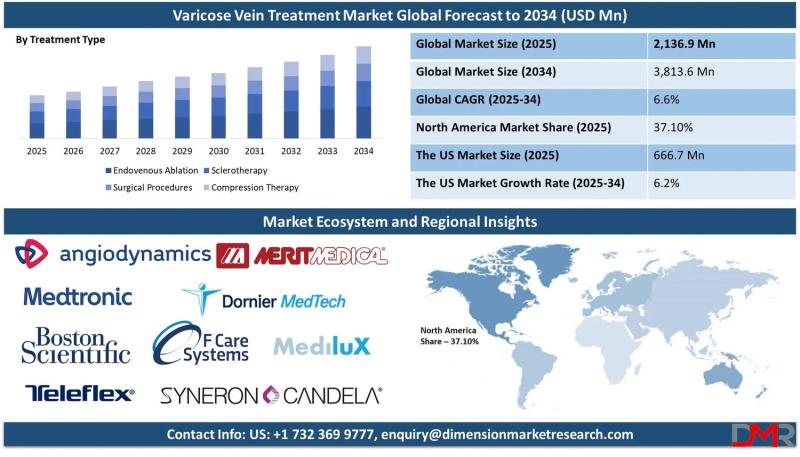
The global Varicose Vein Treatment Market is on a steady growth trajectory, with market valuation projected to rise from an estimated USD 2,136.9 million in 2025 to USD 3,813.6 million by 2034, registering a compound annual growth rate (CAGR) of 6.6%. According to Dimension Market Research, this expansion is being driven by three converging forces: the rising global prevalence of varicose veins and chronic venous insufficiency, the rapid adoption of…
Embroidery Market to Reach $8.0 Billion by 2034 Driven by Smart Textiles and Per …
According to Dimension Market Research, the market is set to grow from USD 3.3 billion in 2025 to USD 8.0 billion by 2034, with the US alone contributing USD 2.2 billion by the end of the forecast period.
A new comprehensive analysis reveals that the global embroidery market is accelerating at a 10.3% compound annual growth rate, powered by two converging forces: consumer demand for hyper-personalized apparel and the rapid digitization…
More Releases for Logistics
Cold Chain Market 2021 Precise Outlook- AmeriCold Logistics, Nichirei Logistics …
The Global Cold Chain Market provides a unique tool for evaluating the market, highlighting opportunities, and supporting strategic and tactical decision-making. This report recognizes that in this rapidly-evolving and competitive environment, up-to-date marketing information is essential to monitor performance and make critical decisions for growth and profitability. The report covers the post-COVID-19 (Corona Virus) impact on various regions and major countries and on the future development of the industry is…
Food Logistics Market Worth Observing Growth | Trinity Logistics, AmeriCold Logi …
The latest launched report on Global Food Logistics Market delivers a transformation framework to understand how megatrends affect industry growth, taking into account the major disrupting forces creating uncertainties for every organisation in the Food Logistics. Based on these outcomes, HTF MI outline plan for these volatile scenarios considering companies such as Port Jersey Logistics, DB Schenker, Henningsen Cold Storage, R2 Logistics, AN Deringer, ODW Logistics, Matson Logistics, Allen Lund,…
Logistics Market Future Outlook | AmeriCold Logistics, Nichirei Logistics, Linea …
WiseGuyRerports.com Presents “Global Logistics Market Size, Status and Forecast 2020-2026” New Document to its Studies Database
The extensive market study presents a complete analysis of the global Logistics market, including the latest developments, current market conditions, and the growth potentialities during the review period. Accurate statistics with regard to the product, methods as well as the share belonging to the key businesses in the market are also given in the report.…
Cold Chain Logistics Market Size 2020 by Top Key Players | Nichirei Logistics Gr …
Cold Chain Logistics Market report' to its research archive which allows exploring the industry in a 360-degree view while marking major insights and highlights accelerating the Cold Chain Logistics market trends. The data sourced from the report enables formulating business plans and helps decision making to improve profitability. The Cold Chain Logistics market has been witnessing consistent growth over the last few years and is projected to continue growing with…
Healthcare Cold Chain Logistics Market 2020 Business Scenario - AmeriCold Logist …
The Global Healthcare Cold Chain Logistics Market will increase at a compound annual growth rate of 10.09% (approx.) between 2020 and 2026.
The Healthcare Cold Chain Logistics Market report is a collection of pragmatic information, quantitative and qualitative estimation by industry experts, the contribution from industry connoisseurs and industry accomplices across the value chain. Furthermore, the report also provides the qualitative results of diverse market factors on its geographies and segments.…
Third-Party Logistics Market: Leading Players – XPO Logistics , Yusen Logistic …
Third-Party Logistics Market report provides key information about the industry, including invaluable facts and figures, expert opinions, and the latest developments across the globe. The Report also calculate the market size, the report considers the revenue generated from the sales of This Report and technologies by various application segments.
Download Sample PDF copy of this report https://supplydemandmarketresearch.com/home/contact/272389?ref=Sample-and-Brochure&toccode=SDMRSE272389&utm_source=s.
The study objectives of this report are:
To analyze global Third-Party Logistics status, future forecast,…